Advanced Synthetic Route for High Optical Purity Valsartan Intermediates and Commercial Scalability
The pharmaceutical industry's relentless pursuit of high-purity antihypertensive agents has led to significant advancements in the synthesis of Valsartan, a critical angiotensin II receptor blocker. Patent CN1844110B introduces a transformative methodology for producing N-[[2'-(1H-tetrazol-5-yl)-(1,1'-diphenyl)-4-yl]-methyl]-L-valine with exceptional optical purity. This technical breakthrough addresses long-standing challenges in chiral stability and process safety, offering a robust alternative to legacy manufacturing protocols. By shifting from hazardous reductive amination or risky alkaline hydrolysis to a controlled acidic deprotection strategy, this innovation ensures that the final Active Pharmaceutical Ingredient (API) meets stringent enantiomeric excess specifications. For global supply chain leaders, this represents a pivotal opportunity to secure a more reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Valsartan has been plagued by complex purification hurdles and safety concerns associated with early synthetic routes. As illustrated in prior art such as Scheme 1 (US 5,399,578), traditional methods often relied on the use of trialkyltin azide at elevated temperatures (120-140°C) to construct the tetrazole ring, introducing severe toxicity risks and complicating waste management. Furthermore, alternative pathways like those described in WO 05/049586 utilized stepwise deprotection strategies where the removal of the carboxyl protecting group under alkaline conditions frequently triggered racemization. This loss of chiral integrity not only diminished the overall yield—sometimes dropping to as low as 50% in deprotection steps—but also necessitated costly recrystallization processes to restore optical purity. The reliance on catalytic hydrogenation equipment in other variants further inflated capital expenditure and operational complexity, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates.
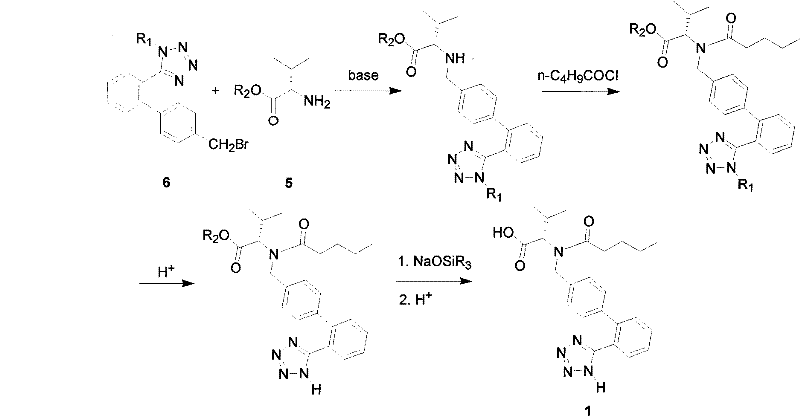
The Novel Approach
The methodology disclosed in CN1844110B fundamentally reengineers the deprotection and acylation sequence to preserve stereochemical integrity. Instead of exposing the sensitive chiral center to basic conditions, the novel process employs a tetrazole-protected 2'-tetrazolyl-4-bromomethylbiphenyl coupled with esterified L-valine via nucleophilic substitution. The critical innovation lies in the subsequent acidic deprotection step, where both the tetrazole and carboxyl protecting groups are removed using inorganic acids like hydrochloric or hydrobromic acid. This acidic environment effectively suppresses the racemization mechanisms that plague alkaline hydrolysis, routinely delivering intermediates with optical purity exceeding 97% and often reaching 99%. Additionally, the final acylation with n-valeryl chloride is conducted in the presence of lower aliphatic alcohols at a controlled pH of 1.5 to 5.0, a subtle yet powerful modification that prevents acid chloride hydrolysis while maintaining the chiral configuration of the valine moiety.
Mechanistic Insights into Acidic Deprotection and Racemization Suppression
The core chemical advantage of this patent lies in the mechanistic stability of the L-valine stereocenter under acidic versus basic conditions. In conventional alkaline hydrolysis, the alpha-proton adjacent to the carbonyl group becomes susceptible to abstraction, leading to the formation of an enolate intermediate that can reprotonate from either face, resulting in racemization. By utilizing strong inorganic acids for deprotection, the reaction medium remains proton-rich, preventing the formation of the reactive enolate species and locking the chiral center in its L-configuration. This mechanistic control is further enhanced during the final acylation step, where the addition of methanol or ethanol acts as a kinetic stabilizer. The alcohol molecules likely coordinate with the reaction intermediates or modify the solvation shell around the chiral amine, creating a steric and electronic environment that disfavors inversion. This dual-strategy of acidic deprotection followed by alcohol-mediated acylation ensures that the high-purity pharmaceutical intermediates produced are virtually free from the D-enantiomer impurities that compromise therapeutic efficacy.
From an impurity profile perspective, this route significantly simplifies the downstream purification landscape. Traditional methods often generate substantial amounts of n-valeric acid due to the hydrolysis of n-valeryl chloride in aqueous or uncontrolled conditions, which co-crystallizes with the product and is difficult to separate. The patented method's strict pH control (1.5-5.0) combined with the alcohol additive minimizes this hydrolysis side reaction. Consequently, the crude product exhibits higher assay values (often >95% by HPLC) and superior optical purity (>96% ee) directly from the reaction mixture. This reduction in impurity load translates to fewer crystallization cycles, lower solvent consumption, and a dramatically improved mass balance, which are critical metrics for R&D directors evaluating process viability.
How to Synthesize Valsartan Intermediate Efficiently
The synthesis protocol outlined in the patent offers a pragmatic pathway for manufacturing teams to implement immediately. The process begins with the nucleophilic substitution of the bromomethyl biphenyl derivative with L-valine ester in the presence of an organic base such as diisopropylethylamine, typically conducted at mild temperatures (0-10°C) to minimize side reactions. Following isolation of the protected intermediate, the material undergoes acidic hydrolysis using mixed solvents like ethylene glycol dimethyl ether and hydrochloric acid, which can be performed as a one-pot reaction to maximize throughput. The final acylation step requires precise addition of n-valeryl chloride and base to maintain the critical pH window, ensuring the preservation of optical activity. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform nucleophilic substitution between tetrazole-protected 2'-tetrazolyl-4-bromomethylbiphenyl and esterified L-valine using an organic base.
- Execute a one-pot or stepwise acidic deprotection to remove both tetrazole and carboxyl protecting groups without racemization.
- Condense the resulting free acid with n-valeryl chloride in the presence of lower aliphatic alcohols while strictly controlling pH between 1.5 and 5.0.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere technical elegance. By eliminating the need for specialized hydrogenation equipment and toxic organotin reagents, the process drastically reduces the barrier to entry for contract manufacturing organizations, thereby expanding the pool of qualified suppliers. The simplified workflow, characterized by fewer unit operations and the avoidance of cryogenic conditions or high-pressure reactors, leads to substantial cost savings in both capital investment and daily operational expenditures. Furthermore, the robustness of the acidic deprotection step ensures consistent batch-to-batch quality, mitigating the risk of production delays caused by failed purity specifications. This reliability is essential for maintaining uninterrupted supply chains for life-saving cardiovascular medications.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like trialkyltin azide removes the need for complex heavy metal removal steps and specialized waste disposal protocols, which are significant cost drivers in traditional synthesis. Additionally, the high optical purity achieved directly from the reaction reduces the number of recrystallization steps required, leading to significant savings in solvent usage and energy consumption. The avoidance of catalytic hydrogenation equipment further lowers capital depreciation costs, allowing for more competitive pricing structures in the final API market.
- Enhanced Supply Chain Reliability: The reliance on readily available industrial raw materials, such as protected bromomethyl biphenyl and L-valine esters, ensures a stable supply base that is less susceptible to geopolitical or logistical disruptions compared to exotic catalysts. The simplified process flow, which avoids sensitive high-pressure or low-temperature steps, enhances the robustness of production schedules, reducing the likelihood of unplanned downtime. This operational stability allows suppliers to offer shorter lead times and more flexible volume commitments to their downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard glass-lined or stainless steel reactors without the need for specialized metallurgy required for handling corrosive tin compounds or high-pressure hydrogen. The reduction in hazardous waste generation, particularly the absence of tin residues and azide byproducts, aligns with increasingly stringent global environmental regulations, simplifying permitting and compliance audits. This eco-friendly profile not only reduces regulatory risk but also enhances the brand reputation of manufacturers committed to sustainable green chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-optical purity synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process capabilities and quality outcomes. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: How does the new acidic deprotection method improve optical purity compared to alkaline hydrolysis?
A: Traditional alkaline deprotection often induces racemization in valsartan intermediates, lowering optical purity. The patented acidic deprotection method (using HCl or HBr) stabilizes the chiral center, consistently achieving optical purity (ee%) above 97%, often exceeding 99%.
Q: Does this synthesis route eliminate the need for toxic tin reagents?
A: Yes. Unlike older methods (e.g., US 5,399,578) that rely on toxic trialkyltin azide for tetrazole formation, this route utilizes pre-protected tetrazole starting materials, significantly improving safety profiles and reducing hazardous waste disposal costs.
Q: What role do lower aliphatic alcohols play in the final acylation step?
A: Adding methanol or ethanol during the reaction with n-valeryl chloride serves a dual purpose: it effectively suppresses racemization of the chiral valine moiety and prevents the hydrolysis of the acid chloride into valeric acid, thereby facilitating easier crystallization and purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven manufacturing capacity. Our facility is equipped to handle diverse synthetic pathways, ranging from early-stage process development to full-scale commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced chiral HPLC and NMR spectroscopy to verify every batch, guaranteeing that our Valsartan intermediates meet the highest global pharmacopeial standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your margin structure. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can drive value for your organization.
