Optimizing Dabigatran Etexilate Production: A Technical Analysis of Patent CN103087046A
Optimizing Dabigatran Etexilate Production: A Technical Analysis of Patent CN103087046A
The pharmaceutical landscape for anticoagulants has been significantly shaped by the introduction of direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule in modern cardiovascular therapy. As global demand for this critical Active Pharmaceutical Ingredient (API) continues to surge, the efficiency and sustainability of its supply chain have become paramount concerns for procurement and R&D leadership alike. Patent CN103087046A presents a transformative synthetic methodology that addresses long-standing bottlenecks in the production of Dabigatran Etexilate intermediates. By strategically substituting costly and hazardous reagents with more accessible alternatives, this innovation not only enhances process safety but also delivers a substantial improvement in overall chemical yield. For stakeholders evaluating reliable dabigatran etexilate intermediate supplier options, understanding the technical nuances of this patent is essential for securing a competitive advantage in the marketplace.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dabigatran Etexilate has been plagued by inefficiencies inherent in the use of specialized coupling agents and unstable intermediates. The prior art, often referenced in early Boehringer Ingelheim filings, relies heavily on N,N'-carbonyldiimidazole (CDI) for the formation of key amide bonds. While chemically effective, CDI is a high-cost reagent that significantly inflates the Bill of Materials (BOM) for large-scale production. Furthermore, the conventional route necessitates the use of volatile acid salts and generates substantial quantities of waste acid solutions during the conversion of intermediates, creating a heavy burden on wastewater treatment facilities and increasing the environmental footprint of the manufacturing process. Perhaps most critically, the traditional pathway involves the synthesis of unstable chloroformate intermediates derived from phosgene or triphosgene, introducing severe safety hazards and requiring specialized containment infrastructure that limits the suitability for industrialized production in many regions.
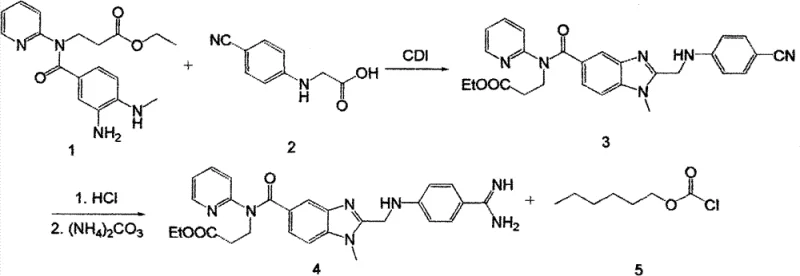
The Novel Approach
In stark contrast to the legacy methods, the methodology disclosed in CN103087046A introduces a robust and economically superior pathway that fundamentally re-engineers the synthesis logic. The most striking innovation is the replacement of the expensive CDI coupling agent with thionyl chloride, a commodity chemical that is not only significantly cheaper but also easier to source globally, thereby mitigating supply chain risks associated with specialty reagents. This substitution simplifies the reaction workup and reduces the complexity of post-treatment procedures. Additionally, the new process employs a sodium ethoxide and ammonium salt system for the cyclization steps, which effectively eliminates the generation of large volumes of waste acid, aligning the process with stricter environmental compliance standards. By utilizing a stable carbamate precursor (Compound 11) instead of the hazardous phosgene-derived species, the method ensures a safer operating environment while simultaneously boosting the total yield from a lackluster 36.6% to an impressive 54.8%, representing a massive gain in material efficiency.
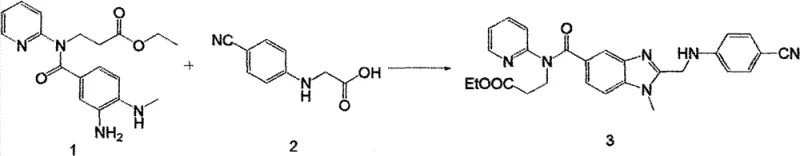
Mechanistic Insights into Thionyl Chloride-Mediated Amidation
The core of this technological breakthrough lies in the mechanistic efficiency of using thionyl chloride (SOCl2) as an activating agent for the carboxylic acid moiety of Compound 2. In this optimized protocol, thionyl chloride reacts with the acid to form a highly reactive acyl chloride intermediate in situ, which is subsequently captured by the amine nucleophile of Compound 1. This transformation is catalyzed by a trace amount of dimethylformamide (DMF), which facilitates the formation of the Vilsmeier-Haack type active species, ensuring rapid and complete conversion under mild reflux conditions. The use of an organic base, such as triethylamine or pyridine, serves a dual purpose: it scavenges the hydrogen chloride byproduct generated during the amidation and maintains the reaction medium at a pH conducive to nucleophilic attack. This mechanistic pathway avoids the formation of imidazole byproducts associated with CDI usage, thereby simplifying the purification profile and reducing the load on downstream chromatography or crystallization steps.
Furthermore, the stabilization of the final coupling step through the use of Compound 11 offers profound implications for impurity control. In traditional routes, the instability of the chloroformate intermediate often leads to hydrolysis and the formation of phenolic impurities that are difficult to separate from the final API. By employing the stable hexyl carbonate derivative (Compound 11), the reaction proceeds via a controlled aminolysis mechanism that minimizes side reactions. The biphasic solvent system utilized in this step, typically comprising tetrahydrofuran and water or ethyl acetate and water, allows for precise temperature control at 0~-5°C, further suppressing thermal degradation pathways. This rigorous control over reaction parameters ensures a cleaner crude product profile, which is critical for meeting the stringent purity specifications required for pharmaceutical intermediates intended for human consumption.
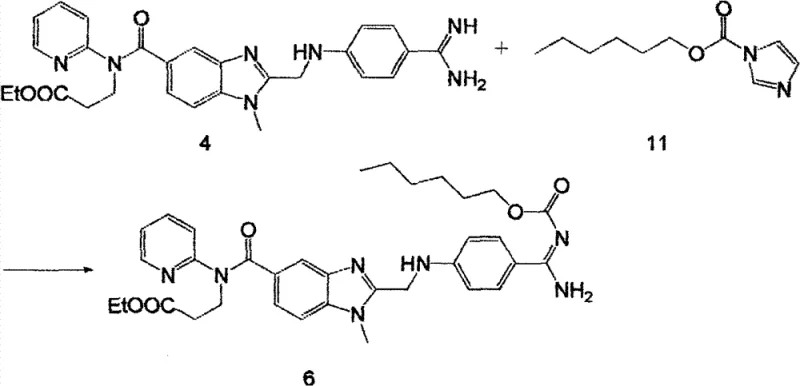
How to Synthesize Dabigatran Etexilate Efficiently
The implementation of this synthetic route requires precise adherence to the optimized conditions described in the patent embodiments to achieve the reported yield enhancements. The process is divided into three distinct stages: the activation and amidation to form Compound 3, the cyclization to form Compound 4, and the final carbamation to yield Dabigatran Etexilate (Compound 6). Each step leverages specific solvent systems and stoichiometric ratios to maximize conversion while minimizing byproduct formation. Operators must pay particular attention to the temperature controls during the addition of thionyl chloride and the final coupling agent, as exothermic events can compromise product quality if not managed correctly. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Activate Compound 2 with thionyl chloride and DMF, then react with Compound 1 in the presence of an organic base to form Compound 3.
- Convert Compound 3 to Compound 4 using sodium ethoxide in ethanol followed by treatment with an ammonium salt.
- React Compound 4 with stable Compound 11 in a biphasic solvent system to yield the final Dabigatran Etexilate (Compound 6).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN103087046A translates directly into tangible strategic benefits that extend beyond simple chemical yield. The shift away from proprietary or high-cost reagents like CDI towards commodity chemicals like thionyl chloride drastically reduces the raw material cost basis, allowing for more competitive pricing structures in long-term supply agreements. Moreover, the elimination of phosgene from the process removes a significant regulatory and logistical hurdle, as phosgene is a scheduled chemical in many jurisdictions requiring extensive security clearance and specialized transport protocols. This simplification of the regulatory landscape accelerates the speed to market and reduces the administrative overhead associated with chemical procurement and inventory management.
- Cost Reduction in Manufacturing: The economic argument for this new process is compelling, driven primarily by the substitution of high-value coupling agents with low-cost commodities. By replacing N,N'-carbonyldiimidazole (CDI) with thionyl chloride, manufacturers can achieve a significant reduction in direct material costs, as thionyl chloride is produced on a massive industrial scale and is readily available at a fraction of the price of CDI. Additionally, the simplified post-treatment procedures, which no longer require the removal of imidazole residues or the neutralization of large volumes of waste acid, lead to reduced utility consumption and lower waste disposal fees. The cumulative effect of these efficiencies results in a leaner manufacturing cost structure that provides substantial cost savings without compromising on product quality or purity.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on stable and widely available starting materials. The conventional dependence on unstable intermediates often necessitates just-in-time manufacturing or complex cold-chain logistics to prevent degradation before use. In contrast, the use of stable Compound 11 allows for the stocking of key intermediates, providing a buffer against supply disruptions and enabling more flexible production scheduling. This stability ensures that production timelines are not held hostage by the shelf-life limitations of reactive species, thereby guaranteeing consistent delivery performance to downstream API manufacturers and reducing the risk of stockouts.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a clear path to sustainable scale-up. The avoidance of phosgene eliminates the need for expensive scrubbing systems and emergency containment protocols, making the technology transferable to a wider range of manufacturing sites, including those in regions with strict safety regulations. Furthermore, the reduction in waste acid generation aligns with green chemistry principles, lowering the environmental impact per kilogram of product. This eco-friendly profile not only reduces compliance costs but also enhances the corporate social responsibility (CSR) credentials of the supply chain, a factor increasingly weighted in vendor selection processes by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in CN103087046A, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement teams assessing vendor capabilities.
Q: How does the new synthesis method improve overall yield compared to prior art?
A: The patented method increases the total yield from 36.6% in conventional routes to 54.8% by replacing expensive reagents like CDI with thionyl chloride and utilizing stable intermediates.
Q: What safety advantages does this route offer regarding phosgene usage?
A: The process eliminates the need for highly toxic phosgene or triphosgene by using a stable Compound 11 for the final carbamation step, significantly enhancing operational safety.
Q: Does this method reduce waste generation?
A: Yes, by adopting a sodium ethoxide and ammonium salt system, the method avoids the generation of large amounts of waste acid solution, simplifying post-treatment and reducing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103087046A are fully realized in a commercial setting. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay. Our commitment to quality assurance means that every batch of Dabigatran Etexilate intermediate is thoroughly vetted to meet the exacting standards required for global regulatory submissions.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this novel route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-quality intermediates into your API manufacturing workflow.
