Advanced Meropenem Synthesis: Eliminating Noble Metal Catalysts for Commercial Scale
Advanced Meropenem Synthesis: Eliminating Noble Metal Catalysts for Commercial Scale
The pharmaceutical landscape for beta-lactam antibiotics is constantly evolving, driven by the need for more efficient and cost-effective manufacturing processes. Patent CN101348486B, filed in 2010, introduces a groundbreaking preparation method for Meropenem, a broad-spectrum carbapenem antibiotic critical for treating severe bacterial infections. This technology addresses long-standing inefficiencies in traditional synthesis routes by eliminating the dependency on scarce and expensive Noble Metal Rhodium catalysts. For R&D directors and procurement specialists, this represents a pivotal shift towards a more sustainable and economically viable supply chain for high-purity API intermediates. The disclosed method streamlines the production into a concise six-step sequence, starting from readily available azetidinone precursors, ensuring robust scalability and reduced environmental impact.
The significance of this patent lies not just in the chemical transformation but in its strategic alignment with green chemistry principles and cost reduction imperatives. By replacing the traditional Rhodium-catalyzed cyclization with an organophosphorus-mediated Wittig reaction, the process mitigates the risks associated with heavy metal contamination and volatile precious metal pricing. This technical advancement offers a reliable meropenem intermediate supplier pathway that is both chemically elegant and commercially superior. As global demand for carbapenems rises, adopting such innovative synthetic strategies becomes essential for maintaining competitive advantage in the generic pharmaceutical market. The following analysis dissects the mechanistic nuances and commercial implications of this rhodium-free methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
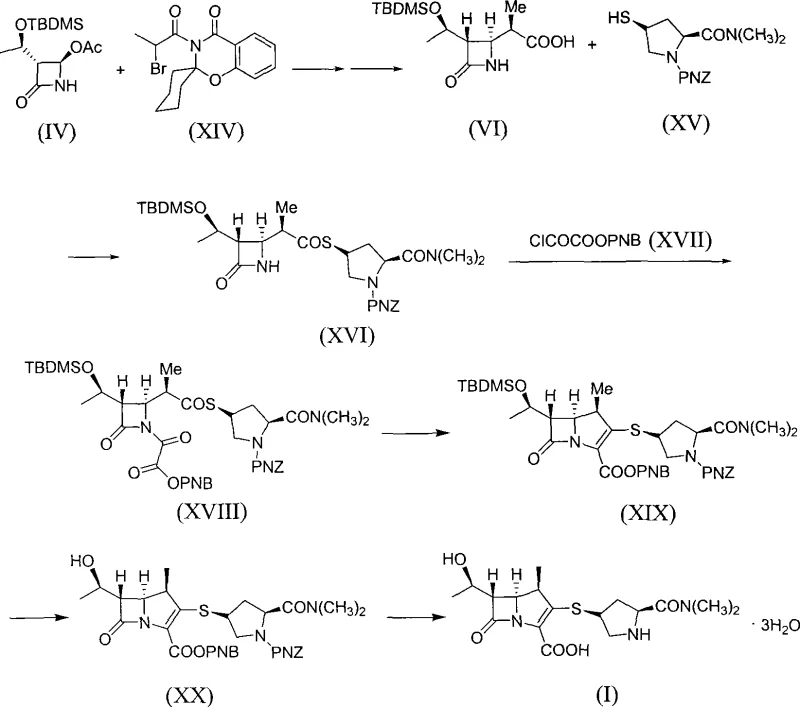
Historically, the industrial synthesis of beta-methyl carbapenem antibiotics has been dominated by routes established by major pharmaceutical entities like Sumitomo and Lederle. These conventional pathways, while effective, suffer from inherent structural inefficiencies that hinder optimal manufacturing economics. As illustrated in prior art such as USP4933333 and USP4990613, these methods typically involve lengthy synthetic sequences exceeding seven steps, necessitating multiple isolation and purification stages for chiral intermediates. A critical bottleneck in these traditional approaches is the reliance on Noble Metal Rhodium complexes to facilitate the key cyclization reaction required to form the carbapenem nucleus. The scarcity and exorbitant cost of Rhodium create a significant financial burden, while the presence of residual heavy metals poses stringent regulatory challenges for final API purity. Furthermore, the atom economy of these older routes is often suboptimal, generating substantial chemical waste and requiring complex solvent recovery systems that inflate operational expenditures.
The Novel Approach
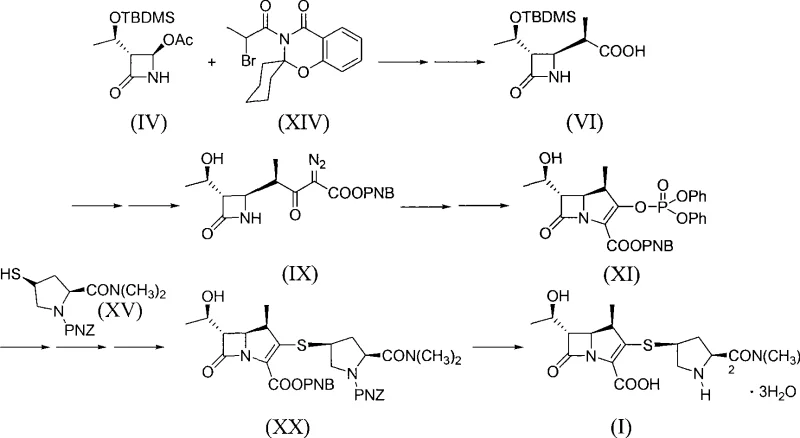
In stark contrast to the legacy methodologies, the process detailed in CN101348486B offers a streamlined alternative that fundamentally reengineers the cyclization step. This novel approach successfully bypasses the need for Rhodium catalysts entirely, substituting them with accessible organophosphorus reagents like triethyl phosphite. The synthetic route is condensed into six distinct steps, starting from compound (IV) and proceeding through a series of highly selective transformations to yield Meropenem (I). By integrating a 'one-pot' philosophy where possible, such as the direct use of crude intermediates in the amide coupling and activation steps, the method drastically reduces processing time and solvent usage. The elimination of noble metals not only lowers the direct material cost but also simplifies the downstream purification process, as there is no need for specialized metal scavenging resins or extensive washing protocols. This results in a cleaner impurity profile and a more robust process capable of meeting the rigorous quality standards demanded by international regulatory bodies for antibiotic production.
Mechanistic Insights into Organophosphorus-Mediated Cyclization
The cornerstone of this innovative synthesis is the intramolecular Wittig reaction utilized to construct the carbapenem double bond, a step that traditionally relied on transition metal catalysis. In this protocol, the precursor compound (XVIII), containing a leaving group at the C1 position and a ketone functionality, is treated with a phosphorus reagent such as triethyl phosphite in the presence of a phenolic catalyst like resorcinol. This reaction generates a phosphorus ylide intermediate in situ, which subsequently undergoes cyclization to form the unsaturated bicyclic system of compound (XIX). The choice of resorcinol as a catalyst is particularly ingenious, as it facilitates the reaction under mild thermal conditions (reflux in xylene) while maintaining high stereoselectivity for the desired trans-configuration. This mechanistic pathway ensures that the critical C5-C6 stereochemistry is preserved throughout the ring closure, yielding a product with HPLC purity exceeding 97% after simple recrystallization. The avoidance of harsh basic conditions often associated with classical Wittig reagents further protects the sensitive beta-lactam ring from hydrolytic degradation.
Following the cyclization, the process employs a highly efficient deprotection and hydrogenation strategy to finalize the API structure. The silyl ether protecting group on the hydroxyethyl side chain is cleaved using fluoride sources like tetrabutyl ammonium fluoride (TBAF) or ammonium bifluoride, conditions that are orthogonal to the ester and amide functionalities present in the molecule. The final transformation involves the catalytic hydrogenation of the p-nitrobenzyl (PNB) and p-nitrobenzyloxycarbonyl (PNZ) protecting groups using Palladium on Carbon (Pd/C). This step is conducted under moderate hydrogen pressure (1.8 MPa) in a mixed solvent system of THF and water, buffered by organic bases like 2,6-lutidine to maintain pH stability. The use of Pd/C, a ubiquitous and inexpensive catalyst, stands in sharp contrast to the Rhodium systems of the past, offering a dramatic reduction in catalyst cost while ensuring complete removal of protecting groups to yield the final Meropenem trihydrate with high optical purity. This sequence demonstrates a masterful balance of reactivity and selectivity, ensuring that the fragile beta-lactam core remains intact throughout the vigorous chemical transformations.
How to Synthesize Meropenem Efficiently
The synthesis of Meropenem via this rhodium-free pathway offers a practical blueprint for manufacturers seeking to optimize their production lines. The process begins with the zinc-mediated coupling of azetidinone (IV) and bromo-propionic amide (XIV) to establish the side chain, followed by amidation with the thiol fragment (XV). Subsequent activation with chlorooxalic acid ester sets the stage for the critical ring closure. Detailed operational parameters, including specific temperature ranges such as -10 to 0°C for the activation step and reflux conditions for the cyclization, are critical for maximizing yield and minimizing byproduct formation. The standardized synthesis steps outlined below provide a comprehensive guide for replicating this high-efficiency route in a GMP-compliant environment, ensuring consistent quality from kilogram to tonne scale.
- React compound (IV) with bromo-propionic amide (XIV) using Zinc powder to form carboxylic acid (VI).
- Couple acid (VI) with thiol side-chain (XV) using CDI or DCC to generate amide (XVI).
- Activate the nitrogen with chlorooxalic acid p-nitrobenzyl ester (XVII) to produce intermediate (XVIII).
- Perform intramolecular Wittig cyclization using triethyl phosphite and resorcinol to close the carbapenem ring (XIX).
- Deprotect the silyl ether using TBAF or ammonium bifluoride to yield precursor (XX).
- Execute catalytic hydrogenation using Palladium/Carbon to remove protecting groups and finalize Meropenem (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the raw material portfolio; by removing Noble Metal Rhodium from the bill of materials, companies can insulate themselves from the geopolitical and market volatility associated with precious metals. This shift allows for more predictable budgeting and long-term cost planning, as organophosphorus reagents and palladium catalysts are commoditized chemicals with stable supply chains. Furthermore, the reduction in synthetic steps from seven or more down to six directly correlates with improved throughput and reduced labor costs per kilogram of API produced. The ability to telescope multiple steps without intermediate purification significantly cuts down on solvent consumption and waste disposal fees, aligning production with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of the expensive Rhodium catalyst is the most significant driver of cost savings in this process. Rhodium is not only costly to purchase but also incurs additional expenses for recovery and recycling infrastructure. By switching to a phosphorus-based cyclization, the direct material cost is substantially lowered. Additionally, the high yields reported in the patent examples, such as the 95.6% yield in the activation step and 92% in the deprotection step, indicate a highly efficient process that minimizes raw material waste. The use of recyclable solvents like toluene and ethyl acetate further enhances the economic profile, allowing for solvent recovery loops that reduce overall operating expenses.
- Enhanced Supply Chain Reliability: Dependence on single-source or limited-supply catalysts creates vulnerability in the supply chain. This novel method utilizes widely available reagents such as zinc powder, triethyl phosphite, and palladium on carbon, all of which are produced by multiple global suppliers. This diversification of the supply base ensures continuity of supply even during market disruptions. Moreover, the simplified purification requirements mean that production bottlenecks associated with complex chromatography or crystallization steps are minimized, leading to shorter lead times and faster turnaround for batch completion. This reliability is crucial for meeting the demands of large-scale generic drug contracts where timely delivery is paramount.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like reflux, filtration, and hydrogenation that are easily transferable from pilot plant to commercial manufacturing. The reduction in the number of isolation steps decreases the physical footprint required for production and lowers the energy intensity of the process. From an environmental perspective, the avoidance of heavy metal residues simplifies wastewater treatment and reduces the ecological footprint of the manufacturing site. This compliance with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, making it a more attractive partner for environmentally conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-free Meropenem synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the feasibility and advantages of this modern approach. Understanding these details is essential for technical teams evaluating the transition from legacy processes to this optimized pathway.
Q: Why is eliminating the Rhodium catalyst significant for Meropenem production?
A: Traditional routes rely on expensive Noble Metal Rhodium catalysts for the cyclization step, which drastically increases raw material costs and introduces supply chain volatility due to metal scarcity. This patent replaces Rhodium with common organophosphorus reagents, significantly lowering production costs and simplifying metal residue control.
Q: How does this 6-step route compare to conventional Sumitomo or Lederle methods?
A: Conventional methods often require 7 or more steps with multiple chiral separations and harsh conditions. This novel approach condenses the synthesis into 6 efficient steps, utilizes easily obtainable raw materials, and avoids complex intermediate purifications, thereby improving atom economy and overall operational simplicity.
Q: What are the key purification advantages of this specific synthetic pathway?
A: The process is designed for telescoping, where crude products from steps like the amide coupling and oxalyl activation can be directly used in subsequent reactions without further purification. Only the critical cyclization intermediate (XIX) requires recrystallization, which streamlines the workflow and reduces solvent consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to remain competitive in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the methodology presented in CN101348486B and possesses the technical capability to implement this rhodium-free route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and robust. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring stringent purity specifications, guaranteeing that every batch of Meropenem intermediate meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific supply chain needs. By leveraging our expertise in process optimization and cost engineering, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this noble-metal-free protocol. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can serve as your strategic partner in delivering high-quality, cost-effective antibiotic intermediates for the global healthcare industry.
