Advanced Manufacturing of Bosutinib Intermediates: A Technical Breakthrough for Scalable Production
Advanced Manufacturing of Bosutinib Intermediates: A Technical Breakthrough for Scalable Production
The pharmaceutical industry continuously seeks robust synthetic pathways for potent kinase inhibitors, and the preparation of Bosutinib (SKI-606) represents a critical area of focus for oncology drug development. Patent CN101792416A introduces a revolutionary preparation process that fundamentally restructures the synthetic approach to this complex quinoline-3-carbonitrile derivative. By shifting the starting material to vanillic acid, a widely available and cost-effective industrial chemical, this methodology addresses the longstanding economic and technical bottlenecks associated with earlier synthetic routes. The innovation lies not merely in the substitution of reagents but in the holistic optimization of reaction conditions, specifically achieving cyclization under mild room temperature environments rather than the extreme thermal or cryogenic conditions previously deemed necessary. This technical advancement paves the way for a more sustainable and economically viable supply chain for high-purity pharmaceutical intermediates.
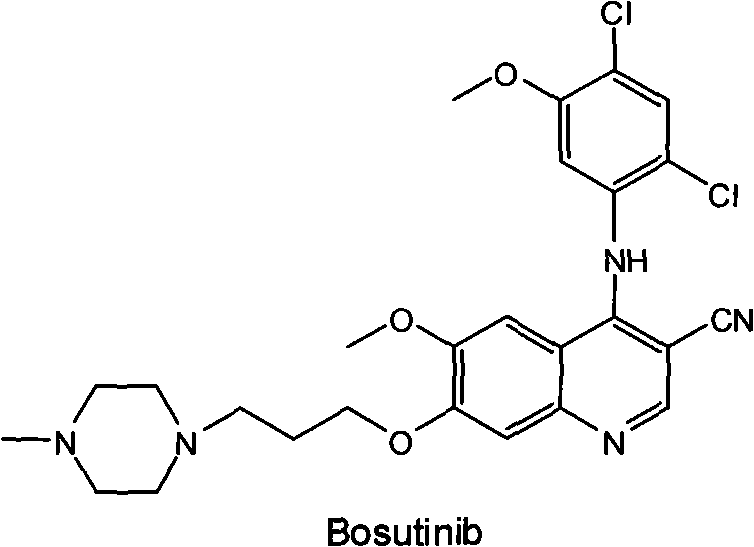
For R&D directors and process chemists evaluating technology transfer opportunities, the structural integrity and purity profile of the final active pharmaceutical ingredient are paramount. The disclosed route minimizes the formation of difficult-to-remove impurities by streamlining the number of unit operations. Unlike legacy methods that require multiple protection and deprotection sequences which inherently increase the risk of side reactions, this novel approach maintains a linear and efficient progression from simple aromatic acids to the complex heterocyclic target. The strategic selection of vanillic acid as the foundational building block ensures that the methoxy and hydroxyl functionalities are positioned optimally for the subsequent alkylation and ring-closing steps, thereby enhancing the overall atom economy of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Bosutinib, such as those described in US 2005/0043537 A1, rely heavily on expensive and less accessible starting materials like 2-methoxy-5-nitrophenol. These conventional routes are plagued by significant operational inefficiencies, including excessively long reaction times that can extend up to 40 hours for specific transformation steps, creating a bottleneck in production throughput. Furthermore, alternative literature routes often necessitate the use of hydroxyl protecting groups, which adds unnecessary synthetic steps and increases the consumption of reagents and solvents. Perhaps most critically from a safety and engineering perspective, some established methods require cryogenic conditions as low as -78°C for the cyclization phase, demanding specialized equipment and substantial energy inputs that are impractical for large-scale commercial manufacturing. These factors collectively result in a high cost of goods sold and a fragile supply chain susceptible to disruptions.
The Novel Approach
In stark contrast, the novel process detailed in CN101792416A leverages the inherent reactivity of vanillic acid derivatives to bypass these historical limitations. By utilizing 1-bromo-3-chloropropane for the alkylation step, the process efficiently installs the requisite propoxy side chain in a single operation with high conversion rates. The most transformative aspect of this new methodology is the cyclization step, which proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling systems. This mild condition not only reduces the operational expenditure significantly but also enhances the safety profile of the manufacturing process by avoiding thermal runaways associated with exothermic reactions at elevated temperatures. The route is notably shorter, reducing the cumulative yield loss typically observed in multi-step syntheses, and ensures that the final product is obtained with superior purity specifications suitable for downstream pharmaceutical formulation.
Mechanistic Insights into Quinoline Cyclization and Substitution
The core of this synthetic innovation revolves around the efficient construction of the quinoline ring system, a structural motif essential for the biological activity of Bosutinib as a Src/Abl dual inhibitor. The mechanism involves the condensation of an aniline intermediate with cyanoacetaldehyde diethyl acetal, followed by an intramolecular cyclization that forms the heterocyclic core. In traditional approaches, this cyclization often requires harsh acidic conditions or high thermal energy to overcome the activation barrier, leading to decomposition of sensitive functional groups. However, the electronic properties of the vanillic acid-derived intermediate, specifically the electron-donating methoxy group at the 6-position, activate the aromatic ring sufficiently to allow cyclization under neutral or mildly basic conditions at ambient temperature. This mechanistic nuance is critical for preserving the integrity of the nitrile group and preventing the hydrolysis or polymerization of the reactive enamine intermediates.
Impurity control is further enhanced during the final nucleophilic aromatic substitution steps where the chloro-quinoline intermediate reacts with 2,4-dichloro-5-anisidine and subsequently N-methylpiperazine. The process employs sodium iodide as a catalyst to facilitate the displacement of the chloride atom by the piperazine moiety, a transformation that is often sluggish due to steric hindrance. By optimizing the molar ratios and solvent systems, the reaction drives towards completion while minimizing the formation of bis-substituted byproducts or unreacted starting materials. The rigorous control over stoichiometry and temperature during these final stages ensures that the impurity profile remains well within the stringent limits required for clinical-grade materials. This level of mechanistic understanding allows process chemists to predict and mitigate potential quality issues before they arise during scale-up.
How to Synthesize Bosutinib Efficiently
The implementation of this synthesis route requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and high yield. The process begins with the esterification of vanillic acid, followed by a sequence of alkylation, nitration, and reduction to generate the key aniline precursor. Each step is designed to be telescoped or worked up with minimal purification to maintain efficiency. The subsequent cyclization and chlorination steps must be monitored closely to prevent over-reaction, while the final coupling reactions benefit from the use of polar aprotic solvents to enhance solubility and reaction kinetics. For a comprehensive breakdown of the specific reagent quantities, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Convert vanillic acid to methyl vanillate via esterification, followed by alkylation with 1-bromo-3-chloropropane to introduce the propoxy chain.
- Perform nitration and subsequent reduction to form the aniline derivative, then condense with cyanoacetaldehyde diethyl acetal.
- Execute mild room-temperature cyclization to form the quinoline core, followed by chlorination, coupling with dichloroanisidine, and final displacement with N-methylpiperazine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain management perspective, the adoption of this vanillic acid-based synthesis route offers profound strategic benefits that extend beyond simple technical feasibility. The shift away from proprietary or scarce starting materials to commodity chemicals like vanillic acid fundamentally alters the cost structure of the intermediate, making it less susceptible to market volatility and supplier monopolies. This transition supports a more resilient supply chain capable of withstanding global disruptions, as the raw materials are sourced from established industrial producers with massive capacity. Furthermore, the simplification of the process flow reduces the dependency on specialized contract manufacturing organizations that possess cryogenic capabilities, thereby broadening the pool of qualified suppliers and increasing competition which drives down costs.
- Cost Reduction in Manufacturing: The elimination of expensive reagents and the removal of protection-deprotection sequences result in a drastic reduction in raw material expenditures. By avoiding the use of cryogenic cooling for the cyclization step, the process achieves substantial energy savings, lowering the utility costs associated with production. Additionally, the shorter reaction times and higher yields mean that the same production equipment can generate more output in less time, effectively amortizing fixed capital costs over a larger volume of product. These factors combine to create a significantly lower cost of goods sold, allowing for more competitive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Utilizing vanillic acid, a bulk chemical with a stable global supply, mitigates the risk of raw material shortages that often plague niche synthetic intermediates. The robustness of the reaction conditions, which do not require exotic catalysts or extreme temperatures, ensures that production can be maintained consistently across different manufacturing sites without significant technology transfer friction. This reliability is crucial for maintaining continuous inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients. The simplified logistics of handling non-hazardous, ambient-temperature reactions further reduce the regulatory burden and transportation costs associated with the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to kilolab and eventually to multi-ton commercial production without the need for re-engineering critical steps. The reduction in solvent usage and the avoidance of hazardous cryogenic fluids contribute to a greener manufacturing footprint, aligning with increasingly strict environmental regulations and corporate sustainability goals. Waste generation is minimized due to the higher atom economy and fewer purification steps, simplifying waste treatment protocols and reducing disposal costs. This environmental compliance not only protects the company from regulatory fines but also enhances its reputation as a responsible manufacturer in the eyes of stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Bosutinib synthesis process. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the vanillic acid route over conventional Bosutinib synthesis methods?
A: The vanillic acid route eliminates the need for expensive starting materials like 2-methoxy-5-nitrophenol and avoids harsh cryogenic conditions (-78°C) required in previous methods. It utilizes industrially available raw materials and features a mild cyclization step at room temperature, significantly simplifying the operational complexity and reducing energy consumption.
Q: How does this process improve impurity control and product purity?
A: By shortening the synthetic route and removing protection-deprotection steps found in older methodologies, the potential for side reactions and byproduct formation is drastically reduced. The specific reaction conditions, particularly the controlled cyclization and chlorination steps, are optimized to minimize the generation of structural analogs, ensuring a cleaner crude profile prior to final purification.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. The use of common solvents, ambient temperature cyclization, and robust reaction parameters makes it highly adaptable for kilolab and plant-scale production, addressing the supply continuity needs of global pharmaceutical supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bosutinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the vanillic acid route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Bosutinib intermediate meets the highest global standards. Our commitment to quality assurance means that we can navigate the complexities of impurity profiling and process validation with confidence, delivering a product that is ready for immediate API synthesis.
We invite forward-thinking procurement leaders and R&D directors to collaborate with us to unlock the full potential of this optimized synthesis route. By leveraging our technical prowess, your organization can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can secure your supply of high-quality Bosutinib intermediates while driving significant value for your organization.
