Advanced Bosutinib Manufacturing: A Greener Condensation-Reduction Strategy for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the synthesis of complex kinase inhibitors, and the preparation method disclosed in patent CN103265482A represents a significant leap forward in the manufacturing of Bosutinib. This pivotal document outlines a novel synthetic strategy that diverges from the hazardous, multi-step classical routes by employing a direct condensation and reduction sequence. By reacting 6-methoxy-7-[3-(4-methyl-1-piperazinyl)propoxy]-4-amino-3-quinolinecarbonitrile with 2,4-dichloro-5-methoxybenzaldehyde, the process achieves the target molecule with remarkable simplicity. This technical breakthrough not only addresses the critical issue of hazardous waste generation but also streamlines the supply chain for this potent Src/Abl inhibitor used in treating chronic myeloid leukemia. For global procurement and R&D teams, understanding this shift from aggressive chlorination to mild coupling is essential for evaluating future sourcing strategies and cost structures.
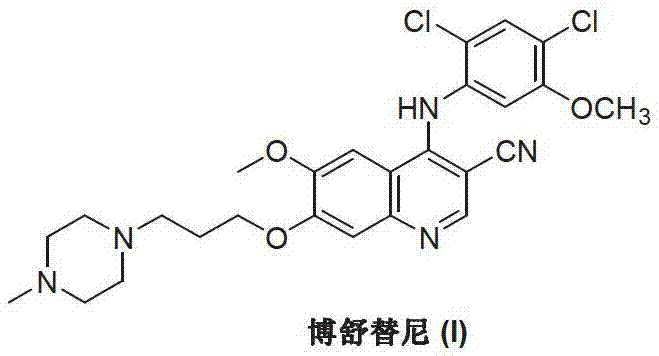
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bosutinib has been dominated by routes that rely heavily on the activation of the quinolinone core through aggressive chlorination. As illustrated in the prior art, classical approaches typically begin with aniline derivatives undergoing condensation and cyclization to form 4-oxo-3-quinoline carbonitrile derivatives. These intermediates must then be subjected to harsh chlorination conditions using reagents such as phosphorus oxychloride (POCl3), phosphorus trichloride, or thionyl chloride to generate the reactive 4-chloro-3-quinoline species. This reliance on hazardous chlorinating agents presents severe drawbacks, including the generation of large volumes of acidic wastewater and the requirement for specialized corrosion-resistant equipment. Furthermore, these conventional pathways often necessitate high-temperature reflux conditions and prolonged reaction times, which increase energy consumption and complicate the purification process due to the formation of thermally induced byproducts.
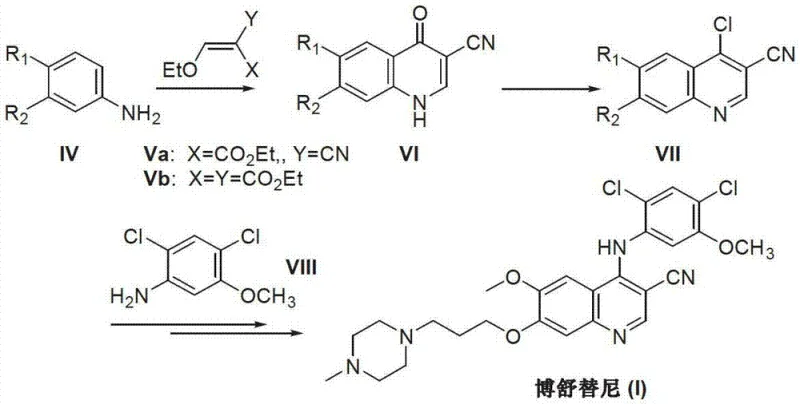
The Novel Approach
In stark contrast to the legacy methodologies, the innovative process described in the patent utilizes a convergent strategy that bypasses the need for dangerous chlorination entirely. The core of this advancement lies in the direct coupling of a pre-functionalized amino-quinoline intermediate with a specific benzaldehyde derivative. This condensation reaction, followed immediately by a reduction step, constructs the critical C-N bond linking the quinoline and phenyl rings under significantly milder conditions. By eliminating the chlorination-dechlorination sequence, the process inherently reduces the number of unit operations and removes the most environmentally damaging step from the manufacturing workflow. This streamlined approach not only enhances the overall atom economy but also simplifies the downstream processing, making it an ideal candidate for cost reduction in API manufacturing where waste disposal and safety compliance are major cost drivers.
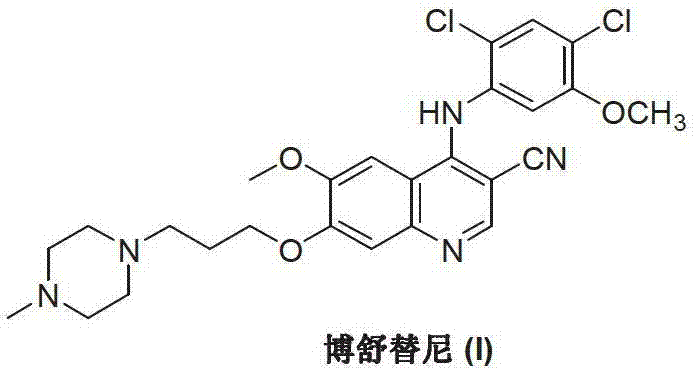
Mechanistic Insights into Condensation-Reduction and Lewis Acid Catalysis
The mechanistic elegance of this new pathway is twofold, involving both the final assembly of the drug molecule and the construction of the key quinoline intermediate. In the final step, the primary amine of the quinoline derivative acts as a nucleophile, attacking the carbonyl carbon of the 2,4-dichloro-5-methoxybenzaldehyde to form an imine intermediate in situ. This imine is subsequently reduced to the secondary amine using mild reducing agents such as sodium borohydride or via catalytic hydrogenation. The choice of reducing agent is critical; for instance, using sodium borohydride at controlled low temperatures (0-5°C) prevents over-reduction or side reactions, ensuring high selectivity. This mechanism avoids the formation of chlorinated impurities that are notoriously difficult to remove in traditional routes, thereby enhancing the purity profile of the final active pharmaceutical ingredient.
Furthermore, the synthesis of the crucial amino-quinoline intermediate itself employs a sophisticated Lewis acid-catalyzed cyclization. The reaction between 3-[3-(4-methyl-1-piperazinyl)propoxy]-4-anisidine, triethyl orthoformate, and malononitrile is facilitated by catalysts such as aluminum chloride. This Lewis acid activates the electrophilic centers, promoting the cyclization to form the quinoline ring system efficiently. The use of aluminum chloride allows the reaction to proceed at reflux temperatures in ethanol, yielding the intermediate in high purity (over 87% in reported examples). This specific catalytic cycle demonstrates a robust method for constructing the heterocyclic core without resorting to the harsh conditions typically associated with quinoline synthesis, providing a reliable foundation for the subsequent condensation step.
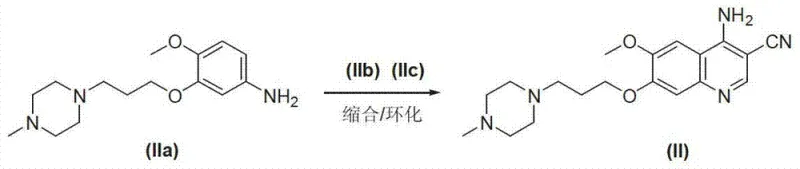
How to Synthesize Bosutinib Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the amino-quinoline intermediate via the aforementioned Lewis acid catalysis, followed by the critical condensation with the dichloro-methoxybenzaldehyde. Operators must maintain strict temperature control during the addition of the aldehyde and the subsequent reduction phase to prevent exothermic runaways. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety during scale-up.
- Prepare the key intermediate 6-methoxy-7-[3-(4-methyl-1-piperazinyl)propoxy]-4-amino-3-quinolinecarbonitrile via Lewis acid catalyzed cyclization.
- Perform condensation between the amino-quinoline intermediate and 2,4-dichloro-5-methoxybenzaldehyde using an acid binding agent at 50-55°C.
- Execute the reduction step using sodium borohydride or catalytic hydrogenation to yield the final Bosutinib product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical novelty. The elimination of phosphorus oxychloride and thionyl chloride from the process map fundamentally alters the cost structure of production by removing the need for expensive hazardous waste treatment and specialized containment infrastructure. Additionally, the use of commercially available and stable starting materials, such as the specific benzaldehyde derivative and the aniline precursor, mitigates the risk of supply disruptions often associated with custom-synthesized, highly reactive intermediates. This stability in raw material sourcing translates directly into enhanced supply chain reliability and predictable lead times for bulk orders.
- Cost Reduction in Manufacturing: The removal of hazardous chlorinating agents significantly lowers the operational expenditure related to environmental compliance and waste disposal. Traditional routes generate substantial amounts of acidic waste that require neutralization and safe disposal, adding hidden costs to the final product price. By adopting a condensation-reduction pathway, manufacturers can achieve substantial cost savings through simplified effluent treatment and reduced energy consumption, as the reaction proceeds at moderate temperatures rather than requiring prolonged high-heat reflux.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable reagents like sodium borohydride and common aldehydes ensures a more resilient supply chain. Unlike processes dependent on moisture-sensitive or highly corrosive reagents that require special shipping and storage protocols, this method utilizes standard chemical logistics. This ease of handling reduces the complexity of inventory management and minimizes the risk of production delays caused by the unavailability of specialized hazardous materials, ensuring a continuous flow of high-purity kinase inhibitor intermediates.
- Scalability and Environmental Compliance: The simplified workup procedure, which often involves straightforward crystallization from alcohol solvents, facilitates easier scale-up from pilot plant to commercial tonnage. The absence of corrosive gases and toxic byproducts aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory tightening. This environmental compatibility not only protects the manufacturer's license to operate but also appeals to end-clients who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific Bosutinib preparation method. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the condensation-reduction approach for potential partners and technical evaluators.
Q: How does this new Bosutinib synthesis method improve environmental safety compared to traditional routes?
A: Traditional methods rely heavily on hazardous chlorinating agents like phosphorus oxychloride (POCl3) and thionyl chloride, which generate significant toxic waste. This novel approach utilizes a mild condensation-reduction strategy that completely eliminates the need for these aggressive reagents, drastically reducing environmental impact and waste treatment costs.
Q: What are the key advantages of the condensation-reduction pathway for large-scale production?
A: The process operates under milder conditions (50-55°C) compared to the high-temperature reflux required in classical cyclization routes. Furthermore, the use of readily available starting materials and simpler workup procedures, such as direct crystallization, enhances operational efficiency and scalability for industrial manufacturing.
Q: Can this method ensure high purity for pharmaceutical grade Bosutinib?
A: Yes, the method avoids the formation of chlorinated byproducts often associated with POCl3 usage. The specific reduction conditions using sodium borohydride or catalytic hydrogenation allow for precise control over the reaction, facilitating the production of high-purity intermediates and final API suitable for stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bosutinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this greener synthetic pathway for the global oncology market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Bosutinib intermediate or API meets the exacting standards required for clinical and commercial use. We are committed to leveraging our technical expertise to optimize this condensation-reduction route for maximum efficiency and yield.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full capabilities of this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive down costs and secure your supply chain for this critical cancer therapeutic.
