Scalable Production of Rod-Shaped Bisphenol A Diether Diphthalimide for High-Performance Polyetherimide Synthesis
Scalable Production of Rod-Shaped Bisphenol A Diether Diphthalimide for High-Performance Polyetherimide Synthesis
The development of high-performance engineering plastics relies heavily on the availability of ultra-pure monomers and intermediates that possess consistent physical properties suitable for industrial processing. Patent CN110218173B introduces a transformative methodology for the preparation of rod-shaped bisphenol A type diether diphthalimide, a critical precursor in the synthesis of polyetherimide (PEI) materials. This innovation addresses a longstanding bottleneck in fine chemical manufacturing where traditional crystallization techniques often yield agglomerated, sticky, or excessively fine particles that complicate downstream handling. By implementing a novel additive-controlled precipitation strategy, this technology ensures the formation of discrete, rod-shaped crystals with exceptional filtration characteristics and high chemical purity. The structural integrity of this key intermediate is fundamental to the performance of the final polymer, and the ability to control its morphology represents a significant advancement in process chemistry.
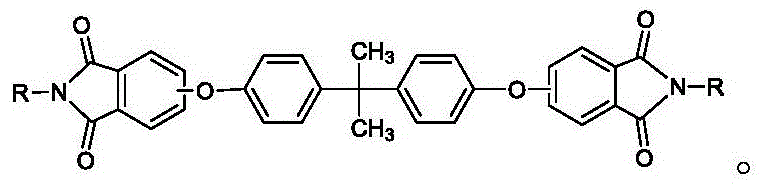
For procurement specialists and supply chain managers, the implications of this technological shift extend far beyond simple laboratory optimization. The transition from unpredictable, agglomerated masses to uniform rod-shaped crystals directly correlates with enhanced operational efficiency in large-scale production environments. Traditional methods often suffer from low filtration rates and high solvent retention in the filter cake, leading to increased energy consumption during drying and potential losses of valuable material. The new approach described in the patent mitigates these risks by engineering the crystal habit at the molecular level, resulting in a product that flows freely, filters rapidly, and retains minimal volatile organic compounds. This level of process control is essential for maintaining the rigorous quality standards required by the aerospace, automotive, and electronics industries where polyetherimides are extensively utilized.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bisphenol A type diether diphthalimide has been plagued by significant challenges related to product isolation and purification. Conventional protocols typically involve cooling the reaction mixture to induce precipitation, a method that frequently results in the formation of amorphous aggregates or extremely fine particulate matter. These physical forms are notoriously difficult to manage; fine particles tend to blind filtration media, causing severe bottlenecks that extend batch cycle times and increase operational costs. Furthermore, when conversion rates are incomplete, the presence of amphiphilic by-products—molecules possessing both hydrophilic phenolate groups and hydrophobic phthalimide moieties—exacerbates the problem by acting as binding agents that cause the product to clump into sticky masses. This agglomeration not only hampers mechanical stirring and heat transfer but also traps impurities within the crystal lattice, making it nearly impossible to achieve the high purity levels necessary for high-performance polymer applications without extensive and costly reprocessing steps.
The Novel Approach
The methodology outlined in patent CN110218173B fundamentally reengineers the crystallization step to overcome these inherent physical limitations. Instead of relying on passive cooling, the process utilizes a dynamic precipitation technique where the reaction solution is introduced into a hot alcohol solvent system containing specific additives. This system employs a synergistic combination of cationic surfactants, such as cetyltrimethylammonium bromide, and organic acids like formic or acetic acid. The cationic surfactant interacts electrostatically with the unilateral by-products, effectively neutralizing their tendency to cause agglomeration, while the organic acid maintains a weakly acidic environment that prevents the deactivation of the surfactant. This dual-action mechanism directs the crystal growth along specific axes, favoring the formation of elongated, rod-shaped structures rather than isotropic clusters. The result is a free-flowing powder with an average particle length ranging from 10 to 30 μm, which exhibits superior dispersion properties and drastically improved filtration kinetics compared to traditional outputs.
Mechanistic Insights into Additive-Controlled Crystallization
The success of this synthesis route lies in the precise manipulation of interfacial phenomena during the nucleation and growth phases of crystallization. In the absence of additives, the supersaturation generated upon mixing the reaction solution with the anti-solvent leads to rapid, uncontrolled nucleation, producing a multitude of small crystals that readily aggregate due to high surface energy. The introduction of cationic surfactants alters the surface tension at the crystal-solvent interface, selectively adsorbing onto specific crystal faces to inhibit growth in those directions while promoting elongation in others. Simultaneously, the organic acid component plays a crucial role in modulating the ionic strength and pH of the medium, ensuring that the surfactant remains active and soluble throughout the process. This careful balance prevents the occlusion of mother liquor and impurities within the crystal lattice, thereby enhancing the overall chemical purity of the isolated solid. The interaction between the additives and the solute molecules is governed by van der Waals forces and hydrogen bonding, which collectively dictate the final morphological outcome of the precipitation.
From a quality control perspective, this mechanistic understanding allows for robust process parameter optimization to ensure batch-to-batch consistency. The patent specifies that the concentration of additives can be adjusted between 1 wt% and 5 wt% to accommodate variations in reaction conversion and residual alkali content, providing a flexible control knob for process engineers. By maintaining the precipitation temperature between 50°C and 78°C and controlling the addition rate of the reaction mixture, the system avoids local zones of excessive supersaturation that could lead to secondary nucleation and fines generation. The subsequent reflux washing step in alcohol further purifies the rod-shaped crystals by dissolving any adhering surface impurities without compromising the crystal integrity. This comprehensive approach to crystal engineering ensures that the final product not only meets stringent chemical specifications with purity levels exceeding 99.8% but also possesses the physical attributes required for efficient automated handling in downstream polymerization reactors.
How to Synthesize Bisphenol A Diether Diphthalimide Efficiently
The synthesis protocol detailed in the patent offers a reproducible pathway for manufacturing this critical intermediate with high yield and superior morphology. The process begins with the formation of the bisphenol A salt in a non-polar solvent, followed by a nucleophilic substitution reaction in a polar aprotic medium, and concludes with the innovative additive-assisted crystallization. This sequence is designed to maximize conversion while minimizing the formation of the problematic amphiphilic by-products that hinder traditional methods. Operators must pay close attention to the stoichiometry of the base and the temperature profiles during the salt formation and reaction stages to ensure optimal feedstock quality for the crystallization step. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for scaling this technology to commercial production volumes.
- Prepare bisphenol A salt by reacting bisphenol A with a strong base in a non-polar solvent like o-xylene at reflux temperatures to remove water.
- Conduct a nucleophilic reaction between the bisphenol A salt and substituted phthalimide in a polar aprotic solvent such as DMAC or DMSO at 100-150°C.
- Precipitate the product into hot alcohol containing cationic surfactants and organic acids to induce rod-shaped crystal growth, followed by reflux washing.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the supply chain of high-performance polymers, the adoption of this rod-shaped crystallization technology offers substantial strategic benefits that extend well beyond the laboratory bench. The primary advantage lies in the drastic improvement of solid-liquid separation efficiency, which is often a rate-limiting step in fine chemical manufacturing. The formation of large, discrete rod-shaped crystals eliminates the blinding of filtration equipment commonly associated with fine powders, allowing for significantly faster cycle times and reduced downtime for cleaning and maintenance. This enhancement in throughput directly translates to lower manufacturing costs per kilogram, as the same production assets can generate higher output volumes without the need for capital-intensive equipment upgrades. Furthermore, the reduced moisture content in the filter cake minimizes the energy load required for drying, contributing to a more sustainable and cost-effective production profile that aligns with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of agglomeration and the prevention of sticky mass formation remove the need for complex reprocessing or manual intervention to break up clumps, which are labor-intensive and costly activities. By achieving high purity directly from the crystallization step, the process reduces the dependency on extensive recrystallization cycles, thereby lowering solvent consumption and waste disposal costs. The high yield reported in the patent examples, often exceeding 90%, indicates a highly efficient utilization of raw materials, minimizing the financial impact of unreacted starting materials and by-product formation. This efficiency is critical for maintaining competitive pricing in the global market for specialty polymer intermediates where margin pressures are constant.
- Enhanced Supply Chain Reliability: The robustness of the additive-controlled crystallization method ensures consistent product quality regardless of minor fluctuations in reaction conversion, providing a stable supply of material for downstream customers. The physical stability of the rod-shaped crystals prevents caking during storage and transportation, ensuring that the material arrives at the customer's facility in optimal condition for immediate use. This reliability reduces the risk of production stoppages at the polymerization stage, which can be exceedingly costly in continuous manufacturing environments. Additionally, the use of common, commercially available solvents and additives simplifies the procurement of raw materials, reducing the risk of supply disruptions associated with exotic or highly regulated reagents.
- Scalability and Environmental Compliance: The process is inherently scalable, as the mechanisms governing crystal growth are independent of vessel size when proper mixing and addition rates are maintained. The use of alcohol solvents for precipitation and washing facilitates solvent recovery and recycling, significantly reducing the volume of hazardous waste generated per unit of product. The low VOC content in the final filter cake further simplifies compliance with increasingly stringent environmental regulations regarding volatile emissions. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important for multinational corporations selecting long-term supply partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of rod-shaped bisphenol A type diether diphthalimide. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into existing polymer synthesis workflows and for assessing the total cost of ownership associated with its procurement.
Q: Why is the rod-shaped morphology critical for polyetherimide intermediates?
A: Rod-shaped crystals exhibit superior filtration characteristics compared to agglomerated fines, significantly reducing processing time and solvent retention in the filter cake, which is vital for large-scale polymer synthesis.
Q: How does the additive system prevent product agglomeration?
A: The combination of cationic surfactants and organic acids modifies the surface energy of the growing crystals and interacts with amphiphilic by-products, preventing them from acting as binding agents that cause stickiness.
Q: What purity levels can be achieved with this crystallization method?
A: This method consistently achieves purity levels exceeding 99.8% by effectively excluding impurities during the controlled crystal growth phase and through efficient reflux washing protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol A Diether Diphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final polymer is inextricably linked to the purity and physical consistency of its monomeric precursors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory successes of advanced crystallization technologies are seamlessly translated into robust industrial processes. We are committed to delivering high-purity bisphenol A type diether diphthalimide that meets stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to control crystal morphology ensures that our clients receive a product that is not only chemically pure but also physically optimized for efficient handling and reaction kinetics in their polyetherimide synthesis operations.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall material costs and improve your production efficiency. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your application needs. Our goal is to establish a long-term strategic partnership that drives innovation and value creation in the high-performance polymer sector, leveraging our expertise in fine chemical synthesis to solve your most complex supply chain challenges.
