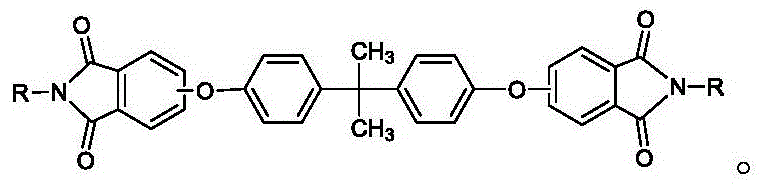
Scaling High-Purity Rod-Shaped Bisphenol A Diether Diphthalimide for Advanced Polyetherimide Production
The global demand for high-performance engineering plastics, specifically polyetherimide (PEI), has necessitated a rigorous re-evaluation of the intermediate supply chain, particularly regarding the quality and physical form of key precursors. Patent CN110218173B introduces a transformative preparation method for rod-shaped bisphenol A type diether diphthalimide, addressing critical bottlenecks that have long plagued the manufacturing of this essential monomer. Traditional synthesis routes often suffer from uncontrolled crystallization, resulting in products that are difficult to filter, wash, and dry, thereby inflating production costs and compromising supply reliability. This patented technology leverages a sophisticated additive-controlled crystallization system to engineer the particle morphology at the molecular level, yielding discrete rod-shaped crystals with superior flow properties and exceptional chemical purity. For R&D directors and procurement strategists, this represents a significant leap forward in process robustness, offering a pathway to more efficient downstream polymerization and reduced operational expenditure in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bisphenol A type diether diphthalimide has been fraught with significant processing challenges that directly impact the bottom line of chemical manufacturing facilities. Conventional techniques typically involve the nucleophilic substitution of bisphenol A salts with phthalimide derivatives, followed by a crude precipitation step where the reaction mixture is simply cooled or poured into a non-solvent. In these traditional scenarios, the resulting product tends to precipitate as fine, irregular particles or, worse, as sticky agglomerates that adhere tenaciously to reactor walls and stirring paddles. This phenomenon is particularly exacerbated when conversion rates are incomplete, leading to the presence of amphiphilic by-products that act as binding agents, causing the entire batch to form a viscous, unmanageable mass. Such physical characteristics create severe filtration bottlenecks, where filter cloths become blinded rapidly, extending cycle times drastically and increasing energy consumption for drying. Furthermore, the high surface area of fine, irregular particles retains excessive amounts of mother liquor, leading to higher solvent residues and necessitating extensive, resource-intensive washing protocols that still may not achieve the stringent purity standards required for high-end electronic or aerospace grade polymers.
The Novel Approach
The methodology disclosed in CN110218173B fundamentally alters the crystallization landscape by introducing a dual-additive system during the precipitation phase, effectively engineering the crystal habit from the ground up. Instead of allowing random nucleation, the process directs the growth of the bisphenol A type diether diphthalimide into well-defined rod-shaped structures with average lengths ranging from 10 to 30 micrometers. This morphological control is achieved by precipitating the reaction solution into a hot alcohol solvent containing specific concentrations of cationic surfactants and organic acids. The rod-shaped geometry inherently possesses superior packing characteristics, creating a filter cake with high porosity and low resistance to fluid flow, which dramatically accelerates filtration rates and reduces the moisture content of the wet cake. By preventing the formation of sticky agglomerates, this approach eliminates the risk of reactor fouling and ensures that the product remains free-flowing throughout the isolation and drying stages. Consequently, the novel approach not only enhances the physical handling properties of the intermediate but also facilitates a more thorough removal of impurities, resulting in a final product with purity levels consistently exceeding 99.8%, which is critical for the subsequent synthesis of high-molecular-weight polyetherimides.
Mechanistic Insights into Additive-Controlled Crystallization
The core innovation of this technology lies in the synergistic interaction between the cationic surfactant and the organic acid within the alcoholic precipitation medium, which acts as a sophisticated crystal habit modifier. During the rapid precipitation event, amphiphilic by-products—typically unilateral species containing both unreacted phenolate groups and phthalimide moieties—tend to adsorb onto the growing crystal faces, disrupting orderly lattice formation and promoting aggregation. The cationic surfactant, such as cetyltrimethylammonium bromide or chloride, selectively interacts with these anionic or polar by-product species through electrostatic forces, effectively sequestering them and preventing them from interfering with the primary crystal growth of the target diphthalimide. Simultaneously, the organic acid component, such as formic or acetic acid, maintains a slightly acidic environment that prevents the deactivation of the cationic surfactant while also modulating the supersaturation levels of the solute. This dual-action mechanism ensures that the solute molecules diffuse to the crystal interface at a controlled rate, favoring longitudinal growth over lateral expansion, which results in the characteristic rod-like morphology. By fine-tuning the concentration of these additives, manufacturers can precisely dictate the aspect ratio and size distribution of the crystals, ensuring batch-to-batch consistency that is vital for automated downstream processing.
Furthermore, this mechanistic control extends to the suppression of occlusion defects, which are common in rapidly precipitated systems. In conventional processes, the rapid collapse of solubility traps solvent molecules and impurities within the crystal lattice, leading to broad melting point ranges and inconsistent reactivity in subsequent polymerization steps. The additive-mediated growth described in this patent promotes a more ordered lattice assembly, minimizing the inclusion of solvent pockets and foreign ions. The result is a crystal structure that is thermodynamically more stable and chemically homogeneous. For the R&D team, this means that the intermediate exhibits predictable reactivity during the subsequent hydrolysis and dehydration steps required to form the dianhydride monomer. The high purity and defined morphology reduce the variability in the polymerization kinetics, allowing for tighter control over the molecular weight and viscosity of the final polyetherimide resin, which are key performance indicators for applications in aerospace composites and medical devices.
How to Synthesize Rod-Shaped Bisphenol A Diether Diphthalimide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for transitioning from laboratory-scale discovery to commercial production, emphasizing precise control over reaction parameters and isolation conditions. The process begins with the formation of the bisphenol A salt in a non-polar solvent, followed by a nucleophilic substitution in a polar aprotic medium, but the critical differentiator is the workup procedure. To achieve the desired rod-shaped morphology, operators must strictly adhere to the temperature and agitation specifications during the precipitation step, ensuring that the reaction mixture is introduced into the hot alcohol anti-solvent at a controlled rate while maintaining high shear mixing.
- Prepare bisphenol A salt by reacting bisphenol A with a strong base like sodium hydroxide in a non-polar solvent such as o-xylene under reflux.
- Conduct nucleophilic reaction between the bisphenol A salt and phthalimide derivatives in a polar aprotic solvent like DMAC at elevated temperatures.
- Precipitate the product in hot alcohol containing cationic surfactants and organic acids to induce rod-shaped crystal growth, followed by reflux washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rod-shaped crystallization technology translates into tangible operational efficiencies and risk mitigation across the value chain. The primary economic driver is the drastic improvement in solid-liquid separation efficiency; because the rod-shaped crystals form a highly permeable filter cake, filtration times are significantly reduced compared to the gelatinous masses produced by older methods. This reduction in cycle time allows for higher throughput in existing equipment without the need for capital-intensive upgrades, effectively debottlenecking the production line. Additionally, the lower moisture content of the filtered cake means that less energy is required for the drying process, leading to substantial cost savings in utility consumption. The elimination of sticky agglomerates also reduces the frequency of reactor cleaning and maintenance downtime, ensuring greater asset availability and production continuity. From a supply security perspective, the robustness of this method against variations in conversion rates means that off-spec batches are minimized, guaranteeing a more reliable supply of high-quality intermediates to downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the reduction in solvent usage due to improved washing efficiency directly lower the variable cost per kilogram of the intermediate. By avoiding the need for extensive recrystallization or specialized filtration equipment to handle fine powders, the overall manufacturing footprint is optimized. The process relies on commodity chemicals like alcohols and common surfactants, avoiding the volatility and cost associated with exotic catalysts or reagents. This streamlined approach ensures that the cost structure remains competitive even in fluctuating raw material markets, providing a stable pricing foundation for long-term supply agreements.
- Enhanced Supply Chain Reliability: The high yield and consistent purity achieved through this method reduce the risk of production delays caused by quality failures or reprocessing requirements. The rod-shaped product's superior flowability facilitates easier packaging and transportation, minimizing the risk of caking or bridging during storage and logistics. This physical stability ensures that the material arrives at the customer's site in optimal condition, ready for immediate use without additional conditioning. For supply chain planners, this predictability simplifies inventory management and reduces the safety stock levels required to buffer against quality-related disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations that are easily replicated from pilot plants to multi-ton reactors. The use of recyclable alcohol solvents and the reduction in wastewater generation due to efficient washing contribute to a lower environmental footprint. The high purity of the final product minimizes the generation of hazardous waste streams associated with purification by-products. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced crystallization technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your supply chain. Understanding these nuances is essential for making informed decisions about sourcing strategies and process integration.
Q: Why is the rod-shaped morphology critical for polyetherimide intermediates?
A: Rod-shaped crystals prevent agglomeration and stickiness during filtration, significantly reducing processing time and solvent retention compared to conventional fine or amorphous particles.
Q: How does the additive system improve product purity?
A: The combination of cationic surfactants and organic acids interacts with amphiphilic by-products, preventing them from incorporating into the crystal lattice and ensuring purity levels exceeding 99.8%.
Q: Can this process be scaled for industrial polyetherimide production?
A: Yes, the method utilizes standard solvents and avoids complex transition metal catalysts, making it highly scalable from pilot batches to multi-ton commercial manufacturing with consistent crystal quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol A Diether Diphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final polymer is inextricably linked to the precision of its building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate crystallization controls required for rod-shaped bisphenol A diether diphthalimide are maintained at every scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of crystal morphology and residual solvent content. We understand that for high-performance applications, consistency is not just a metric but a mandate, and our process validation protocols are designed to deliver batch-after-batch reliability that meets the exacting standards of the global electronics and aerospace industries.
We invite you to collaborate with our technical procurement team to explore how this advanced intermediate can optimize your polyetherimide manufacturing process. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential efficiency gains in your specific production environment. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to validate the superior performance of our rod-shaped intermediates against your current supply baseline. Let us partner with you to drive innovation and efficiency in your high-performance polymer supply chain.
