Advanced Synthesis of Rod-Shaped Bisphenol A Diether Diphthalimide for Commercial Polyetherimide Production
The global demand for high-performance engineering plastics, particularly polyetherimide (PEI), continues to surge across aerospace, automotive, and electronics sectors, driving an urgent need for reliable suppliers of critical monomers. Patent CN110218173B introduces a transformative preparation method for rod-shaped bisphenol A type diether diphthalimide, a pivotal intermediate in PEI synthesis. This technology addresses long-standing bottlenecks in crystallization and purification that have historically plagued the manufacturing of this complex aromatic imide. By shifting from traditional precipitation techniques to a sophisticated additive-controlled crystallization process, the patent enables the production of high-purity intermediates with superior physical properties. For R&D directors and procurement strategists, this innovation represents a significant leap forward in process robustness, offering a pathway to reduce operational expenditures while ensuring the stringent quality standards required for advanced polymer applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bisphenol A type diether diphthalimide has been fraught with significant downstream processing challenges that directly impact manufacturing economics. Traditional protocols typically involve cooling the reaction mixture to precipitate the product, a method that frequently results in severe agglomeration and the formation of sticky, viscous masses, particularly when reaction conversion is incomplete. These agglomerates adhere tenaciously to stirring paddles and reactor walls, necessitating labor-intensive manual cleaning and leading to substantial product loss during recovery. Furthermore, even when agglomeration is minimized, conventional methods often yield extremely fine crystal particles that create high resistance during filtration, causing frequent filter cloth blinding and drastically extending cycle times. The presence of amphiphilic by-products, specifically unilateral species containing both phenolate and phthalimide groups, exacerbates these issues by acting as surfactants that stabilize unwanted emulsions or gels, making the isolation of the pure diether diphthalimide inefficient and costly in terms of both time and solvent consumption.
The Novel Approach
The methodology disclosed in CN110218173B fundamentally re-engineers the isolation step to overcome these physical limitations through precise crystal habit modification. Instead of simple cooling, the process employs a controlled precipitation into a hot alcohol solvent system fortified with a specific combination of cationic surfactants and organic acids. This dual-additive strategy effectively disrupts the intermolecular forces that lead to particle sticking, guiding the solute molecules to assemble into well-defined, discrete rod-shaped crystals with lengths ranging from 10 to 30 micrometers. 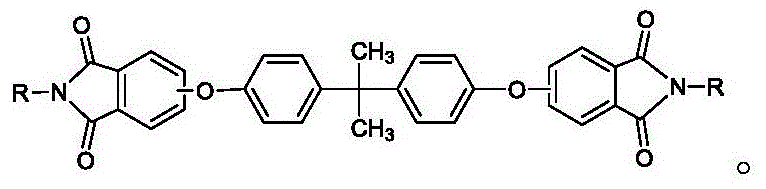 This morphological control not only prevents the formation of filter-clogging fines but also produces a filter cake with significantly lower wet content, thereby reducing the energy load required for subsequent drying operations. The result is a streamlined workflow where filtration proceeds rapidly without blockage, and the final product exhibits exceptional purity levels exceeding 99.8%, ready for immediate downstream polymerization without extensive recrystallization.
This morphological control not only prevents the formation of filter-clogging fines but also produces a filter cake with significantly lower wet content, thereby reducing the energy load required for subsequent drying operations. The result is a streamlined workflow where filtration proceeds rapidly without blockage, and the final product exhibits exceptional purity levels exceeding 99.8%, ready for immediate downstream polymerization without extensive recrystallization.
Mechanistic Insights into Additive-Controlled Crystallization
The core scientific breakthrough of this patent lies in the intricate interplay between the reaction by-products and the added crystallization modifiers during the phase transition from solution to solid. During the nucleophilic substitution reaction, incomplete conversion inevitably generates unilateral intermediates which possess both hydrophilic phenolate moieties and hydrophobic phthalimide groups, rendering them amphiphilic. In standard processes, these molecules accumulate at the crystal-liquid interface, acting as binding agents that fuse primary particles into large, unmanageable clumps. The introduction of cationic surfactants, such as cetyltrimethylammonium bromide or chloride, creates a competitive adsorption environment where the surfactant molecules interact electrostatically with the anionic sites of the by-products, effectively neutralizing their stickiness and preventing them from bridging between growing crystal faces. Simultaneously, the inclusion of organic acids like formic or acetic acid serves a critical protective function by maintaining a mildly acidic microenvironment that prevents the hydrolysis or deactivation of the cationic surfactant, ensuring its efficacy throughout the precipitation window.
Beyond merely preventing agglomeration, this additive system actively directs the supramolecular assembly of the bisphenol A diether diphthalimide molecules into a rod-like lattice structure. The additives function similarly to crystal growth modifiers in biomineralization, selectively adsorbing onto specific crystallographic planes to inhibit growth in certain directions while promoting elongation in others. This controlled diffusion and interfacial mass transfer result in the formation of uniform rods rather than irregular aggregates, which dramatically enhances the bulk density and flowability of the powder. For process chemists, understanding this mechanism is vital for scaling, as it implies that the process is robust against minor fluctuations in reaction conversion; the additives can be tuned to compensate for varying levels of impurities, ensuring consistent product morphology batch after batch. This level of control over solid-state properties is rare in fine chemical synthesis and provides a distinct competitive advantage in manufacturing high-value polymer precursors.
How to Synthesize Bisphenol A Diether Diphthalimide Efficiently
The synthesis protocol outlined in the patent is designed for scalability and reproducibility, utilizing common industrial solvents and reagents to minimize supply chain risks. The process begins with the formation of the bisphenol A salt in a non-polar medium, followed by a high-temperature nucleophilic attack in a polar aprotic solvent, and concludes with the critical additive-assisted precipitation. This sequence ensures high conversion rates while managing the thermal profile to prevent degradation of the sensitive imide rings. The detailed standardized operating procedures, including specific temperature ramps, stirring velocities, and additive concentrations required to achieve the optimal rod-shaped morphology, are essential for transferring this laboratory success to commercial production lines.
- Prepare bisphenol A salt by reacting bisphenol A with a strong base like NaOH or KOH in a non-polar solvent such as o-xylene or toluene under reflux conditions.
- Conduct a nucleophilic reaction between the bisphenol A salt and substituted phthalimide in a polar aprotic solvent like DMAC or DMSO at temperatures between 100-150°C.
- Precipitate the product in hot alcohol containing cationic surfactants and organic acids to induce rod-shaped crystal formation, followed by reflux washing and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rod-shaped crystal technology translates directly into tangible operational efficiencies and cost reductions across the manufacturing value chain. The primary economic driver is the drastic improvement in solid-liquid separation efficiency; by eliminating the formation of fine, clogging particles and sticky agglomerates, the process significantly reduces filtration time and extends the lifespan of filtration media. This enhancement allows for faster batch turnover rates and lowers the utility costs associated with vacuum generation and pump operation, contributing to a leaner manufacturing footprint. Furthermore, the high purity achieved directly from the crystallization step minimizes the need for resource-intensive recrystallization cycles, thereby reducing solvent consumption and waste disposal volumes, which aligns with increasingly stringent environmental compliance regulations.
- Cost Reduction in Manufacturing: The elimination of agglomeration and the formation of free-flowing rod-shaped crystals lead to substantial savings in downstream processing costs. Traditional methods often suffer from significant product loss due to material adhering to reactor walls or passing through filters as fines, whereas this novel approach maximizes yield recovery through efficient cake formation. Additionally, the lower moisture content of the rod-shaped filter cake reduces the thermal energy required for drying, resulting in lower natural gas or steam consumption per kilogram of product. By avoiding the use of expensive transition metal catalysts and relying on readily available bases and surfactants, the raw material cost structure remains optimized and stable against market volatility.
- Enhanced Supply Chain Reliability: The robustness of the additive-controlled crystallization process ensures consistent product quality even when facing variations in raw material grades or slight deviations in reaction conversion. This reliability reduces the risk of off-spec batches that could disrupt downstream polymerization schedules, providing a more predictable supply of critical PEI intermediates. The use of common solvents like toluene, ethanol, and DMAC ensures that sourcing remains flexible and不受限于 exotic reagents, mitigating the risk of supply disruptions. Moreover, the improved physical handling characteristics of the rod-shaped powder facilitate safer and faster packaging and logistics operations, reducing the potential for dust generation and spillage during transport.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is facilitated by the straightforward nature of the unit operations involved, which do not require specialized high-pressure or cryogenic equipment. The significant reduction in solvent usage for washing and the higher efficiency of solvent recovery due to simpler distillation streams contribute to a greener manufacturing profile. The process inherently generates less solid waste due to higher yields and reduces the volume of wastewater contaminated with fine particulate matter, simplifying effluent treatment requirements. This alignment with green chemistry principles not only lowers compliance costs but also enhances the sustainability credentials of the final polyetherimide products for end-users in regulated industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced crystallization technology for bisphenol A diether diphthalimide. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical benefits observed during development. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this method into existing production facilities.
Q: How does the novel crystallization method solve filtration issues in diether diphthalimide production?
A: Conventional methods often result in agglomerated, sticky masses or fine particles that clog filters. This patent utilizes a dual-additive system (cationic surfactant and organic acid) during precipitation in hot alcohol, which controls crystal growth to form discrete rod-shaped particles (10-30μm). This morphology significantly improves filtration efficiency and reduces product loss.
Q: What is the role of the additives in the crystallization process?
A: The cationic surfactant interacts with amphiphilic by-products (unilateral reaction products) to prevent them from causing agglomeration and stickiness. The organic acid maintains a weakly acidic environment to prevent the deactivation of the surfactant, ensuring consistent crystal morphology even with variations in reaction conversion rates.
Q: Why is the rod-shaped morphology beneficial for downstream polyetherimide manufacturing?
A: Rod-shaped crystals exhibit superior flow properties compared to irregular or agglomerated powders, facilitating easier feeding into downstream polymerization reactors. Additionally, this morphology results in a filter cake with lower moisture content and reduced VOC retention, lowering drying energy costs and improving overall process safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol A Diether Diphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patented laboratory methods to commercial reality requires deep technical expertise and rigorous process validation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate crystal engineering principles described in CN110218173B are faithfully reproduced at scale. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the rod-shaped morphology and chemical integrity of every batch, guaranteeing that the intermediates supplied meet the exacting demands of high-performance polymer manufacturers.
We invite global partners to collaborate with us to leverage this innovative synthesis route for their polyetherimide supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this rod-shaped crystal technology can optimize your total cost of ownership. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a supply partner committed to quality, efficiency, and long-term reliability.
