Revolutionizing Asymmetric Catalysis with Modular Axial Chiral Phosphine-Ene Ligands
Revolutionizing Asymmetric Catalysis with Modular Axial Chiral Phosphine-Ene Ligands
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient, modular, and cost-effective chiral ligands. Patent CN111718372B introduces a groundbreaking class of axial chiral phosphine-ene ligands, characterized by a robust biaryl skeleton that combines the weak coordination of a carbon-carbon double bond with the strong coordination of a phosphorus atom. This unique structural motif addresses long-standing challenges in the field, offering a versatile platform for transition metal catalysis. Unlike traditional ligands that often suffer from synthetic complexity and limited tunability, this innovation leverages a streamlined two-step synthesis from commodity aryl halides. For R&D directors and procurement managers alike, this represents a pivotal shift towards sustainable and economically viable manufacturing of high-value chiral intermediates. The technology promises to enhance the enantioselectivity of palladium-catalyzed reactions while drastically simplifying the supply chain for critical catalytic components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral phosphine-ene ligands has been hindered by intricate synthetic routes that rely heavily on the chiral pool or resolution of racemates. Conventional methods often involve numerous protection and deprotection steps, leading to low overall yields and substantial waste generation. Furthermore, many existing ligands possess rigid structures that are difficult to modify, limiting their applicability across different substrate classes. This lack of modularity forces process chemists to screen extensive libraries of expensive, custom-synthesized ligands for each new reaction, inflating R&D costs and extending project timelines. Additionally, the reliance on scarce natural products as starting materials creates supply chain vulnerabilities, making large-scale production risky and unpredictable. These factors collectively impede the rapid deployment of asymmetric catalysis in industrial settings, particularly for generic pharmaceutical intermediates where cost margins are tight.
The Novel Approach
The methodology disclosed in CN111718372B circumvents these bottlenecks through a clever integration of palladium catalysis and chiral norbornene mediation. By utilizing simple aryl iodides, aryl bromides, and alkenes as starting materials, the process constructs the axially chiral biaryl backbone in a single catalytic step. This approach eliminates the need for pre-existing chirality in the starting materials, as the axial chirality is induced dynamically during the coupling reaction. The subsequent reduction of the phosphine oxide intermediate is equally straightforward, employing standard reagents like trichlorosilane. This two-step sequence not only reduces the number of unit operations but also allows for rapid structural diversification. Chemists can easily tune the steric and electronic properties of the ligand by simply varying the substituents on the aryl rings, enabling precise optimization for specific transformations without redesigning the entire synthetic route.
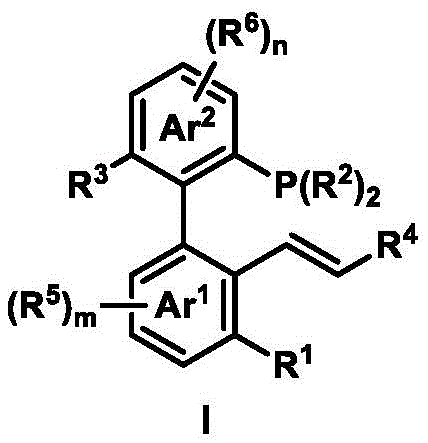
Mechanistically, the synthesis relies on a cooperative catalysis system where palladium and the chiral norbornene derivative work in tandem to activate inert C-H bonds and forge the biaryl axis with high stereocontrol. This Catellani-type reaction pathway ensures that the bulky substituents are positioned ortho to the biaryl bond, locking the conformation and preventing racemization at elevated temperatures. The resulting ligand possesses a unique "hemilabile" character; the alkene moiety can reversibly dissociate from the metal center, creating an open coordination site for substrate binding while the phosphine maintains a stable anchor. This dynamic behavior is crucial for achieving high turnover numbers and exceptional enantioselectivity in asymmetric allylic substitutions. The ability to fine-tune the bite angle and electronic density through the R-groups on the biaryl scaffold further enhances the ligand's versatility, making it suitable for a wide array of nucleophiles ranging from soft carbon nucleophiles to harder oxygen and nitrogen donors.
From an impurity control perspective, this synthetic route offers distinct advantages. The use of well-defined aryl halide precursors minimizes the formation of regioisomers, a common issue in direct C-H functionalization strategies. Moreover, the intermediate phosphine oxide is significantly more stable towards oxidation than the final phosphine product, allowing for purification via column chromatography before the final reduction step. This "protect-as-you-go" strategy ensures that the final ligand is obtained with high chemical purity, which is critical for maintaining consistent catalytic performance in sensitive pharmaceutical syntheses. The robustness of the biaryl axis also means the ligand can withstand the rigorous conditions often required for scale-up, reducing the risk of decomposition or loss of optical purity during manufacturing.
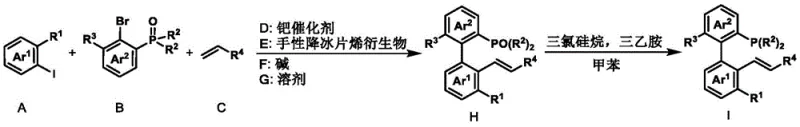
How to Synthesize Axial Chiral Phosphine-Ene Ligand Efficiently
The preparation of these high-performance ligands is designed for operational simplicity and scalability. The process begins with the assembly of the chiral backbone using a palladium-catalyzed coupling reaction in acetonitrile, followed by a reduction step in toluene. This streamlined workflow minimizes solvent exchanges and purification burdens, making it highly attractive for kilogram-scale production. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and quality control.
- Perform a palladium-catalyzed coupling of aryl iodide, aryl bromide, and alkene using a chiral norbornene derivative to form the phosphine oxide intermediate.
- Reduce the phosphine oxide intermediate using trichlorosilane and triethylamine in toluene at elevated temperatures.
- Utilize the resulting ligand in palladium-catalyzed asymmetric allylic substitution reactions with various nucleophiles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology translates into tangible strategic benefits. The shift from complex, multi-step syntheses to a modular two-step process significantly de-risks the supply of critical chiral catalysts. By decoupling ligand production from the volatile market of natural chiral pools, manufacturers can secure a more stable and predictable supply chain. The use of commodity chemicals like aryl bromides and styrenes as feedstocks ensures that raw material costs remain low and availability is high, even during global supply disruptions. Furthermore, the high efficiency of the catalytic system means that lower loadings of the precious palladium metal may be feasible, directly impacting the cost of goods sold for the final API intermediate.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the simplification of the synthetic route. Eliminating multiple synthetic steps reduces labor costs, energy consumption, and solvent usage, leading to a drastically simplified cost structure. The ability to use inexpensive, off-the-shelf aryl halides instead of specialized chiral building blocks removes a major cost driver associated with traditional ligand synthesis. Additionally, the high yields reported in the patent examples suggest minimal material loss, further enhancing the overall process economy. This cost efficiency allows pharmaceutical companies to allocate resources to other critical areas of drug development while maintaining competitive pricing for their final products.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the modular nature of the ligand synthesis. Since the core structure is built from widely available industrial chemicals, the risk of single-source dependency is minimized. Manufacturers can source precursors from multiple global suppliers, ensuring continuity of supply even if one vendor faces production issues. The robustness of the reaction conditions, which tolerate standard laboratory equipment and do not require cryogenic temperatures or ultra-high pressures, facilitates production in diverse geographic locations. This flexibility enables regional manufacturing strategies, reducing lead times and logistics costs associated with transporting hazardous or sensitive chiral materials across borders.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is facilitated by the use of standard organic solvents like acetonitrile and toluene, which are well-understood in industrial settings. The reaction temperatures around 105°C are easily achievable with standard heating media such as steam or thermal oil, avoiding the need for specialized cooling or heating infrastructure. From an environmental standpoint, the atom economy of the coupling reaction is favorable, and the reduction step generates manageable byproducts. The high selectivity of the reaction reduces the burden on downstream purification processes, minimizing waste generation and aligning with green chemistry principles increasingly demanded by regulatory bodies and corporate sustainability goals.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral phosphine-ene ligand technology. These insights are derived directly from the patent specifications and are intended to assist technical teams in evaluating the feasibility of adopting this catalyst for their specific process needs. Understanding these nuances is key to leveraging the full potential of the ligand in asymmetric synthesis.
Q: What are the primary advantages of this novel axial chiral ligand over conventional options?
A: Unlike conventional ligands that often require complex, multi-step syntheses from the chiral pool, this novel ligand features a simple structure derived from readily available aryl iodides and bromides. Its modular design allows for easy structural modification, and it demonstrates superior catalytic activity and enantioselectivity under mild reaction conditions.
Q: What specific reaction conditions are required for the ligand synthesis?
A: The synthesis involves a two-step process. The first step utilizes a palladium catalyst (such as Pd(OAc)2) and a chiral norbornene derivative in acetonitrile at 105°C. The second step involves reduction with trichlorosilane and triethylamine in toluene, also at 105°C, ensuring a robust and scalable process.
Q: Which types of nucleophiles are compatible with this ligand in asymmetric substitution?
A: This ligand exhibits broad substrate scope in palladium-catalyzed asymmetric allylic substitution. It effectively facilitates reactions with diverse nucleophiles including indoles, malonates, amines (such as morpholine), and alcohols (such as benzyl alcohol), delivering products with high yields and excellent enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine-Ene Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the axial chiral phosphine-ene ligand described in CN111718372B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and residual metal content. We understand that consistency is paramount in pharmaceutical manufacturing, and our dedicated process development teams work tirelessly to optimize every parameter of the synthesis to meet your exacting standards.
We invite you to collaborate with us to unlock the full value of this innovative ligand for your pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Whether you need specific COA data for regulatory filings or comprehensive route feasibility assessments to de-risk your scale-up, we are here to support your success. Contact us today to discuss how our expertise in chiral catalysis can accelerate your project and reduce your time to market.
