Advanced Axial Chiral Phosphine-Ene Ligands for Efficient Asymmetric Synthesis and Commercial Manufacturing
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the demand for more efficient and accessible chiral ligands. A pivotal advancement in this field is detailed in patent CN111718372B, which discloses a novel class of axial chiral phosphine-ene ligands characterized by a robust biaryl skeleton. These ligands address critical bottlenecks in modern organic synthesis by combining the weak coordination capacity of a carbon-carbon double bond with the strong coordination of a phosphorus atom. This unique dual-coordination capability allows for superior control over stereoselectivity in transition metal-catalyzed reactions. For R&D directors and process chemists, this represents a shift away from convoluted ligand architectures toward modular, easily tunable systems that can be synthesized from commodity chemicals. The technology promises to streamline the production of high-value chiral intermediates essential for the pharmaceutical and agrochemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral phosphine-ene ligands has been hindered by synthetic complexity and limited structural diversity. Since their initial reports in the early 2000s, many existing ligands have required laborious multi-step syntheses involving expensive chiral pool starting materials or difficult resolution processes. These conventional methods often suffer from poor atom economy and generate significant waste, which poses challenges for both cost efficiency and environmental compliance in large-scale manufacturing. Furthermore, the rigid structures of many traditional ligands limit their adaptability to different substrate classes, often necessitating the design of entirely new ligand families for specific transformations. This lack of modularity increases the time and resources required for process development, creating a barrier to rapid innovation in drug discovery and fine chemical production.
The Novel Approach
The methodology presented in the patent offers a groundbreaking solution by utilizing a palladium-catalyzed Catellani-type reaction to construct the axially chiral biaryl framework directly from simple aryl iodides, aryl bromides, and alkenes. This approach bypasses the need for pre-functionalized chiral building blocks, allowing for the rapid assembly of complex ligand architectures in just two steps. The ability to easily modify the aryl and alkene components means that chemists can fine-tune the steric and electronic properties of the ligand to optimize performance for specific reactions without redesigning the entire synthetic route. This modularity not only accelerates the screening process but also ensures that the resulting ligands possess the necessary stability and activity for demanding industrial applications, effectively bridging the gap between academic innovation and commercial viability.
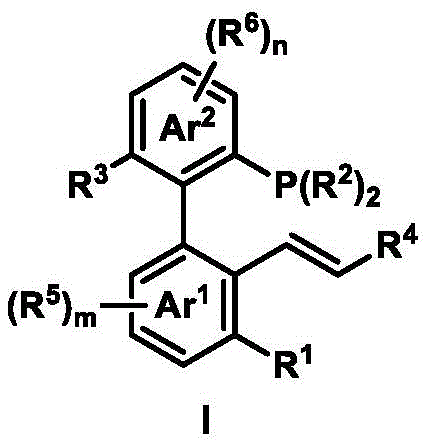
Mechanistic Insights into Palladium-Catalyzed Asymmetric Allylic Substitution
The core utility of these axial chiral phosphine-ene ligands lies in their application for palladium-catalyzed asymmetric allylic substitution reactions. Mechanistically, the ligand coordinates to the palladium center through both the phosphorus atom and the alkene moiety, creating a rigid chiral environment around the metal. This dual coordination stabilizes the pi-allyl palladium intermediate and directs the approach of the nucleophile with high precision. The axial chirality of the biaryl backbone serves as the primary source of stereoinduction, effectively differentiating the enantiotopic faces of the allylic system. This results in the formation of chiral products with exceptional enantiomeric excess, often exceeding 90% ee even with diverse nucleophiles such as indoles, malonates, and amines. The mild reaction conditions, typically proceeding at room temperature in common solvents like dichloromethane, further underscore the robustness of this catalytic system.

From an impurity control perspective, the high selectivity of this catalyst system significantly reduces the formation of regioisomers and byproducts that are common in non-enantioselective processes. The specific geometry imposed by the bulky biaryl groups prevents unwanted side reactions, ensuring a cleaner reaction profile. This purity is critical for downstream processing in pharmaceutical manufacturing, where strict limits on impurities are enforced. By minimizing the generation of difficult-to-separate stereoisomers, the process simplifies purification workflows, reducing the reliance on extensive chromatography or recrystallization steps. This mechanistic efficiency translates directly into higher overall yields and reduced material loss, making the technology highly attractive for the synthesis of complex active pharmaceutical ingredients where every percentage point of yield impacts the bottom line.
How to Synthesize Axial Chiral Phosphine-Ene Ligand Efficiently
The synthesis of these high-performance ligands is achieved through a streamlined two-step protocol that leverages readily available starting materials. The process begins with a palladium-catalyzed coupling reaction involving an aryl iodide, an aryl bromide containing a phosphine oxide group, and an alkene, mediated by a chiral norbornene derivative. This key step establishes the axially chiral biaryl bond with high stereocontrol. Following the initial coupling, the phosphine oxide intermediate undergoes a deoxygenation reaction using trichlorosilane and triethylamine to yield the final active phosphine ligand. This route is notable for its operational simplicity and scalability, avoiding the need for exotic reagents or extreme conditions. Detailed standardized synthesis steps for implementing this protocol in your facility are provided in the guide below.
- Perform a palladium-catalyzed coupling of aryl iodide, aryl bromide, and alkene using a chiral norbornene derivative to form the phosphine oxide intermediate.
- Reduce the phosphine oxide intermediate using trichlorosilane and triethylamine in toluene at elevated temperatures to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology offers substantial strategic advantages rooted in cost efficiency and supply reliability. The reliance on simple, commodity-grade starting materials such as aryl halides and styrenes eliminates the volatility associated with sourcing complex, proprietary chiral building blocks. This shift to a modular synthesis model ensures a more stable supply chain, as the raw materials are widely produced by multiple chemical manufacturers globally. Consequently, the risk of supply disruption due to single-source dependency is drastically minimized, providing greater security for long-term production planning. Furthermore, the simplified two-step synthesis reduces the overall manufacturing footprint, requiring fewer unit operations and less specialized equipment compared to traditional ligand production methods.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven by the elimination of expensive chiral resolution steps and the use of low-cost feedstocks. By constructing the chiral axis directly during the synthesis rather than resolving a racemic mixture, the process inherently avoids the 50% yield loss typical of resolution methods. Additionally, the high catalytic activity allows for lower catalyst loading in downstream applications, further reducing the consumption of precious palladium metals. These factors combine to deliver significant cost savings in the production of chiral intermediates, enhancing the overall margin profile for fine chemical manufacturing without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent quality and availability, which is paramount for maintaining continuous production schedules. The use of standard solvents like acetonitrile and toluene, along with common bases such as potassium carbonate, means that the process can be easily replicated across different manufacturing sites without requiring specialized infrastructure. This flexibility allows for diversified production strategies, enabling companies to mitigate regional risks and optimize logistics. The high yields reported in the patent examples indicate a mature and reliable process that minimizes batch failures, ensuring that delivery commitments to downstream customers are met consistently and reliably.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with green chemistry principles by reducing waste generation and energy consumption. The mild reaction conditions, particularly for the application step which proceeds at room temperature, lower the energy demand for heating and cooling. Moreover, the high selectivity reduces the burden on waste treatment facilities by minimizing the volume of hazardous byproducts. This ease of scale-up, combined with a favorable environmental profile, facilitates faster regulatory approval and smoother technology transfer from pilot plant to commercial scale, accelerating time-to-market for new products derived from this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral phosphine-ene ligand technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for evaluating the feasibility of integrating this catalyst system into existing production workflows. The answers provide clarity on performance metrics, substrate scope, and operational parameters to support informed decision-making for R&D and procurement teams.
Q: What distinguishes this axial chiral phosphine-ene ligand from conventional chiral ligands?
A: Unlike traditional ligands that often require complex multi-step syntheses or resolution of racemates, this novel ligand features a robust axially chiral biaryl skeleton constructed directly from simple aryl halides and alkenes. This modular approach allows for easy structural modification and avoids the costly purification steps associated with older generation ligands.
Q: What represent the primary industrial applications for this catalyst system?
A: The ligand is specifically designed for palladium-catalyzed asymmetric allylic substitution reactions. It demonstrates exceptional versatility with various nucleophiles including indoles, malonates, amines, and alcohols, making it highly valuable for constructing chiral centers in active pharmaceutical ingredients (APIs) and fine chemical intermediates.
Q: Is the synthesis of this ligand suitable for large-scale production?
A: Yes, the preparation method utilizes readily available starting materials such as aryl iodides and bromides and operates under relatively mild conditions. The two-step process avoids extreme pressures or cryogenic temperatures, facilitating straightforward scale-up from laboratory to commercial manufacturing volumes while maintaining high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Phosphine-Ene Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies in driving innovation within the fine chemical and pharmaceutical industries. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN111718372B can be successfully translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or intermediate delivered meets the highest international standards. We are committed to supporting our partners in overcoming synthetic challenges through our deep expertise in organometallic chemistry and process optimization.
We invite you to collaborate with us to unlock the full commercial potential of this novel ligand technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this methodology can enhance your operational efficiency. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your development timeline and secure a competitive advantage in the global market through superior catalytic solutions.
