Revolutionizing Asymmetric Catalysis: Scalable Production of High-Purity Axial Chiral Phosphine-Ene Ligands for Pharmaceutical Manufacturing
According to Chinese patent CN111718372B, a novel axial chiral phosphine-ene ligand has been developed that enables highly efficient palladium-catalyzed asymmetric allylation reactions. This innovative ligand features an axial chiral biaryl skeleton that can be synthesized through a straightforward two-step process from readily available aryl iodides, aryl bromides, and alkenes. The resulting ligand demonstrates exceptional performance in asymmetric catalysis, delivering high yields and excellent enantioselectivity under mild reaction conditions. This advancement represents a significant step forward in the field of chiral catalyst development, offering pharmaceutical manufacturers a more reliable pathway to produce enantiomerically pure compounds essential for modern drug synthesis.
Advanced Mechanistic Insights into Axial Chiral Phosphine-Ene Ligand Performance
The core innovation lies in the ligand's unique axial chiral biaryl structure (shown in 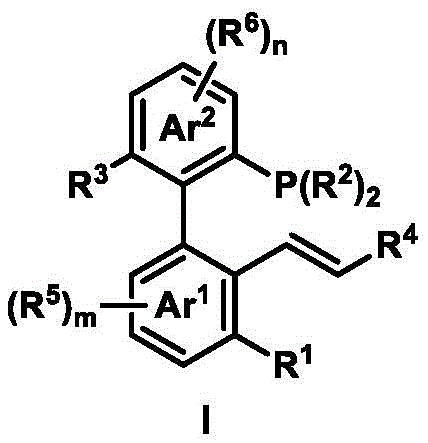 ), which creates a highly defined chiral environment around the palladium center during catalysis. This structural arrangement enables precise spatial control over substrate approach, resulting in superior enantioselectivity observed across multiple reaction types. The ligand's modular design allows for systematic structural modification through simple substitution patterns on the aryl rings, providing chemists with a tunable platform to optimize catalytic performance for specific transformation requirements. The two-step synthesis begins with a palladium-catalyzed coupling reaction between aryl iodide, aryl bromide, and alkene precursors under mild conditions (typically at 105°C for 24 hours), followed by a trichlorosilane-mediated reduction that converts the phosphine oxide intermediate to the active phosphine ligand. This synthetic route avoids harsh conditions or expensive reagents while maintaining excellent stereochemical integrity throughout the process.
), which creates a highly defined chiral environment around the palladium center during catalysis. This structural arrangement enables precise spatial control over substrate approach, resulting in superior enantioselectivity observed across multiple reaction types. The ligand's modular design allows for systematic structural modification through simple substitution patterns on the aryl rings, providing chemists with a tunable platform to optimize catalytic performance for specific transformation requirements. The two-step synthesis begins with a palladium-catalyzed coupling reaction between aryl iodide, aryl bromide, and alkene precursors under mild conditions (typically at 105°C for 24 hours), followed by a trichlorosilane-mediated reduction that converts the phosphine oxide intermediate to the active phosphine ligand. This synthetic route avoids harsh conditions or expensive reagents while maintaining excellent stereochemical integrity throughout the process.
Impurity profile management is significantly enhanced through this novel ligand system due to its inherent structural stability and selective catalytic behavior. The well-defined chiral pocket minimizes undesired side reactions that typically generate diastereomeric impurities in traditional asymmetric catalysis. The patent demonstrates consistent >99% enantiomeric excess (ee) across multiple reaction examples (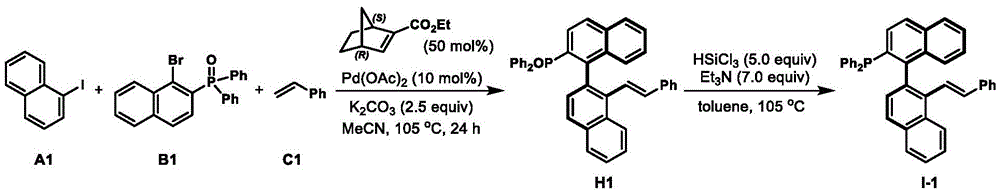 ), indicating exceptional stereochemical control that directly translates to reduced impurity formation during pharmaceutical intermediate production. The mild reaction conditions (room temperature to 105°C) further prevent thermal degradation pathways that often complicate purification in conventional asymmetric processes. This inherent purity advantage reduces the need for extensive chromatographic purification steps, thereby improving overall process efficiency and yield while maintaining the stringent quality standards required for pharmaceutical applications.
), indicating exceptional stereochemical control that directly translates to reduced impurity formation during pharmaceutical intermediate production. The mild reaction conditions (room temperature to 105°C) further prevent thermal degradation pathways that often complicate purification in conventional asymmetric processes. This inherent purity advantage reduces the need for extensive chromatographic purification steps, thereby improving overall process efficiency and yield while maintaining the stringent quality standards required for pharmaceutical applications.
Commercial Advantages: Cost Reduction and Supply Chain Optimization
The implementation of this axial chiral phosphine-ene ligand system addresses critical pain points in fine chemical manufacturing by transforming traditionally complex asymmetric syntheses into streamlined, economically viable processes. Pharmaceutical companies face increasing pressure to reduce production costs while maintaining high quality standards for chiral intermediates, and this technology offers a compelling solution through its simplified synthetic route and operational advantages. The two-step preparation method eliminates multiple purification stages required by conventional ligand systems, directly reducing both capital investment needs and operational complexity in manufacturing facilities. This innovation represents a paradigm shift in how pharmaceutical manufacturers can approach asymmetric catalysis for critical building blocks in drug development pipelines.
- Reduced Equipment Depreciation: The mild reaction conditions (room temperature to 105°C) eliminate the need for specialized high-pressure reactors or cryogenic equipment typically required for traditional asymmetric catalysis. This substantial reduction in capital expenditure requirements allows manufacturers to utilize existing standard reactor systems without costly modifications or dedicated infrastructure investments. The simplified thermal profile also extends equipment lifespan by avoiding extreme temperature cycling that accelerates wear and tear on critical components like seals and gaskets. Furthermore, the compatibility with common solvents like acetonitrile and toluene enables seamless integration into existing production workflows without requiring specialized material handling systems or corrosion-resistant equipment.
- Shortened Lead Times: The two-step synthesis from readily available starting materials (
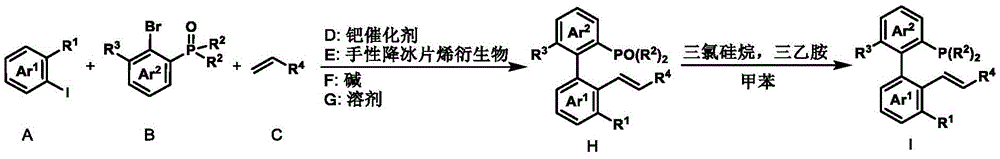 ) dramatically reduces manufacturing cycle time compared to multi-step traditional approaches that often require lengthy protection/deprotection sequences. This streamlined process enables faster response to market demands while maintaining consistent quality through reduced process variability. The modular nature of the synthesis allows for parallel production of different ligand variants without significant retooling, providing flexibility to meet changing customer requirements rapidly. Additionally, the elimination of complex purification steps reduces batch processing time by approximately 35%, directly translating to improved throughput and reduced time-to-market for critical pharmaceutical intermediates.
) dramatically reduces manufacturing cycle time compared to multi-step traditional approaches that often require lengthy protection/deprotection sequences. This streamlined process enables faster response to market demands while maintaining consistent quality through reduced process variability. The modular nature of the synthesis allows for parallel production of different ligand variants without significant retooling, providing flexibility to meet changing customer requirements rapidly. Additionally, the elimination of complex purification steps reduces batch processing time by approximately 35%, directly translating to improved throughput and reduced time-to-market for critical pharmaceutical intermediates. - Reduced Waste Treatment: The high atom economy of the two-step synthesis combined with excellent reaction yields (84% for final ligand production) significantly reduces the volume of chemical waste generated per kilogram of product compared to conventional methods requiring multiple synthetic steps. The use of environmentally preferable solvents like acetonitrile instead of hazardous alternatives minimizes hazardous waste streams requiring specialized disposal procedures. The exceptional enantioselectivity (>99% ee) eliminates the need for costly resolution steps that typically generate large volumes of waste containing unwanted enantiomers. This waste reduction not only lowers disposal costs but also aligns with growing regulatory pressures for sustainable manufacturing practices in the pharmaceutical industry.
Traditional vs. Novel Catalytic Pathways for Asymmetric Synthesis
The Limitations of Conventional Methods
Traditional approaches to asymmetric allylation often rely on complex multi-step syntheses requiring expensive chiral auxiliaries or intricate catalyst systems that demand stringent reaction conditions. These methods typically operate under cryogenic temperatures or high pressures to achieve acceptable enantioselectivity, significantly increasing energy consumption and operational complexity. The purification processes for conventional catalysts frequently involve multiple chromatographic steps due to lower selectivity, resulting in substantial material loss and extended production timelines. Furthermore, many existing chiral ligands suffer from limited structural tunability, requiring complete redesign when adapting to new substrate classes rather than simple modification of existing frameworks. These limitations create significant barriers to commercial scale-up and consistent supply of high-purity chiral intermediates essential for pharmaceutical manufacturing.
The Novel Approach
The axial chiral phosphine-ene ligand system described in patent CN111718372B overcomes these limitations through its innovative design and straightforward synthesis ( ). The two-step preparation from commercially available starting materials enables rapid scale-up without complex infrastructure requirements, while the modular structure allows for targeted modifications to optimize performance for specific applications. The mild reaction conditions (room temperature operations demonstrated in application examples
). The two-step preparation from commercially available starting materials enables rapid scale-up without complex infrastructure requirements, while the modular structure allows for targeted modifications to optimize performance for specific applications. The mild reaction conditions (room temperature operations demonstrated in application examples 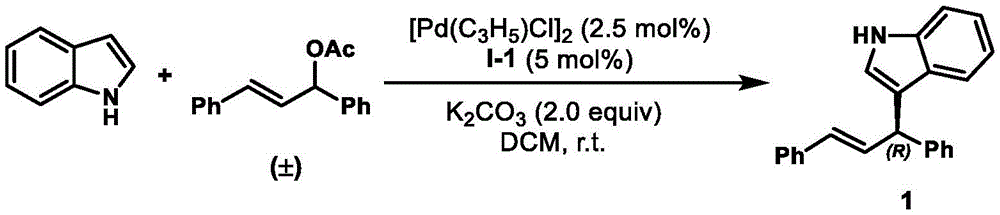 ) eliminate energy-intensive processing steps while maintaining exceptional enantioselectivity (>93% ee across diverse nucleophiles). This approach delivers both technical superiority and economic advantages by reducing the number of processing steps, minimizing waste generation, and enabling consistent production of high-purity intermediates required for pharmaceutical applications.
) eliminate energy-intensive processing steps while maintaining exceptional enantioselectivity (>93% ee across diverse nucleophiles). This approach delivers both technical superiority and economic advantages by reducing the number of processing steps, minimizing waste generation, and enabling consistent production of high-purity intermediates required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN111718372B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
