Industrial Scale Synthesis of Iodobenzene Diacetate via Safe Oxidation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, safe, and scalable synthetic routes for critical oxidizing agents, and the technology disclosed in patent CN101575293A represents a significant leap forward in the manufacturing of iodobenzene diacetate. This hypervalent iodine compound serves as a pivotal reagent in the synthesis of complex active pharmaceutical ingredients, offering selective oxidation capabilities that traditional heavy metal oxidants cannot match without generating toxic waste. The core innovation lies in replacing hazardous liquid peracetic acid with stable sodium perborate tetrahydrate, fundamentally altering the safety profile and logistical feasibility of large-scale production. By utilizing a mixed solvent system of glacial acetic acid and acetic anhydride, this method achieves high conversion rates under mild thermal conditions, effectively mitigating the explosion risks that have historically constrained the supply chain of this valuable intermediate. For global procurement teams and R&D directors, this patent outlines a pathway to secure a reliable iodobenzene diacetate supplier capable of meeting stringent safety and purity standards required for GMP environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of iodobenzene diacetate has been heavily reliant on the use of high-concentration peracetic acid, typically requiring concentrations upwards of 40% to drive the oxidation of iodobenzene efficiently. This traditional approach presents severe safety hazards, as peracetic acid at such concentrations is inherently unstable and possesses significant explosive potential during storage, transportation, and handling within a reactor. Furthermore, the market availability of peracetic acid above 40% is extremely limited due to strict regulatory controls on hazardous chemicals, forcing manufacturers to synthesize it in situ or deal with complex logistics that increase lead time for high-purity pharmaceutical intermediates. The inherent instability of the oxidant often leads to inconsistent reaction kinetics, resulting in lower yields and the formation of difficult-to-remove byproducts that compromise the quality of the final API intermediate. These factors collectively create a bottleneck in the commercial scale-up of complex polymer additives and pharmaceutical building blocks, driving up costs and introducing unacceptable risks to the manufacturing facility.
The Novel Approach
In stark contrast, the novel methodology utilizes sodium perborate tetrahydrate, a stable, solid-state oxidizing agent that is widely available and safe to handle, effectively eliminating the volatility associated with liquid peracids. By reacting this solid oxidant with iodobenzene in a carefully optimized mixture of glacial acetic acid and acetic anhydride, the process generates the necessary oxidative species in a controlled manner without the risk of thermal runaway or explosion. This shift from a hazardous liquid reagent to a stable solid allows for much simpler operational procedures, reducing the need for specialized explosion-proof equipment and lowering the barrier for cost reduction in electronic chemical manufacturing and pharma sectors. The ability to operate at moderate temperatures between 30°C and 45°C further enhances energy efficiency and safety, making this route exceptionally suitable for industrial mass production where consistency and operator safety are paramount concerns for any responsible chemical enterprise.
Mechanistic Insights into Sodium Perborate Mediated Oxidation
The chemical mechanism underlying this transformation involves the in-situ generation of reactive oxygen species facilitated by the interaction between sodium perborate and the acetic acid/anhydride solvent system. Upon addition to the acidic medium, the perborate releases hydrogen peroxide, which subsequently reacts with acetic anhydride to form peracetic acid locally within the reaction mixture, immediately consuming it to oxidize the iodine center of the iodobenzene substrate. This "just-in-time" generation of the active oxidant prevents the accumulation of dangerous levels of peracids, thereby maintaining a safe reaction profile while ensuring high atom economy. The presence of acetic anhydride is critical not only as a dehydrating agent to drive the equilibrium towards the diacetate product but also as a participant in the acylation step that stabilizes the hypervalent iodine structure. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize the process for specific impurity profiles, as the ratio of anhydride to acid directly influences the rate of acylation versus hydrolysis side reactions.
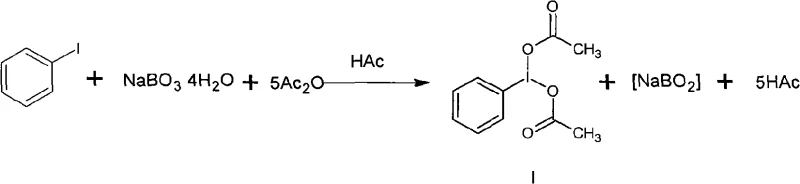
Controlling the impurity profile in the synthesis of high-purity OLED material or pharmaceutical intermediates requires precise management of the reaction stoichiometry and workup conditions. The patent specifies a molar ratio of sodium perborate to iodobenzene ranging from 3:1 to 10:1, ensuring a substantial excess of oxidant to drive the reaction to completion while minimizing unreacted starting material. Following the reaction, the addition of ice water serves a dual purpose: it quenches any remaining reactive species and induces the precipitation of the crude product, leveraging the solubility differences between the product and inorganic salts. Subsequent recrystallization from a glacial acetic acid and petroleum ether system is employed to remove trace organic impurities and residual iodine species, yielding a product with exceptional purity suitable for sensitive downstream applications. This rigorous purification protocol ensures that the final material meets the stringent specifications required by top-tier pharmaceutical clients who demand consistent quality in their supply chain.
How to Synthesize Iodobenzene Diacetate Efficiently
Implementing this synthesis route requires careful attention to the addition rate of the solid oxidant and the maintenance of the reaction temperature within the narrow window of 30°C to 45°C to maximize yield and safety. The process begins by preparing the solvent system, followed by the batched addition of sodium perborate to control the exotherm and ensure uniform mixing throughout the reaction vessel. Detailed standard operating procedures regarding stirring speeds, cooling capacities, and filtration techniques are essential to replicate the high yields reported in the patent embodiments consistently. For a comprehensive understanding of the specific operational parameters and step-by-step instructions required for successful implementation, please refer to the standardized synthesis guide provided below.
- Prepare the reaction mixture by combining iodobenzene with a glacial acetic acid and acetic anhydride solution.
- Add sodium perborate tetrahydrate in batches while maintaining temperature between 30°C and 45°C.
- Quench with ice water and recrystallize the crude product using glacial acetic acid and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, adopting this sodium perborate-based methodology offers profound advantages in terms of supply chain resilience and total cost of ownership for chemical manufacturing. By removing the dependency on hazardous high-concentration peracetic acid, companies can significantly reduce insurance premiums, safety compliance costs, and the logistical complexities associated with transporting dangerous goods across international borders. The use of stable, solid raw materials simplifies inventory management and warehousing requirements, allowing for larger stockpiles without the degradation risks inherent to liquid peroxides, thus ensuring continuous production capability even during market fluctuations. This stability translates directly into enhanced supply chain reliability, as manufacturers are less vulnerable to disruptions caused by regulatory changes or transportation accidents involving hazardous chemicals.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents leads to substantial cost savings by reducing the need for specialized containment systems and emergency response infrastructure. Furthermore, the simplified workup procedure minimizes solvent consumption and waste treatment costs, contributing to a more lean and efficient production model that aligns with modern sustainability goals. The ability to recycle byproduct iodobenzene further enhances the economic viability of the process, creating a closed-loop system that maximizes raw material utilization and minimizes waste disposal fees.
- Enhanced Supply Chain Reliability: Sourcing stable solid oxidants like sodium perborate is significantly more straightforward than procuring regulated liquid peracids, leading to drastically simplified procurement cycles and reduced lead times. This reliability ensures that production schedules can be maintained without interruption, providing downstream customers with a consistent flow of high-quality intermediates essential for their own manufacturing timelines. The robustness of the supply chain is further bolstered by the widespread availability of the raw materials, reducing the risk of single-source dependency.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal catalysts make this process inherently greener and easier to scale from pilot plant to full commercial production without significant re-engineering. The reduction in toxic waste streams simplifies environmental compliance and permitting processes, allowing facilities to expand capacity more rapidly in response to market demand. This scalability ensures that the technology can support the growing global demand for hypervalent iodine reagents in diverse applications ranging from agrochemicals to advanced materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology for iodobenzene diacetate. These insights are derived directly from the patent data and practical industry experience, aimed at clarifying the operational advantages and safety improvements offered by this method. Understanding these details is crucial for technical decision-makers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is sodium perborate preferred over peracetic acid for PIDA synthesis?
A: Sodium perborate is a stable solid that eliminates the explosion risks associated with transporting and storing high-concentration peracetic acid.
Q: What is the typical yield of this novel oxidation method?
A: Optimized embodiments demonstrate yields exceeding 70%, significantly higher than conventional methods lacking acetic anhydride activation.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (30-45°C) and use of stable solid reagents make it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodobenzene Diacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of safety and quality in the production of sensitive oxidizing agents like iodobenzene diacetate, and we are fully equipped to leverage this advanced patent technology for your commercial needs. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements without compromising on our stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical techniques to verify every batch, guaranteeing that the material you receive is consistent, safe, and ready for immediate use in your most demanding synthetic applications.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific project requirements and provide a Customized Cost-Saving Analysis for your supply chain. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your raw material strategy. Let us help you secure a stable, high-quality supply of iodobenzene diacetate that drives innovation and efficiency in your manufacturing operations.
