Revolutionizing Iodobenzene Diacetate Production: A Safe, Scalable Commercial Route
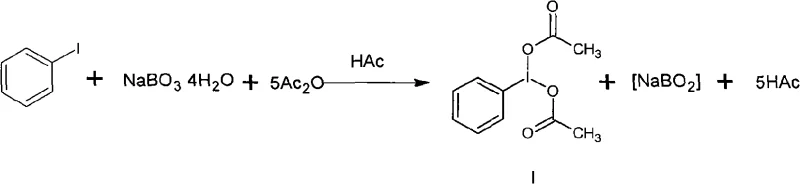
The chemical industry is currently witnessing a pivotal shift towards safer, more sustainable oxidation methodologies, particularly for high-value intermediates like iodobenzene diacetate. Patent CN101575293A introduces a groundbreaking preparation method that fundamentally alters the risk profile of synthesizing this critical hypervalent iodine reagent. By replacing hazardous concentrated peracetic acid with stable sodium perborate tetrahydrate, this technology addresses long-standing safety concerns that have plagued the manufacturing of organic iodine oxidants. For R&D directors and procurement specialists, this represents not just a chemical improvement, but a strategic supply chain advantage, enabling the reliable sourcing of high-purity iodobenzene diacetate without the logistical nightmares associated with explosive precursors. The patent details a robust acylation reaction performed in a glacial acetic acid and acetic anhydride mixed solution, operating under mild thermal conditions that are inherently easier to control on a multi-ton scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of iodobenzene diacetate has been bottlenecked by the reliance on peracetic acid with concentrations exceeding 40%, a reagent known for its extreme instability and explosive potential. The background technology section of the patent highlights that storing, transporting, and utilizing such high-concentration peroxides introduces severe safety hazards that can destabilize production facilities and endanger personnel. Furthermore, the market availability of peracetic acid above 40% is extremely limited, with most commercial supplies capped at 18-23% concentration, which is insufficient for efficient synthesis without complex concentration steps. These constraints have severely restricted the industrial scale of traditional production, leading to low output volumes and inconsistent supply for downstream pharmaceutical applications. The inherent danger of the old process also drives up insurance and compliance costs, making the final product economically inefficient for large-scale API manufacturing.
The Novel Approach
The innovative method described in the patent circumvents these dangers by utilizing sodium perborate tetrahydrate, a stable, solid oxidant widely available and commonly used in the detergent industry. This approach generates the necessary oxidative species in situ within a glacial acetic acid and acetic anhydride medium, effectively bypassing the need to handle bulk liquid peroxides. The process operates at a mild temperature range of 30 to 45 degrees Celsius, significantly reducing energy consumption and thermal runaway risks compared to more aggressive oxidation protocols. By shifting to a solid-state oxidant, the method simplifies the operational workflow, allowing for batch additions that are easier to monitor and control. This transition from a hazardous liquid oxidant to a benign solid reagent transforms the production landscape, making it feasible to manufacture iodobenzene diacetate in substantial quantities with a dramatically improved safety profile.
Mechanistic Insights into Sodium Perborate Mediated Oxidation
The core of this technological advancement lies in the unique reactivity of sodium perborate tetrahydrate within an anhydrous acidic environment. In the presence of glacial acetic acid and acetic anhydride, the perborate acts as an oxygen donor, facilitating the oxidation of the iodine atom in iodobenzene from the +1 to the +3 oxidation state. The acetic anhydride plays a dual role: it serves as a dehydrating agent to drive the equilibrium forward and as an acylating agent to form the stable diacetate structure on the iodine center. This mechanism avoids the formation of free radical species that often lead to side reactions and impurity generation in less controlled oxidation systems. The stoichiometry is carefully balanced, with a molar ratio of sodium perborate to iodobenzene ranging from 3:1 to 10:1, ensuring complete conversion while minimizing waste.
Understanding the impurity profile is crucial for pharmaceutical grade materials, and this mechanism offers superior control over byproduct formation. Traditional methods using unstable peracids often result in over-oxidation or decomposition products that are difficult to separate. In contrast, the byproduct of this novel reaction is sodium metaborate and acetic acid, both of which are easily removed during the aqueous workup and recrystallization steps. The patent specifies recrystallization using a glacial acetic acid and petroleum ether system, which effectively purges inorganic salts and unreacted starting materials. This results in a final product with purity levels exceeding 99%, as evidenced by the melting point data and content analysis in the patent embodiments. For R&D teams, this means a cleaner reaction profile that reduces the burden on downstream purification processes.
How to Synthesize Iodobenzene Diacetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise temperature control and reagent addition rates. The process begins with the preparation of the solvent system, followed by the controlled addition of the solid oxidant to manage exotherms. Detailed standard operating procedures regarding stirring speeds, addition times, and quenching protocols are essential for replicating the high yields reported in the embodiments. For a comprehensive guide on the exact step-by-step execution of this synthesis, please refer to the standardized protocol below.
- Prepare a mixed solution of glacial acetic acid and acetic anhydride, then add iodobenzene and heat to 35-40°C.
- Batch add sodium perborate tetrahydrate over 30-60 minutes while maintaining the reaction temperature between 40-45°C.
- After reacting for 6-12 hours, pour into ice water to precipitate the crude product, then recrystallize using glacial acetic acid and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sodium perborate-based route offers transformative benefits that extend far beyond simple chemical yield. The primary advantage is the drastic reduction in supply chain risk associated with raw material sourcing. Unlike concentrated peracetic acid, which requires specialized hazardous material transport and storage infrastructure, sodium perborate tetrahydrate is a stable, non-hazardous solid that can be sourced globally from multiple suppliers. This diversification of the supply base ensures continuity of supply and protects against price volatility caused by regulatory crackdowns on explosive precursors. Furthermore, the elimination of high-risk reagents simplifies facility permitting and reduces the capital expenditure required for blast-proof reactors and containment systems.
- Cost Reduction in Manufacturing: The shift to sodium perborate significantly lowers the total cost of ownership for producing iodobenzene diacetate. By removing the need for expensive, high-concentration peracetic acid and the associated safety infrastructure, manufacturers can achieve substantial cost savings in both raw material procurement and facility operations. The process utilizes acetic anhydride and glacial acetic acid, which are commodity chemicals available at competitive prices, further driving down the variable cost per kilogram. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it by decoupling production from the volatile market of hazardous peroxides. Since sodium perborate is a stable solid with a long shelf life, manufacturers can maintain strategic stockpiles without degradation concerns, ensuring uninterrupted production even during upstream supply disruptions. The robustness of the reaction conditions also means fewer batch failures due to reagent instability, leading to more predictable delivery schedules for downstream clients. This stability allows for better production planning and inventory management, reducing the need for expedited shipping and emergency procurement.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles and regulatory trends. The byproducts are benign and easily treatable, reducing the environmental footprint of the manufacturing process and simplifying wastewater treatment requirements. The mild reaction temperatures and absence of explosive hazards make the process inherently scalable, allowing for seamless transition from kilogram to multi-ton production without major engineering redesigns. This scalability ensures that suppliers can rapidly ramp up capacity to meet surging demand for iodobenzene diacetate in the growing pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of iodobenzene diacetate using this patented methodology. These answers are derived directly from the technical specifications and comparative data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is critical for evaluating the feasibility of integrating this material into your specific synthetic pathways.
Q: Why is the sodium perborate method safer than traditional peracetic acid synthesis?
A: Traditional methods require concentrated peracetic acid (>40%), which is highly explosive and difficult to transport. The new method uses solid sodium perborate tetrahydrate, a stable oxidant commonly used in detergents, eliminating explosion risks and storage hazards.
Q: What is the typical yield and purity of iodobenzene diacetate using this patent method?
A: According to patent embodiments, the process achieves yields ranging from 66% to over 71%, with product purity consistently exceeding 99% after recrystallization, making it suitable for high-grade pharmaceutical applications.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly highlights industrial suitability. The use of stable solid raw materials and moderate temperatures (30-45°C) allows for safe scaling without the complex safety infrastructure required for handling explosive liquid peroxides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodobenzene Diacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality oxidizing agents play in the synthesis of complex pharmaceutical intermediates. Our technical team has extensively analyzed the pathway described in CN101575293A and possesses the expertise to implement this safer, more efficient route at commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify every batch against the highest international standards, guaranteeing that the iodobenzene diacetate we supply is free from the heavy metal contaminants often associated with older oxidation technologies.
We invite you to collaborate with us to optimize your supply chain for hypervalent iodine reagents. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and production goals efficiently and safely.
