Advanced Beta-Lactam Intermediates for Scalable Taxane Manufacturing and Cost Reduction
Advanced Beta-Lactam Intermediates for Scalable Taxane Manufacturing and Cost Reduction
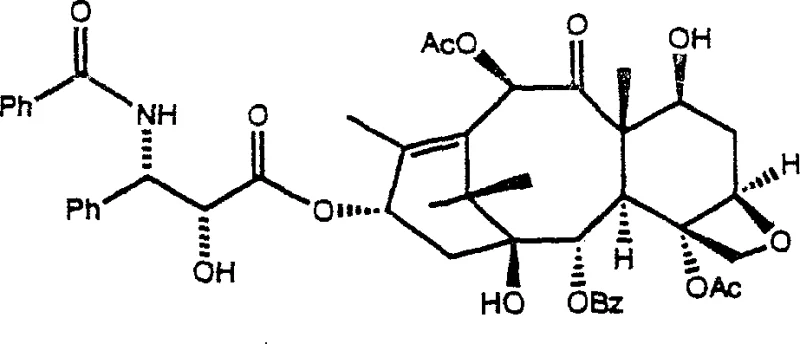
The global demand for potent anticancer agents like paclitaxel continues to drive innovation in semi-synthetic manufacturing pathways, particularly focusing on the efficiency and purity of key intermediates. Patent CN1080256C introduces a groundbreaking methodology involving novel beta-lactam compounds that serve as critical precursors in the preparation of sidechain-bearing taxanes. These intermediates address longstanding challenges in the industry regarding stereochemical control and the physical state of reactants, shifting from difficult-to-handle oils to stable crystalline solids. By leveraging specific protecting groups such as the 1-methoxy-1-methylethoxy moiety, this technology enables the production of taxane derivatives with significantly reduced impurity profiles. For pharmaceutical manufacturers, adopting this patented approach represents a strategic move towards more robust and reliable supply chains for high-value oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing taxane sidechains often rely on beta-lactam intermediates that exist as liquids or oils at room temperature, creating substantial bottlenecks in large-scale processing. These liquid forms are notoriously difficult to purify to the stringent standards required for active pharmaceutical ingredients, often trapping solvent residues and impurities that complicate downstream reactions. Furthermore, conventional routes frequently generate mixtures of stereoisomers due to the presence of asymmetric carbon atoms at critical positions, necessitating complex and yield-reducing separation steps. The inefficiency of starting material usage in these older processes leads to higher raw material costs and increased waste generation, which is unsustainable for modern green chemistry initiatives. Additionally, the instability of certain liquid intermediates can result in degradation during storage or transport, further compromising the consistency of the final drug product.
The Novel Approach
The innovative strategy outlined in the patent utilizes specifically designed beta-lactam structures that crystallize readily, offering a transformative advantage in handling and purification efficiency. By introducing substituents where R1 and R2 form identical alkyl groups or cyclic structures, the resulting compounds exhibit superior physical properties that facilitate easy filtration and washing. 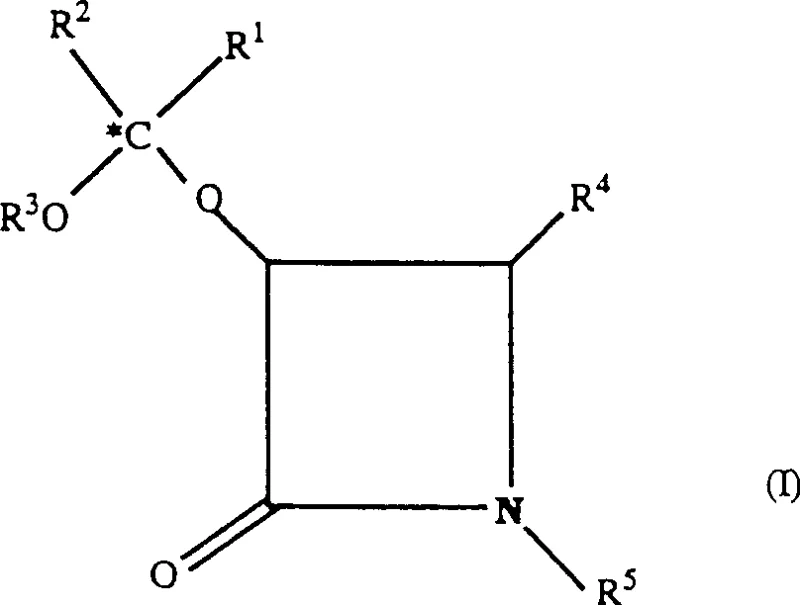 This crystalline nature ensures that the intermediates can be obtained in high purity forms, directly translating to cleaner coupling reactions with the taxane core. The method specifically avoids the formation of unwanted diastereomeric mixtures by controlling the stereochemistry at the marked carbon atoms, ensuring that the final taxane product possesses the exact configuration required for biological activity. This approach not only streamlines the manufacturing workflow but also significantly enhances the overall yield of the valuable final anticancer agent.
This crystalline nature ensures that the intermediates can be obtained in high purity forms, directly translating to cleaner coupling reactions with the taxane core. The method specifically avoids the formation of unwanted diastereomeric mixtures by controlling the stereochemistry at the marked carbon atoms, ensuring that the final taxane product possesses the exact configuration required for biological activity. This approach not only streamlines the manufacturing workflow but also significantly enhances the overall yield of the valuable final anticancer agent.
Mechanistic Insights into Acid-Catalyzed Protection and Coupling
The core of this technological advancement lies in the precise acid-catalyzed protection of the hydroxyl group on the beta-lactam ring, which is essential for maintaining stereochemical integrity during subsequent reactions. The process involves reacting a hydroxy-beta-lactam precursor with reagents like 2-methoxypropene in the presence of organic sulfonic acids such as pyridinium p-toluenesulfonate (PPTS). 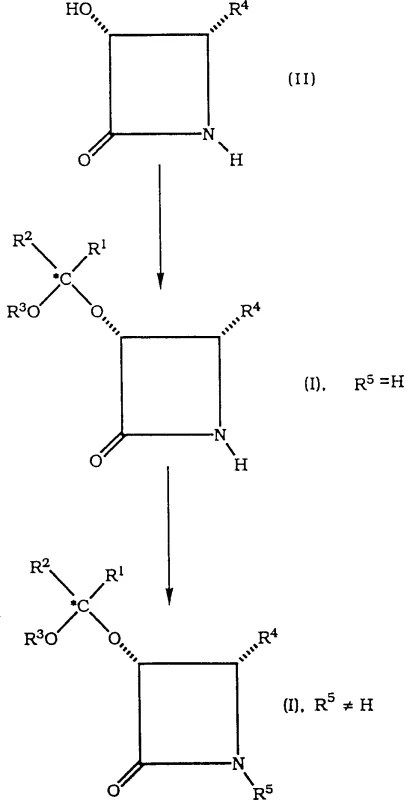 This reaction is carefully controlled at temperatures ranging from -30°C to 30°C, preferably around 0°C, to prevent racemization and ensure the formation of the desired cis-configuration. The choice of solvent, such as acetone or dichloromethane, plays a critical role in solubilizing the reactants while maintaining the stability of the acid catalyst. The mechanism effectively blocks the hydroxyl group from participating in unwanted side reactions, thereby preserving the chiral center that dictates the pharmacological efficacy of the final taxane molecule.
This reaction is carefully controlled at temperatures ranging from -30°C to 30°C, preferably around 0°C, to prevent racemization and ensure the formation of the desired cis-configuration. The choice of solvent, such as acetone or dichloromethane, plays a critical role in solubilizing the reactants while maintaining the stability of the acid catalyst. The mechanism effectively blocks the hydroxyl group from participating in unwanted side reactions, thereby preserving the chiral center that dictates the pharmacological efficacy of the final taxane molecule.
Following the formation of the protected intermediate, the coupling with the taxane moiety is executed using specialized bases that minimize substrate degradation. The patent highlights the use of alkali metal silylamide bases, particularly lithium hexamethyldisilylamide (LHMDS), which act as hindered, non-nucleophilic bases. This specific choice of base is crucial because it deprotonates the C-13 hydroxyl group of the baccatin III core without attacking the sensitive beta-lactam ring or the ester functionalities present in the molecule. The reaction proceeds efficiently at low temperatures, typically between -70°C and 0°C, ensuring that the kinetic control favors the formation of the desired ester linkage. This mechanistic precision results in coupling yields often exceeding 90%, with purity levels reaching upwards of 98%, demonstrating the robustness of the chemical pathway for industrial application.
How to Synthesize Novel Beta-Lactam Intermediates Efficiently
The synthesis of these high-value intermediates requires strict adherence to anhydrous conditions and precise temperature control to maximize yield and stereochemical purity. Operators must ensure that all solvents are dried thoroughly, often using molecular sieves or distillation over sodium, to prevent hydrolysis of the sensitive beta-lactam ring during the protection step. The addition of the protecting group reagent must be performed dropwise to manage exotherms and maintain the reaction mixture within the optimal temperature window specified in the protocol. Following the reaction, the crystallization process is a critical unit operation where the choice of anti-solvent, such as hexane added to an ethyl acetate solution, determines the crystal habit and purity of the isolated solid. Proper seeding and controlled cooling rates are essential to avoid oiling out and to ensure the formation of the thermodynamically stable crystalline polymorph.
- React a hydroxy-beta-lactam precursor with 2-methoxypropene in the presence of an acid catalyst like PPTS at 0°C to form the protected intermediate.
- Purify the resulting crystalline solid via recrystallization from ethyl acetate and hexane mixtures to ensure high stereochemical purity.
- Couple the purified beta-lactam with a baccatin III derivative using a hindered base like LHMDS at low temperatures to form the side-chain bearing taxane.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers profound economic and logistical benefits for organizations managing the procurement of complex pharmaceutical intermediates. The shift from liquid to crystalline intermediates fundamentally alters the cost structure of manufacturing by eliminating the need for extensive chromatographic purification steps that are both time-consuming and expensive. This simplification of the purification process directly correlates to a reduction in solvent consumption and waste disposal costs, aligning with increasingly strict environmental regulations in the chemical sector. Furthermore, the stability of the crystalline intermediates allows for longer shelf-life and safer transportation, reducing the risk of supply chain disruptions caused by material degradation. These factors collectively contribute to a more predictable and cost-effective production schedule for downstream API manufacturers.
- Cost Reduction in Manufacturing: The ability to produce intermediates in high purity through simple crystallization rather than complex chromatography leads to substantial operational expenditure savings. By avoiding the use of expensive silica gel columns and large volumes of elution solvents, manufacturers can significantly lower the variable costs associated with each batch production. The high yield of the coupling reaction, often reported above 90%, ensures that expensive starting materials like baccatin III are utilized with maximum efficiency, minimizing material loss. Additionally, the reduced need for reprocessing off-spec material due to high initial purity further drives down the overall cost of goods sold for the final taxane product.
- Enhanced Supply Chain Reliability: The robustness of the crystalline intermediates ensures a consistent supply of high-quality material, which is critical for maintaining continuous API production lines. Unlike liquid intermediates that may vary in quality or degrade over time, these solids provide a stable inventory buffer that can be stored safely for extended periods without significant loss of potency. This stability mitigates the risks associated with just-in-time delivery models, allowing procurement teams to build strategic stockpiles during favorable market conditions. The simplified synthesis route also reduces dependency on specialized contract manufacturing organizations capable of handling difficult liquid chemistries, thereby broadening the potential supplier base.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory to commercial tonnage due to the reliance on standard unit operations like crystallization and filtration rather than specialized separation techniques. The use of common organic solvents such as ethyl acetate and hexane facilitates easier solvent recovery and recycling, supporting sustainability goals within the manufacturing facility. Moreover, the high atom economy of the coupling reaction reduces the generation of hazardous waste streams, simplifying compliance with environmental discharge permits. This scalability ensures that the technology can meet growing global demand for paclitaxel without requiring disproportionate increases in manufacturing footprint or waste treatment capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel beta-lactam technology in taxane synthesis. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for technical decision-making.
Q: Why are crystalline beta-lactam intermediates preferred over liquid forms in taxane synthesis?
A: Crystalline intermediates offer significant advantages in large-scale production as they are easier to prepare, handle, and purify compared to liquid oils, leading to higher overall purity of the final taxane product.
Q: What is the role of the 1-methoxy-1-methylethoxy protecting group in this process?
A: This specific protecting group prevents the formation of asymmetric carbon centers that lead to diastereomeric mixtures, thereby simplifying isolation and ensuring the desired stereochemistry for pharmaceutical activity.
Q: How does the use of LHMDS improve the coupling reaction yield?
A: Lithium hexamethyldisilylamide (LHMDS) acts as a non-nucleophilic hindered base that minimizes degradation of the sensitive taxane substrate while facilitating efficient coupling at the C-13 position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Lactam Intermediates Supplier
NINGBO INNO PHARMCHEM stands at the forefront of delivering high-quality pharmaceutical intermediates, leveraging deep expertise in complex organic synthesis to support global drug development pipelines. Our facilities are equipped to handle the precise temperature controls and anhydrous conditions required for the production of sensitive beta-lactam compounds described in patent CN1080256C. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our rigorous QC labs enforce stringent purity specifications, utilizing advanced analytical techniques to verify stereochemical integrity and impurity profiles before shipment. This commitment to quality assurance makes us a trusted partner for pharmaceutical companies seeking to optimize their taxane supply chains.
We invite procurement leaders and technical directors to engage with our team for a Customized Cost-Saving Analysis tailored to your specific production requirements. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to our optimized intermediates. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or secure a long-term supply agreement, NINGBO INNO PHARMCHEM is ready to support your strategic goals with reliability and scientific excellence.
