Scalable Production of 2-Amino-5-Chloropyridine via Ionic Liquid Catalysis for Global Pharma Supply Chains
Introduction to Advanced Synthetic Methodology
The pharmaceutical industry continuously seeks robust synthetic routes for critical heterocyclic intermediates, particularly those serving as building blocks for central nervous system agents like zopiclone. Patent CN106632014B introduces a transformative preparation method for 2-amino-5-chloropyridine that addresses longstanding challenges in selectivity and environmental impact. This innovation utilizes N-fluoro-N-chlorobenzenesulfonamide as a precise chlorinating agent in conjunction with imidazole-based ionic liquids, achieving exceptional yields under remarkably mild conditions. By shifting away from hazardous gaseous chlorine and corrosive mineral acids, this technology offers a sustainable pathway for manufacturing high-purity pharmaceutical intermediates. The process operates efficiently at temperatures ranging from 0°C to 40°C, demonstrating significant potential for energy savings and safer operational protocols in large-scale facilities. Furthermore, the ability to recycle the catalytic system multiple times underscores its economic viability for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-chloropyridine has been plagued by severe operational hazards and poor selectivity profiles that hinder efficient manufacturing. Traditional routes often rely on the salification of 2-aminopyridine in concentrated sulfuric acid followed by chlorination, a process that generates substantial amounts of acidic waste and causes rapid equipment corrosion due to the aggressive nature of the reagents. Moreover, controlling the stoichiometry of chlorine gas in these reactions is notoriously difficult, frequently leading to the formation of unwanted dichlorinated byproducts such as 2-amino-3,5-dichloropyridine, which complicates downstream purification and reduces overall yield to approximately 50 percent. Alternative methods utilizing potassium chlorate in hydrochloric acid improve yields slightly but still struggle with byproduct formation and require complex separation procedures to isolate the desired mono-chlorinated species. These legacy processes impose heavy burdens on waste treatment infrastructure and pose significant safety risks to personnel handling toxic gases and strong acids.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages the unique properties of ionic liquids to achieve unprecedented control over the chlorination reaction. By employing N-fluoro-N-chlorobenzenesulfonamide as a stable, solid chlorinating source, the process eliminates the need for handling dangerous chlorine gas while ensuring precise delivery of the electrophile. The reaction proceeds smoothly in common organic solvents like dichloromethane or acetonitrile, facilitated by a catalytic amount of imidazole ionic liquid which directs the substitution exclusively to the 5-position. 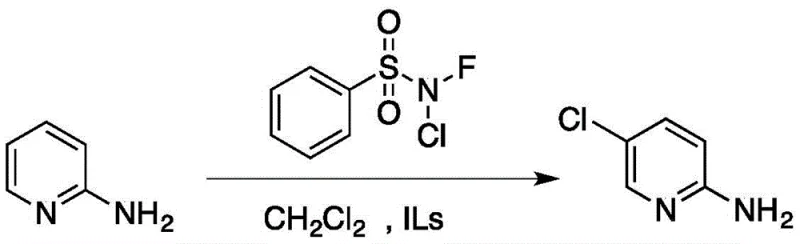 This novel approach not only boosts yields to nearly 99 percent but also simplifies the workup procedure, as the product can be easily isolated via recrystallization. The mild reaction conditions and the absence of corrosive byproducts make this route highly attractive for modern green chemistry initiatives and cost reduction in pharmaceutical intermediate manufacturing.
This novel approach not only boosts yields to nearly 99 percent but also simplifies the workup procedure, as the product can be easily isolated via recrystallization. The mild reaction conditions and the absence of corrosive byproducts make this route highly attractive for modern green chemistry initiatives and cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ionic Liquid Catalyzed Chlorination
The success of this synthetic route hinges on the sophisticated interaction between the imidazole ionic liquid catalyst and the substrate, which fundamentally alters the electronic landscape of the pyridine ring. The ionic liquid, typically comprising a cation such as 1-methyl-3-methylimidazolium and a halide anion, acts as more than just a solvent or phase transfer agent; it actively participates in the reaction mechanism through coordination. 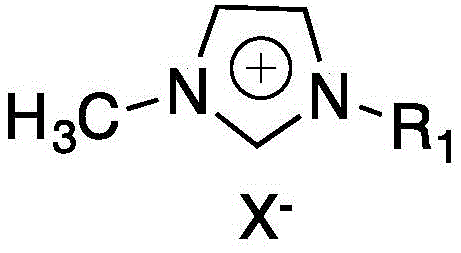 The nitrogen atom of the amino group on the 2-aminopyridine forms a complex with the ionic liquid species, creating a steric and electronic shield around the adjacent C-3 position. This complexation effectively deactivates the C-3 site towards electrophilic attack, thereby preventing the formation of the thermodynamically favorable but undesirable 3,5-dichloro byproduct. Consequently, the chlorinating agent is directed exclusively to the C-5 position, resulting in high regioselectivity. This mechanistic understanding allows chemists to fine-tune the catalyst structure, adjusting the alkyl chain length on the imidazole ring to optimize solubility and catalytic efficiency for specific batch sizes.
The nitrogen atom of the amino group on the 2-aminopyridine forms a complex with the ionic liquid species, creating a steric and electronic shield around the adjacent C-3 position. This complexation effectively deactivates the C-3 site towards electrophilic attack, thereby preventing the formation of the thermodynamically favorable but undesirable 3,5-dichloro byproduct. Consequently, the chlorinating agent is directed exclusively to the C-5 position, resulting in high regioselectivity. This mechanistic understanding allows chemists to fine-tune the catalyst structure, adjusting the alkyl chain length on the imidazole ring to optimize solubility and catalytic efficiency for specific batch sizes.
Furthermore, the stability of the ionic liquid under reaction conditions ensures that the catalytic cycle remains intact throughout the process, allowing for consistent performance over multiple runs. The absence of transition metals in this catalytic system is another critical advantage, as it removes the risk of heavy metal contamination in the final active pharmaceutical ingredient. This is particularly vital for regulatory compliance, where strict limits on residual metals must be met. The mechanism also explains why the reaction can proceed at ambient temperatures without the need for harsh Lewis acids; the ionic liquid provides sufficient activation energy through its unique solvation environment. Understanding these mechanistic nuances is essential for R&D teams aiming to adapt this chemistry for continuous flow processing or other advanced manufacturing technologies.
How to Synthesize 2-Amino-5-Chloropyridine Efficiently
Implementing this synthesis requires careful attention to the molar ratios of reagents and the selection of the appropriate ionic liquid to maximize yield and purity. The standard protocol involves dissolving 2-aminopyridine and N-fluoro-N-chlorobenzenesulfonamide in a solvent like dichloromethane, followed by the addition of the ionic liquid catalyst. Reaction progress is conveniently monitored via thin-layer chromatography, with completion typically achieved within a few hours at room temperature.
- Dissolve 2-aminopyridine and N-fluoro-N-chlorobenzenesulfonamide in an organic solvent such as dichloromethane.
- Add a catalytic amount of imidazole ionic liquid (e.g., 1-methyl-3-methylimidazolium chloride) and stir at 0-40°C.
- Monitor reaction by TLC, remove solvent, recrystallize residue with ethanol, and recover the ionic liquid catalyst from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-catalyzed process represents a strategic opportunity to optimize costs and secure a more resilient supply of critical intermediates. The elimination of corrosive acids and toxic gases significantly reduces the capital expenditure required for specialized corrosion-resistant reactors and scrubbing systems, leading to substantial cost savings in facility maintenance and operation. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which streamlines the purification process and reduces the consumption of solvents and energy associated with extensive chromatographic separations. The ability to recycle the ionic liquid catalyst further enhances the economic profile of the process, as the catalyst cost is amortized over numerous production batches rather than being a single-use expense.
- Cost Reduction in Manufacturing: The process achieves significant cost efficiency by replacing expensive and hazardous chlorinating agents with stable solids and recyclable catalysts. By avoiding the use of concentrated sulfuric acid and chlorine gas, manufacturers can drastically lower their expenditure on waste disposal and environmental compliance measures. The high yield of nearly 99 percent means that raw material utilization is maximized, reducing the cost per kilogram of the final product. Furthermore, the simplified workup procedure involving simple recrystallization instead of complex distillation or column chromatography lowers labor and utility costs significantly.
- Enhanced Supply Chain Reliability: Utilizing stable solid reagents like N-fluoro-N-chlorobenzenesulfonamide mitigates the supply risks associated with gaseous chlorine, which is subject to strict transportation regulations and availability fluctuations. The mild reaction conditions allow for production in a wider range of facilities without requiring specialized high-pressure or high-temperature equipment, thereby diversifying the potential manufacturing base. The robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining uninterrupted supply lines to downstream API manufacturers.
- Scalability and Environmental Compliance: The green chemistry attributes of this method align perfectly with increasingly stringent global environmental regulations, facilitating easier permitting and long-term operational sustainability. The absence of heavy metal catalysts simplifies the regulatory filing process for drug master files, as there is no need for extensive metal clearance validation. The process generates minimal hazardous waste, primarily consisting of benign sulfonamide byproducts that are easier to treat than acidic sludge. This environmental friendliness enhances the corporate social responsibility profile of the manufacturer and future-proofs the supply chain against tightening ecological laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: How does the ionic liquid catalyst prevent dichlorination byproducts?
A: The imidazole ionic liquid forms a complex with the amino group on the pyridine ring. This coordination effectively blocks the C-3 position sterically and electronically, preventing the chlorinating agent from attacking that site and ensuring high selectivity for the 5-chloro product.
Q: Can the ionic liquid catalyst be recycled for industrial production?
A: Yes, the patent data demonstrates that the imidazole ionic liquid can be recovered from the filtrate after solvent removal and reused for more than 10 cycles without significant loss in catalytic activity or product yield.
Q: What are the safety advantages of this method over traditional chlorination?
A: Unlike traditional methods that utilize toxic chlorine gas or corrosive concentrated sulfuric acid, this process employs solid N-fluoro-N-chlorobenzenesulfonamide under mild temperatures (0-40°C), significantly reducing equipment corrosion risks and hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Chloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that our 2-amino-5-chloropyridine meets the highest standards required for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle the specific solvent and catalyst recovery systems needed for this advanced ionic liquid technology, maximizing efficiency and sustainability.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value for your organization. Partner with us to secure a reliable, cost-effective, and high-quality supply of this essential pharmaceutical intermediate.
