Advanced Manufacturing of 2,4,5-Trifluorophenylacetic Acid for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, particularly for high-value antidiabetic medications like Sitagliptin phosphate. Patent CN112851493A discloses a groundbreaking preparation method for 2,4,5-trifluorophenylacetic acid, a pivotal building block in the synthesis of DPP-4 inhibitors. This technical insight report analyzes the novel multi-step pathway that replaces expensive starting materials with cost-effective dichloronitrotoluene isomers, leveraging nucleophilic fluorination and catalytic hydrogenation to achieve superior yields. For R&D directors and procurement specialists, understanding this process is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity compounds at scale. The methodology outlined in the patent addresses long-standing challenges in fluorine chemistry, offering a streamlined approach that enhances both economic efficiency and supply chain stability for global manufacturing networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2,4,5-trifluorophenylacetic acid have historically relied on 2,4,5-trifluoronitrobenzene or 1,2,4-trifluorobenzene as primary starting materials, which present significant economic and logistical barriers. The market price of 2,4,5-trifluoronitrobenzene is notoriously high, driving up the overall production cost and making the final API intermediate less competitive in price-sensitive markets. Furthermore, alternative pathways involving the condensation of trifluoronitrobenzene with diethyl malonate often suffer from low yields in the initial steps, frequently failing to exceed 66% efficiency. These inefficiencies necessitate the consumption of excessive raw materials to achieve target output volumes, thereby increasing waste generation and complicating downstream purification processes. For supply chain heads, these limitations translate into volatile pricing and potential bottlenecks, as the availability of high-cost fluorinated precursors can be inconsistent. The reliance on such expensive feedstocks restricts the ability of manufacturers to offer cost reduction in pharmaceutical intermediates manufacturing, ultimately impacting the affordability of the final therapeutic agents.
The Novel Approach
The innovative strategy detailed in the patent circumvents these economic hurdles by utilizing readily available and inexpensive dichloronitrotoluene isomers, such as 3,4-dichloro-6-nitrotoluene, as the foundational raw materials. This shift in starting material selection drastically lowers the entry cost for the synthesis, enabling a more competitive pricing structure for the final 2,4,5-trifluorophenylacetic acid. The process employs a sequential fluorination technique using potassium fluoride in sulfolane, which allows for precise control over the substitution pattern, ensuring high selectivity and minimizing the formation of unwanted isomers. By optimizing reaction temperatures between 150°C and 230°C, the method achieves complete conversion of the chloro groups to fluoro groups without degrading the nitro functionality. This approach not only improves the overall yield of the first intermediate but also simplifies the purification workflow, as the reaction proceeds cleanly with fewer byproducts. For procurement managers, this novel approach represents a tangible opportunity for substantial cost savings, as the raw material basket is composed of commodity chemicals rather than specialized fluorinated compounds.
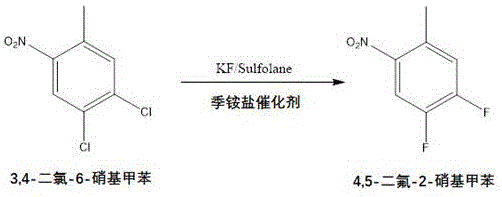
Mechanistic Insights into KF-Catalyzed Nucleophilic Fluorination
The core of this synthetic breakthrough lies in the efficient nucleophilic aromatic substitution facilitated by potassium fluoride in a polar aprotic solvent system. The reaction mechanism involves the activation of the fluorine anion by the quaternary ammonium salt catalyst, which enhances its nucleophilicity towards the electron-deficient aromatic ring of the dichloronitrotoluene. Operating at elevated temperatures, typically around 180°C to 230°C, ensures that the activation energy barrier for the displacement of the chlorine atoms is overcome, leading to the formation of the difluoronitrotoluene intermediate with high fidelity. The use of sulfolane as a solvent is critical, as it stabilizes the transition state and solubilizes the inorganic fluoride salt, allowing for homogeneous reaction conditions that promote uniform heat transfer and mixing. This mechanistic precision is vital for R&D directors focused on purity and impurity profiles, as it prevents the formation of partially fluorinated species that could complicate subsequent steps. The ability to control the stoichiometry of potassium fluoride, often using a molar ratio of 1:1.5 to 1:3.5 relative to the substrate, further ensures that the reaction drives to completion without excessive waste of the fluorinating agent.
Following the initial fluorination, the process employs a catalytic hydrogenation step to reduce the nitro group to an amine, which is a prerequisite for the subsequent Balz-Schiemann reaction. This reduction is typically carried out using Raney nickel or palladium on carbon catalysts under hydrogen pressure ranging from 0.1 MPa to 1.0 MPa at moderate temperatures of 50°C to 75°C. The choice of catalyst and conditions is optimized to prevent over-reduction or hydrogenolysis of the newly introduced fluorine atoms, preserving the integrity of the fluorinated aromatic core. The resulting aminotoluene intermediate is then subjected to diazotization using fluoroboric acid and sodium nitrite at low temperatures, typically between -40°C and 0°C, to form the stable diazonium tetrafluoroborate salt. This low-temperature control is essential for safety and yield, as it prevents the premature decomposition of the diazonium species. The final thermal decomposition of this salt releases nitrogen and boron trifluoride, installing the third fluorine atom at the desired position to yield 2,4,5-trifluorotoluene with purity exceeding 98%.
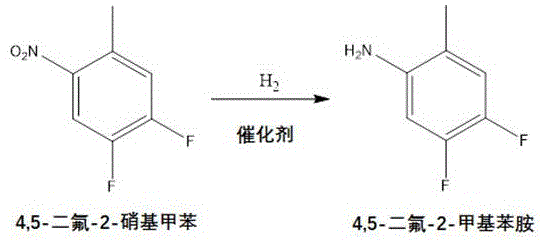
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
Implementing this synthesis route requires careful attention to the sequential transformation of the aromatic core, starting from the cheap dichloronitrotoluene precursors and ending with the hydrolysis of the nitrile side chain. The process is designed to be scalable, with each step building upon the high purity of the previous intermediate to minimize the need for extensive chromatographic purification. Operators must strictly adhere to the temperature profiles specified for the fluorination and diazotization steps to ensure safety and maximize yield, particularly during the exothermic addition of sodium nitrite. The detailed standardized synthesis steps see the guide below, which outlines the specific reagent ratios and workup procedures necessary to replicate the high yields reported in the patent data. This structured approach allows manufacturing teams to transition from laboratory scale to commercial production with confidence, knowing that the critical process parameters have been validated for robustness and reproducibility.
- Perform nucleophilic fluorination on dichloronitrotoluene using potassium fluoride and sulfolane to generate difluoronitrotoluene intermediates.
- Execute catalytic hydrogenation to reduce the nitro group to an amine, followed by diazotization and thermal decomposition to introduce the third fluorine atom.
- Complete the synthesis via side-chain halogenation, cyanation, and acidic hydrolysis to yield the final 2,4,5-trifluorophenylacetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing route offers profound advantages for procurement and supply chain teams seeking to optimize their sourcing strategies for complex pharmaceutical intermediates. The primary benefit stems from the substitution of high-cost fluorinated starting materials with inexpensive chlorinated analogs, which are produced in vast quantities globally and are subject to less price volatility. This fundamental shift in raw material sourcing translates directly into significant cost reduction in pharmaceutical intermediates manufacturing, allowing buyers to negotiate more favorable terms and secure long-term supply agreements without the risk of sudden price spikes. Furthermore, the high yields achieved at each step of the synthesis mean that less raw material is required to produce a kilogram of the final product, enhancing overall material efficiency and reducing the environmental footprint associated with waste disposal. For supply chain heads, the use of common solvents like sulfolane and methanol, along with standard catalysts like Raney nickel, ensures that the supply of consumables is reliable and not dependent on niche specialty chemical vendors.
- Cost Reduction in Manufacturing: The elimination of expensive trifluoronitrobenzene from the bill of materials results in a drastic simplification of the cost structure, enabling manufacturers to pass savings on to clients while maintaining healthy margins. By avoiding the need for complex purification of early-stage intermediates, the process reduces solvent consumption and energy usage associated with distillation and recrystallization, further driving down operational expenditures. The ability to recover and reuse byproducts, such as boron trifluoride generated during the pyrolysis step, adds an additional layer of economic efficiency, turning potential waste streams into valuable resources for other chemical processes. This holistic approach to cost management ensures that the final 2,4,5-trifluorophenylacetic acid is priced competitively in the global market, making it an attractive option for generic drug manufacturers.
- Enhanced Supply Chain Reliability: Sourcing dichloronitrotoluene isomers is significantly more straightforward than procuring specialized fluorinated aromatics, as these chemicals are produced by a wide network of industrial suppliers. This diversity in the supply base mitigates the risk of single-source dependency, ensuring that production schedules are not disrupted by vendor-specific issues or logistical bottlenecks. The robustness of the synthetic route, characterized by high yields and stable intermediates, means that production campaigns can be run continuously with minimal downtime for equipment cleaning or process adjustments. For procurement managers, this reliability translates into reducing lead time for high-purity pharmaceutical intermediates, allowing for leaner inventory management and faster response times to market demand fluctuations. The consistent quality of the output also reduces the need for extensive incoming quality control testing, streamlining the intake process.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing reaction conditions and equipment that are standard in modern chemical plants. The absence of exotic reagents or extreme pressure requirements simplifies the engineering challenges associated with scaling from pilot to production scale, reducing the time and capital investment needed to bring new capacity online. Additionally, the high atom economy of the fluorination steps and the ability to treat byproducts like boron trifluoride safely align with stringent environmental regulations, minimizing the regulatory burden on the manufacturing site. This compliance advantage is crucial for multinational corporations that must adhere to global sustainability standards, ensuring that the supply chain remains resilient against evolving environmental legislation. The reduced generation of hazardous waste also lowers disposal costs, contributing to the overall economic viability of the project.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,4,5-trifluorophenylacetic acid, based on the detailed specifications and beneficial effects outlined in the patent literature. These answers are designed to provide clarity on the feasibility, purity, and scalability of the described method, helping stakeholders make informed decisions about integrating this intermediate into their supply chains. Understanding the nuances of the fluorination mechanism and the subsequent functional group transformations is key to appreciating the value proposition of this technology. The responses below reflect a deep analysis of the process parameters and their implications for industrial application.
Q: What are the primary cost advantages of this synthesis route?
A: The process utilizes low-cost dichloronitrotoluene isomers as starting materials instead of expensive trifluoronitrobenzene, significantly reducing raw material expenditure while maintaining high yields.
Q: How does this method ensure high purity for pharmaceutical use?
A: The stepwise fluorination and hydrogenation strategy minimizes byproduct formation, achieving intermediate purities exceeding 98% and final product purity suitable for DPP-4 inhibitor synthesis.
Q: Is this process scalable for industrial production?
A: Yes, the method avoids complex purification steps for key intermediates like 2,4,5-trifluorotoluene and uses common solvents like sulfolane, facilitating large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the technical potential of this patent is fully realized in a commercial setting. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2,4,5-trifluorophenylacetic acid meets the exacting standards required for API synthesis. We understand the critical nature of supply continuity for pharmaceutical clients and have optimized our operations to deliver consistent quality and volume, mitigating the risks associated with complex fluorine chemistry. Our team of experts is ready to collaborate with your R&D department to validate the route and ensure seamless technology transfer, leveraging our infrastructure to support your long-term manufacturing goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this method for your supply chain. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to evaluate the compatibility of our production capabilities with your quality systems. Our commitment to transparency and technical excellence ensures that you receive not just a chemical product, but a comprehensive solution that enhances your competitive position in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
