Advanced Synthesis of 2,4,5-Trifluorophenylacetic Acid for Commercial Scale-Up and Procurement Optimization
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates, particularly those serving blockbuster drugs like Sitagliptin. Patent CN101244994A introduces a transformative methodology for the preparation of 2,4,5-trifluorophenylacetic acid, a pivotal building block in the synthesis of DPP-IV inhibitors. This innovative approach leverages a three-step sequence comprising Friedel-Crafts acetylation, a Willgerodt-Kindler reaction, and final hydrolysis to achieve exceptional purity levels exceeding 99.9%. By shifting away from hazardous reagents and complex catalytic systems, this technology addresses the growing demand for safer, more efficient, and cost-effective manufacturing processes within the global supply chain. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for optimizing long-term sourcing strategies and ensuring regulatory compliance in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
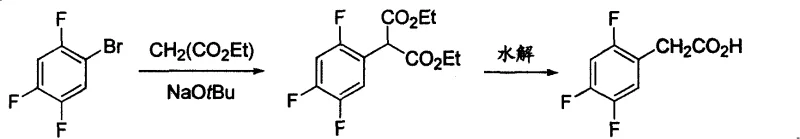
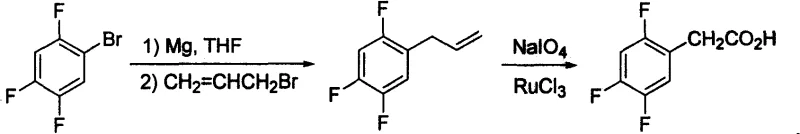
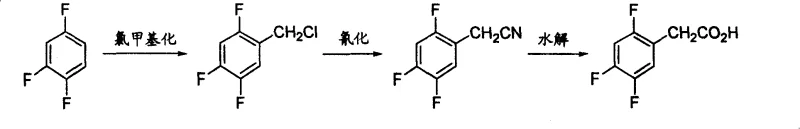
Historically, the synthesis of 2,4,5-trifluorophenylacetic acid has relied on methodologies that present significant industrial challenges. For instance, earlier patents such as US20040068141 describe a route involving the substitution of 2,4,5-trifluorobromobenzene with diethyl malonate under alkaline conditions. This pathway often demands rigorous reaction parameters that are difficult to maintain consistently at scale, leading to potential yield fluctuations and increased operational complexity. Furthermore, alternative routes like those disclosed in US20040077901 utilize Grignard reagents followed by oxidation with Ruthenium trichloride and sodium periodate. The reliance on expensive noble metal catalysts and strong oxidants not only inflates raw material costs but also introduces heavy metal contamination risks that require extensive downstream purification. Additionally, methods involving cyanation, as seen in CN1749232, pose severe safety hazards due to the use of sodium cyanide, creating substantial environmental and occupational health liabilities for manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN101244994A utilizes 1,2,4-trifluorobenzene as a readily available starting material, undergoing a streamlined transformation that prioritizes safety and efficiency. The process initiates with a Friedel-Crafts acetylation to form the ketone intermediate, followed by a Willgerodt-Kindler reaction which elegantly converts the ketone into a thioamide without the need for toxic cyanide sources. This strategic shift eliminates the handling of highly hazardous substances, thereby reducing the regulatory burden and safety infrastructure costs associated with production. The final hydrolysis step is conducted under mild alkaline or acidic conditions, ensuring high conversion rates while minimizing the formation of difficult-to-remove byproducts. This cohesive strategy results in a synthesis pathway that is not only chemically superior but also economically advantageous for commercial-scale operations seeking to reduce lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Friedel-Crafts Acetylation and Willgerodt-Kindler Reaction
The core of this synthetic innovation lies in the precise execution of the Friedel-Crafts acetylation, where 1,2,4-trifluorobenzene reacts with an acetylating agent in the presence of a Lewis acid catalyst such as aluminum trichloride. This electrophilic aromatic substitution is highly regioselective, favoring the formation of 2,4,5-trifluoroacetophenone due to the directing effects of the fluorine substituents on the aromatic ring. The choice of aluminum trichloride is critical, as it provides the necessary activation energy for the acylation while remaining cost-effective compared to other Lewis acids. Following this, the Willgerodt-Kindler reaction facilitates the migration of the carbonyl group and the introduction of the nitrogen and sulfur atoms required for the subsequent chain extension. This transformation occurs through the formation of a morpholine or amine-based intermediate which reacts with elemental sulfur, creating a thioamide structure that serves as a stable precursor for the final acid. The mechanistic elegance of this sequence ensures that the carbon skeleton is preserved with high fidelity, minimizing the generation of structural isomers that could complicate downstream purification efforts.
Impurity control is a paramount concern for R&D directors overseeing the production of API intermediates, and this pathway offers distinct advantages in managing the impurity profile. The mild conditions employed during the hydrolysis of the thioamide intermediate prevent the degradation of the sensitive trifluorophenyl moiety, which can be susceptible to defluorination under harsh acidic or basic environments. By optimizing the temperature and pH during the hydrolysis step, the process effectively suppresses the formation of side products such as dehalogenated analogs or over-oxidized species. Furthermore, the use of recrystallization from ethanol in the final isolation step provides an additional layer of purification, ensuring that the final 2,4,5-trifluorophenylacetic acid meets the stringent purity specifications required for pharmaceutical applications. This robust control over the impurity spectrum translates directly into higher quality raw materials for drug substance manufacturing, reducing the risk of batch failures and ensuring consistent product performance.
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The process begins with the acetylation step, where temperature control is vital to prevent exothermic runaway, followed by the thioamide formation which requires sustained heating in a polar aprotic solvent. The final hydrolysis step demands precise pH adjustment to ensure complete conversion while facilitating easy product isolation. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Perform Friedel-Crafts acetylation of 1,2,4-trifluorobenzene using aluminum trichloride and acetyl chloride to form 2,4,5-trifluoroacetophenone.
- Conduct Willgerodt-Kindler reaction with sulfur and dimethylamine hydrochloride in DMF to generate the thioamide intermediate.
- Execute alkaline hydrolysis of the thioamide followed by acidification and recrystallization to isolate the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere chemical efficiency. By eliminating the need for expensive noble metal catalysts and toxic cyanide reagents, the overall cost of goods sold is significantly reduced, allowing for more competitive pricing structures in the global market. The simplified operational workflow reduces the complexity of waste treatment and environmental compliance, leading to lower overhead costs and a smaller carbon footprint for the manufacturing facility. Additionally, the use of readily available starting materials like 1,2,4-trifluorobenzene ensures a stable supply chain that is less susceptible to geopolitical disruptions or raw material shortages. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of multinational pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of costly Ruthenium catalysts and hazardous cyanide reagents drastically lowers raw material expenses and waste disposal costs. By utilizing common Lewis acids and simple amine salts, the process achieves significant cost savings without compromising on reaction efficiency or product quality. This economic advantage allows suppliers to offer more competitive pricing models while maintaining healthy profit margins in a volatile market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 1,2,4-trifluorobenzene and acetyl chloride ensures that raw material sourcing is robust and resilient against supply shocks. Unlike specialized reagents that may have limited suppliers, these inputs are widely available from multiple global vendors, reducing the risk of production delays. This stability translates into consistent lead times and improved on-time delivery performance for downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic substances make this process inherently safer and easier to scale from pilot plant to commercial production. The reduced environmental hazard profile simplifies regulatory approvals and permits, accelerating the time to market for new projects. Furthermore, the simplified waste stream facilitates easier treatment and disposal, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,4,5-trifluorophenylacetic acid. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on the feasibility and advantages of this synthesis route for potential partners.
Q: Why is the Willgerodt-Kindler route preferred over cyanation for this intermediate?
A: The Willgerodt-Kindler route eliminates the use of highly toxic sodium cyanide required in older methods, significantly improving operational safety and reducing environmental compliance burdens for industrial manufacturing.
Q: What purity levels can be achieved with this synthesis method?
A: The patented process specifies that the final product content can exceed 99.9% after recrystallization, meeting the stringent quality requirements for API intermediate production.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes mild reaction conditions and avoids expensive noble metal catalysts like Ruthenium, making it economically viable and technically feasible for ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to full-scale manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2,4,5-trifluorophenylacetic acid meets the highest industry standards. Our expertise in process optimization allows us to adapt this patented methodology to your specific volume requirements while maximizing efficiency and safety.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us help you secure a reliable supply of this critical intermediate for your Sitagliptin production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
