Advanced Manufacturing of 2,4,5-Trifluorophenylacetic Acid for Global Diabetes Drug Supply Chains
The pharmaceutical industry's relentless pursuit of efficient, cost-effective, and environmentally sustainable manufacturing processes has led to significant innovations in the synthesis of key drug intermediates. A pivotal development in this domain is detailed in patent CN102690166A, which outlines a novel preparation method for 2,4,5-trifluorobenzyl chloride and its subsequent conversion into 2,4,5-trifluorophenylacetic acid. This specific carboxylic acid derivative serves as a critical building block in the synthesis of Sitagliptin, a widely prescribed medication for the treatment of type 2 diabetes. The traditional methods for producing this intermediate have long been plagued by issues related to hazardous reagents, excessive waste generation, and operational instability. The disclosed technology addresses these challenges by introducing a refined chloromethylation strategy that utilizes a saturated hydrogen chloride sulfuric acid solution. This approach not only enhances the reaction yield to over 90% but also fundamentally alters the waste profile of the manufacturing process. For global supply chain leaders and R&D directors, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality and sustainability standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,4,5-trifluorobenzyl chloride has relied heavily on chlorinating agents such as chlorosulfonic acid or mixtures containing zinc chloride and hydrochloric acid. While chemically effective, these traditional reagents present substantial operational and environmental drawbacks that hinder modern manufacturing efficiency. Chlorosulfonic acid, for instance, is known for its extremely pungent and irritating odor, posing significant safety risks to personnel and requiring specialized containment infrastructure. Furthermore, its chemical instability can lead to unpredictable reaction outcomes, complicating process control. Alternatively, methods employing zinc chloride or sodium chloride generate massive quantities of inorganic salt byproducts. This accumulation of inorganic waste creates a severe burden on wastewater treatment facilities and drastically increases the cost of waste disposal. The presence of heavy metals like zinc also necessitates additional purification steps to ensure the final product meets the rigorous purity specifications required for active pharmaceutical ingredient (API) synthesis. These factors collectively contribute to higher production costs and a larger environmental footprint, making conventional routes less attractive for long-term commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to legacy methods, the innovative process described in the patent leverages a chlorinating agent composed of hydrogen chloride gas dissolved in concentrated sulfuric acid (80-95% concentration). This strategic substitution eliminates the need for chlorosulfonic acid and metal salts, thereby streamlining the reaction workflow and mitigating safety hazards. The use of gaseous hydrogen chloride allows for precise stoichiometric control, while the concentrated sulfuric acid acts as both a solvent and a dehydrating agent, driving the equilibrium towards the desired product. Crucially, this system facilitates the separation of the organic product layer from the acid layer, enabling the potential recycling of the sulfuric acid after concentration adjustment. This closed-loop capability significantly reduces the discharge of waste liquor, aligning with green chemistry principles. The result is a cleaner, more robust synthetic pathway that delivers high-purity 2,4,5-trifluorobenzyl chloride, which can then be seamlessly converted into the target acid through cyanation and hydrolysis. This holistic improvement in process design offers a compelling value proposition for cost reduction in API manufacturing.
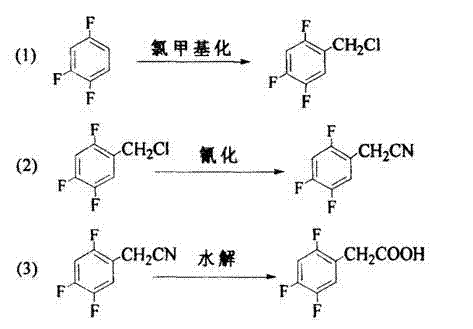
Mechanistic Insights into HCl-H2SO4 Catalyzed Chloromethylation
The core of this technological advancement lies in the mechanistic efficiency of the chloromethylation reaction performed under acidic conditions. In this system, paraformaldehyde depolymerizes in the presence of strong acid to generate formaldehyde, which is subsequently protonated to form a highly electrophilic hydroxymethyl cation species. The electron-rich aromatic ring of 1,2,4-trifluorobenzene, activated by the fluorine substituents, undergoes electrophilic aromatic substitution at the position ortho to the existing fluorine atoms, guided by steric and electronic factors. The introduction of hydrogen chloride gas into the sulfuric acid matrix ensures a high local concentration of chloride ions, which rapidly intercept the intermediate benzylic alcohol to form the stable benzyl chloride bond. The specific concentration range of 80-95% sulfuric acid is critical; concentrations below this range may lack sufficient dehydrating power, while higher concentrations could lead to sulfonation side reactions or excessive viscosity. By maintaining the reaction temperature between 0-30°C, the process minimizes thermal degradation and poly-chlorination, ensuring that the mono-chloromethylated product is formed with exceptional selectivity. This precise control over reaction parameters is what enables the reported yields to consistently exceed 90%, providing a reliable foundation for downstream processing.
Impurity control is another vital aspect where this mechanism excels, particularly concerning the profile of the final 2,4,5-trifluorophenylacetic acid. In traditional zinc chloride-mediated reactions, trace metal contamination is a persistent issue that requires expensive chelating agents or recrystallization steps to remove. The metal-free nature of the HCl-H2SO4 system inherently prevents the introduction of heavy metal impurities, simplifying the purification train. Furthermore, the phase separation between the organic product and the aqueous acid layer is distinct and rapid, reducing the likelihood of emulsion formation which often traps impurities. During the subsequent cyanation and hydrolysis steps, the high purity of the starting benzyl chloride translates directly into a cleaner nitrile intermediate and最终 acid product. The hydrolysis step, whether conducted under acidic or alkaline conditions, proceeds efficiently because the absence of interfering metal salts prevents catalyst poisoning or side reactions. This mechanistic clarity ensures that the final product meets the stringent purity specifications demanded by regulatory bodies for diabetes medication intermediates.
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
Implementing this synthesis route requires careful attention to reagent preparation and temperature control to maximize the benefits of the patented method. The process begins with the saturation of sulfuric acid with hydrogen chloride gas, a step that must be managed to ensure consistent acid strength and chloride availability. Following the chloromethylation, the crude benzyl chloride is subjected to nucleophilic substitution with a cyanide source, typically sodium cyanide, in a polar organic solvent. The final transformation involves the hydrolysis of the nitrile group, which can be tuned via pH and temperature to optimize crystal formation and recovery. While the general chemistry is straightforward, the specific operational parameters defined in the patent are key to achieving the reported high yields and purity levels. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the procedure.
- Perform chloromethylation of 1,2,4-trifluorobenzene using paraformaldehyde and saturated hydrogen chloride in 80-95% sulfuric acid at 0-30°C.
- Conduct cyanation of the resulting 2,4,5-trifluorobenzyl chloride with sodium cyanide in an organic solvent at elevated temperatures.
- Execute acidic or alkaline hydrolysis of the nitrile intermediate to yield the final 2,4,5-trifluorophenylacetic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the waste management profile. By eliminating the generation of heavy metal sludge and excessive inorganic salts, manufacturers can significantly reduce the costs associated with hazardous waste disposal and environmental compliance. This reduction in regulatory burden allows for more flexible production scheduling and lowers the risk of shutdowns due to environmental violations. Furthermore, the ability to recycle the sulfuric acid layer introduces a circular economy element to the process, decreasing the consumption of raw materials and insulating the supply chain from volatility in acid pricing. These factors combine to create a more resilient and cost-efficient manufacturing operation that is better equipped to handle the demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like chlorosulfonic acid directly lowers the bill of materials for each production batch. Additionally, the removal of zinc chloride means that costly downstream purification steps, such as heavy metal scavenging or extensive washing to remove salts, are no longer necessary. This streamlining of the process reduces labor hours, energy consumption, and solvent usage, leading to substantial cost savings per kilogram of product. The high yield of the chloromethylation step further amplifies these savings by maximizing the throughput of the starting material, ensuring that less raw material is wasted on side products. Consequently, the overall cost of goods sold (COGS) for the intermediate is optimized, providing a competitive edge in price negotiations.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, specifically hydrogen chloride gas and sulfuric acid, are commodity chemicals with robust and stable global supply chains. Unlike specialized catalysts or complex organometallic reagents that may suffer from supply disruptions, these basic industrial chemicals are readily available from multiple sources. This abundance ensures continuity of supply even during periods of market fluctuation. Moreover, the simplified workup procedure reduces the cycle time for each batch, allowing manufacturers to respond more quickly to urgent orders. The robustness of the reaction conditions also means that the process is less prone to failure due to minor variations in raw material quality, further stabilizing the delivery schedule for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the use of standard reactor materials and well-understood unit operations. The absence of corrosive chlorosulfonic acid reduces the requirement for exotic alloy reactors, allowing for the use of glass-lined or standard stainless steel equipment which is more readily available. From an environmental perspective, the significant reduction in liquid and solid waste discharge aligns with increasingly strict global environmental regulations. This compliance minimizes the risk of fines and permits delays, ensuring uninterrupted operation. The greener profile of the manufacturing process also enhances the brand reputation of the supplier, appealing to multinational corporations that prioritize sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,4,5-trifluorophenylacetic acid using this advanced methodology. These insights are derived directly from the patent data and practical manufacturing experience, aiming to clarify the operational benefits and feasibility of the route. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or long-term supply partnerships.
Q: What are the primary environmental advantages of this synthesis route?
A: This method replaces chlorosulfonic acid and zinc chloride with hydrogen chloride gas and sulfuric acid, significantly reducing inorganic salt waste and eliminating the need for complex heavy metal removal processes.
Q: How does the new chloromethylation agent improve product purity?
A: By utilizing a saturated hydrogen chloride sulfuric acid solution, the reaction achieves higher selectivity and yields exceeding 90%, minimizing side reactions and simplifying downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses common industrial reagents and allows for the recycling of the sulfuric acid layer, making it highly scalable and economically viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving drugs like Sitagliptin depends on a secure and high-quality supply of critical intermediates. Our technical team has extensively analyzed the patented routes for 2,4,5-trifluorophenylacetic acid and possesses the expertise to implement this green chemistry approach at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for API synthesis. We are committed to being a partner that not only supplies chemicals but also drives value through process optimization.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain needs. By leveraging our manufacturing capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your project's volume and timeline. We encourage you to request specific COA data and route feasibility assessments to verify the superiority of our production methods. Let us collaborate to secure a sustainable and efficient supply of 2,4,5-trifluorophenylacetic acid for your global operations.
