Revolutionizing Cephalosporin Synthesis: A Stable Active Ester Route for Industrial Scale-Up
Revolutionizing Cephalosporin Synthesis: A Stable Active Ester Route for Industrial Scale-Up
The pharmaceutical landscape for first-generation cephalosporins is undergoing a significant technical shift, driven by the need for more robust and scalable synthetic pathways. Patent CN102267953A introduces a groundbreaking intermediate compound, D-2-(tert-butoxycarbonyl)amino-2-(1,4-cyclohexadiene)acetate, which serves as a pivotal precursor for the synthesis of Cephradine and Cefroxadine. Unlike traditional methods that rely on highly unstable acid chloride derivatives, this innovation leverages a stable active ester strategy that fundamentally alters the risk profile of manufacturing these critical antibiotics. By replacing moisture-sensitive acyl chlorides with a Boc-protected active ester, the process achieves superior control over stereochemistry and impurity profiles. This technical advancement is not merely a laboratory curiosity but a viable industrial solution that addresses long-standing bottlenecks in beta-lactam antibiotic production, offering a pathway to higher yields and consistent quality.
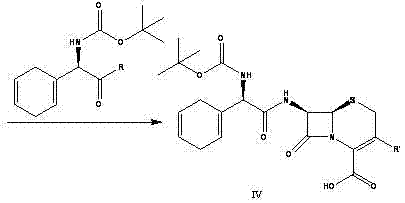
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cephradine and Cefroxadine has been dominated by the use of D-2-amino-2-(1,4-cyclohexadiene)acetyl chloride hydrochloride as the acylating agent. As illustrated in prior art routes such as those disclosed in US5034522, this conventional approach presents severe operational challenges that hinder efficient manufacturing. The acid chloride intermediate is inherently unstable, prone to rapid hydrolysis upon exposure to atmospheric moisture, and requires stringent anhydrous conditions throughout the process. Furthermore, the preparation of this acid chloride often necessitates the use of hazardous chlorinating agents and cryogenic temperatures to prevent decomposition, leading to high energy consumption and complex safety protocols. The instability of the starting material also translates into variable reaction kinetics, making it difficult to maintain consistent batch-to-batch quality and often resulting in lower overall yields due to side reactions.
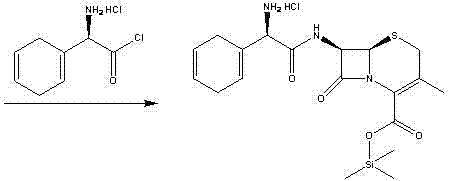
The Novel Approach
In stark contrast, the novel methodology presented in this patent utilizes a Boc-protected active ester intermediate that circumvents the volatility and instability associated with acid chlorides. The new route begins with the protection of the amino group using di-tert-butyl dicarbonate (Boc2O), creating a robust carbamate that shields the amine from unwanted side reactions during subsequent steps. This protected acid is then converted into an active ester, such as an N-hydroxysuccinimide (NHS) or N-hydroxybenzotriazole (HOBt) ester, using mild condensing agents like DCC or HATU. This transformation allows the acylation of the cephalosporin nucleus to proceed under温和 conditions, typically between 0°C and 50°C, without the need for rigorous exclusion of moisture. The result is a streamlined process where the intermediate can be isolated, stored, and transported with stability, decoupling the synthesis of the side chain from the final coupling step and providing unprecedented flexibility in production scheduling.
Mechanistic Insights into Active Ester Coupling and Boc Protection
The core mechanistic advantage of this synthesis lies in the strategic use of the tert-butoxycarbonyl (Boc) protecting group combined with active ester chemistry. In the initial step, the nucleophilic attack of the amine on Boc2O is facilitated by mild bases such as sodium hydroxide or potassium bicarbonate in a biphasic system of water and organic solvents like THF. This protection step is crucial because it prevents the free amine from participating in self-condensation or reacting prematurely with the activated carboxyl group. Following protection, the carboxylic acid is activated by converting it into an active ester. Mechanistically, the condensing agent activates the carbonyl carbon, making it susceptible to nucleophilic attack by the hydroxylamine (NHS or HOBt). The resulting active ester possesses a leaving group that is sufficiently reactive to acylate the 7-amino group of the cephalosporin nucleus but stable enough to prevent significant hydrolysis during the reaction timeframe. This balance of reactivity is key to minimizing the formation of hydrolyzed byproducts and ensuring high conversion rates.
Furthermore, this mechanism offers superior control over the stereochemical integrity of the final product. In acid chloride mediated couplings, the harsh conditions and the presence of base can sometimes lead to epimerization at the chiral alpha-carbon, resulting in diastereomeric impurities that are difficult to remove. The milder conditions of the active ester coupling, performed at near-neutral or slightly basic pH levels using catalysts like triethylamine or DMAP, significantly reduce the risk of racemization. The final deprotection step involves treating the coupled product with acids such as hydrochloric acid or trifluoroacetic acid, which cleanly removes the Boc group to reveal the free amine of the antibiotic. This acidolysis is highly selective and does not affect the sensitive beta-lactam ring, ensuring that the biological activity of the final Cephradine or Cefroxadine is preserved while achieving high pharmaceutical purity standards.
How to Synthesize D-2-(tert-butoxycarbonyl)amino-2-(1,4-cyclohexadiene)acetate Efficiently
The synthesis of this critical intermediate is designed for operational simplicity and high throughput. The process initiates with the dissolution of D-2-amino-2-(1,4-cyclohexadiene)acetic acid in a mixture of water and an organic co-solvent, followed by the addition of a base and Boc anhydride. After the protection is complete, the intermediate is extracted and subsequently activated using standard peptide coupling reagents. This modular approach allows manufacturers to produce the active ester in bulk and store it until needed for the final coupling with 7-ACA or 7-ADCA derivatives. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and temperature profiles optimized for maximum yield, are outlined in the technical guide below.
- Protect D-2-amino-2-(1,4-cyclohexadiene)acetic acid with Boc2O under mild alkaline conditions to form the stable carbamate intermediate.
- Activate the protected acid using condensing agents like DCC or HATU with N-hydroxy succinimide to generate the reactive active ester.
- Couple the active ester with 7-ACA or 7-ADCA derivatives followed by acidic deprotection to yield high-purity Cephradine or Cefroxadine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from acid chloride chemistry to this active ester route represents a significant opportunity for cost optimization and risk mitigation. The elimination of unstable intermediates reduces the need for specialized storage infrastructure and minimizes the loss of raw materials due to degradation during warehousing. Additionally, the ability to perform reactions at ambient temperatures drastically lowers energy costs associated with cryogenic cooling, which is a major expense in traditional beta-lactam synthesis. The simplified workup procedures, which rely on standard filtration and pH-adjusted crystallization rather than complex chromatographic separations, further contribute to reduced processing times and lower utility consumption per kilogram of product.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and hazardous chlorinating reagents required to generate acid chlorides, replacing them with widely available and cost-effective condensing agents. By avoiding the strict anhydrous conditions and low-temperature requirements of the legacy routes, the facility can utilize standard glass-lined or stainless steel reactors without specialized cryogenic jackets, leading to substantial capital expenditure savings and lower operational overheads. The higher purity of the crude product also reduces the burden on downstream purification units, decreasing solvent usage and waste disposal costs significantly.
- Enhanced Supply Chain Reliability: The stability of the Boc-protected active ester intermediate allows for the decoupling of the side-chain synthesis from the final antibiotic assembly. This means that the intermediate can be manufactured in advance during off-peak times and stockpiled without fear of degradation, ensuring a continuous supply of key materials even if there are disruptions in the upstream supply of fresh reagents. This inventory buffer provides a critical safety net for maintaining production schedules and meeting tight delivery deadlines for global pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and more environmentally friendly, utilizing solvents like ethyl acetate and THF which are easier to recover and recycle compared to the halogenated solvents often required for acid chloride handling. The absence of corrosive hydrogen chloride gas evolution during the coupling step improves workplace safety and reduces the load on scrubber systems. These factors make the process highly scalable from pilot batches to multi-ton commercial production, facilitating a smoother technology transfer and faster time-to-market for generic versions of these essential antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this active ester synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable basis for feasibility assessments and technical planning.
Q: Why is the active ester method superior to the acid chloride route for Cephradine?
A: The traditional acid chloride route requires unstable intermediates that are sensitive to moisture and temperature, often necessitating cryogenic conditions. The active ester method described in CN102267953A utilizes a Boc-protected intermediate that is chemically stable, allowing for reactions at ambient temperatures (0-50°C) and significantly simplifying storage and handling logistics.
Q: What represents the key quality advantage of this synthesis route?
A: The use of active esters minimizes side reactions such as hydrolysis and racemization which are common in acid chloride couplings. This results in a cleaner reaction profile, higher optical purity of the final antibiotic, and a simplified purification process involving straightforward pH adjustment and crystallization.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial suitability. It avoids hazardous reagents and extreme conditions, utilizes common organic solvents like THF and Ethyl Acetate, and features robust workup procedures (filtration and extraction) that are easily scalable from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephradine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to remain competitive in the global antibiotic market. Our R&D team has thoroughly evaluated the active ester route described in CN102267953A and confirmed its potential for delivering high-purity intermediates with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every step of the Boc protection and activation process, guaranteeing that our products meet the most stringent purity specifications required by international pharmacopoeias.
We invite you to collaborate with us to optimize your supply chain for Cephradine and Cefroxadine production. By leveraging our expertise in this novel chemistry, your organization can achieve significant operational efficiencies and secure a stable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
