Revolutionizing Cephradine Production: Anaerobic Enzymatic Synthesis for Superior Purity and Scalability
The pharmaceutical industry is constantly seeking robust methodologies to enhance the purity and sustainability of beta-lactam antibiotic production. Patent CN102656274A introduces a groundbreaking approach to synthesizing cephradine, a widely used first-generation cephalosporin, by leveraging enzymatic catalysis under strictly anaerobic conditions. This innovation addresses a persistent challenge in semi-synthetic antibiotic manufacturing: the oxidative degradation of side chains that leads to unacceptable impurity profiles. By shifting the reaction environment from aerobic to anaerobic, the process significantly mitigates the formation of cephalexin, a structurally similar analog that complicates purification and regulatory compliance. For R&D directors and procurement specialists, this technology represents a pivotal shift towards greener chemistry that does not compromise on yield or quality standards. The integration of oxygen scavengers and inert gas purging ensures that the delicate D-dihydrophenylglycine (DHPG) side chain remains intact throughout the activation and coupling phases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cephradine has relied heavily on chemical synthesis routes that utilize hazardous organic solvents such as dichloromethane and dimethylformamide (DMF). These traditional methods involve acylating the amino group of 7-aminodesacetoxycephalosporanic acid (7-ADCA) with a DHPG side chain in a biphasic system. A major drawback of this approach is the generation of substantial chemical waste, posing severe environmental compliance burdens and escalating disposal costs for supply chain managers. Furthermore, chemical synthesis often struggles with stereochemical control, leading to products that require extensive downstream purification to meet pharmacopoeial standards. Even in earlier enzymatic attempts conducted under normal atmospheric conditions, the susceptibility of DHPG to oxidation resulted in the formation of D-phenylglycine (PG). This oxidation byproduct inevitably couples with the beta-lactam nucleus to form cephalexin, frequently pushing impurity levels beyond the acceptable 5.0% threshold mandated by global health authorities.
The Novel Approach
The novel methodology disclosed in the patent fundamentally alters the reaction landscape by enforcing anaerobic conditions during both the activation of the side chain and the enzymatic coupling steps. By reducing oxygen levels by at least 90%, and preferably up to 99%, through the use of nitrogen purging and chemical oxygen scavengers like sodium bisulfite, the process effectively halts the oxidative pathway that generates PG. This strategic modification allows for the use of technical grade DHPG, which may naturally contain trace amounts of oxidized impurities, without compromising the final product quality. The result is a streamlined aqueous process that eliminates the need for chlorinated solvents entirely, aligning with modern green chemistry principles. For a reliable cephradine supplier, adopting this anaerobic protocol translates directly into a more stable supply chain with reduced dependency on volatile organic compounds and simplified waste treatment infrastructure.
Mechanistic Insights into Anaerobic Enzymatic Coupling
The core of this technological advancement lies in the precise control of the reaction atmosphere to preserve the integrity of the DHPG side chain. In standard aerobic environments, DHPG undergoes spontaneous oxidation to form PG, which acts as a competitive substrate for penicillin acylase. When PG couples with 7-ADCA, it yields cephalexin, an impurity that is chemically difficult to separate from cephradine due to their structural similarities. The patent details a mechanism where the concentration of reactive oxygen is minimized using agents such as ascorbic acid, dithiothreitol, or most preferably, bisulfites. This creates a reducing environment that stabilizes the dihydro-structure of the side chain. Additionally, the process utilizes immobilized penicillin G acylase, often mutant variants like Phe-B24-Ala, which exhibit superior Synthesis/Hydrolysis (S/H) ratios. These engineered enzymes favor the formation of the amide bond over hydrolysis, even at elevated temperatures between 15°C and 30°C, thereby driving the equilibrium towards the desired cephradine product.
Controlling the ratio of activated ester to free acid is another critical parameter defined in the mechanistic framework. The efficiency of the coupling reaction is heavily dependent on the purity of the activated DHPG ester prior to its introduction to the enzyme reactor. The patent specifies that the ratio of DHPG ester to the total sum of ester and free acid must be maintained at high levels, ideally exceeding 99%, to maximize conversion rates. This high ratio ensures that the enzyme is presented primarily with the reactive acyl donor rather than the hydrolyzed acid form, which would otherwise drive the reverse reaction. By optimizing this ratio through repeated concentration and alcohol addition cycles under anaerobic conditions, the process achieves conversion rates of 7-ADCA to cephradine exceeding 90%. This level of control minimizes the accumulation of unreacted starting materials, simplifying the subsequent crystallization and isolation steps.
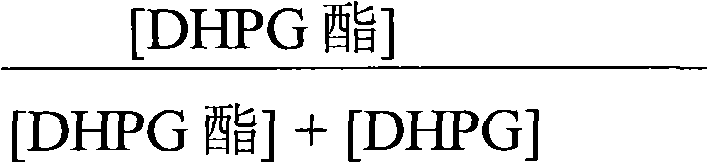
How to Synthesize Cephradine Efficiently
Implementing this synthesis route requires a disciplined approach to process engineering, specifically focusing on the exclusion of oxygen and the optimization of enzyme kinetics. The procedure begins with the activation of DHPG, where the amino acid is converted into its methyl or ethyl ester form using alcohol and a strong acid catalyst under a nitrogen blanket. This step is crucial, as any exposure to air here can seed the reaction with oxidative impurities that propagate through the entire batch. Following activation, the mixture is carefully neutralized and introduced into an aqueous reactor containing 7-ADCA and the immobilized biocatalyst. The reaction is monitored closely for pH and temperature, typically maintained between 6.0 and 8.5 and 20°C to 30°C respectively, to balance enzyme activity with product stability. Detailed standardized synthesis steps see the guide below.
- Convert D-dihydrophenylglycine (DHPG) into its activated ester form (DHPGa) using alcohol and acid under strict anaerobic conditions to prevent oxidation.
- React the activated DHPGa with 7-aminodesacetoxycephalosporanic acid (7-ADCA) in an aqueous mixture using immobilized penicillin acylase, maintaining low oxygen levels.
- Crystallize the resulting cephradine from the aqueous solution at controlled pH and temperature to ensure high stability and low cephalexin impurity levels.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this anaerobic enzymatic process offers compelling economic and operational benefits that extend beyond simple yield improvements. The most significant advantage is the complete elimination of chlorinated organic solvents like dichloromethane, which are subject to increasingly stringent environmental regulations and volatile pricing. By operating in an aqueous medium, manufacturers can drastically reduce the capital expenditure associated with solvent recovery systems and hazardous waste disposal. This shift not only lowers the direct cost of goods sold but also mitigates the risk of production stoppages due to environmental compliance violations. Furthermore, the ability to use technical grade raw materials without extensive pre-purification reduces the upstream procurement burden, allowing for more flexible sourcing strategies in a fluctuating global market.
- Cost Reduction in Manufacturing: The removal of organic solvents from the process workflow results in substantial cost savings by eliminating the need for expensive solvent purchase, storage, and recycling infrastructure. Additionally, the high conversion efficiency achieved through optimized S/H ratio enzymes reduces the consumption of the valuable 7-ADCA core, which is often a cost-driving component in beta-lactam production. The reduction in cephalexin impurity also means less material is lost during recrystallization purification steps, directly improving the overall mass balance and yield of the final API intermediate. These factors combine to create a leaner manufacturing process with a significantly lower overhead per kilogram of produced cephradine.
- Enhanced Supply Chain Reliability: Operating under anaerobic conditions enhances the stability of the reaction intermediates, reducing the likelihood of batch failures caused by unexpected oxidation events. This reliability is critical for maintaining consistent delivery schedules to downstream pharmaceutical clients who operate on tight just-in-time inventory models. The use of immobilized enzymes further supports supply continuity, as these biocatalysts can be reused for multiple cycles, reducing the frequency of enzyme procurement and the associated lead times. By stabilizing the supply of high-purity cephradine, manufacturers can build stronger long-term contracts with generic drug producers who require guaranteed quality specifications.
- Scalability and Environmental Compliance: The aqueous nature of this synthesis makes it inherently easier to scale from pilot plant to commercial production compared to complex multiphase chemical systems. The absence of toxic solvents simplifies the safety profile of the facility, lowering insurance premiums and reducing the regulatory hurdles for capacity expansion. Moreover, the process aligns with global sustainability goals by minimizing the carbon footprint associated with solvent manufacture and incineration. This environmental stewardship is increasingly becoming a prerequisite for inclusion in the supply chains of major multinational pharmaceutical corporations committed to green manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of anaerobic enzymatic synthesis for cephradine. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process optimization projects.
Q: Why are anaerobic conditions critical for cephradine synthesis?
A: Anaerobic conditions prevent the oxidation of D-dihydrophenylglycine (DHPG) into D-phenylglycine (PG). If PG is present, it couples with the beta-lactam core to form cephalexin, a critical impurity that must be kept below pharmacopoeia limits (typically <5%).
Q: How does this enzymatic method compare to traditional chemical synthesis?
A: Traditional chemical synthesis relies on hazardous organic solvents like dichloromethane and DMF, generating significant waste. The enzymatic method operates in aqueous environments, eliminating solvent costs and disposal issues while offering higher stereo-selectivity.
Q: What enzymes are suitable for this process?
A: Penicillin G acylase (PGA), particularly immobilized forms from E. coli, is preferred. Mutant variants with improved Synthesis/Hydrolysis (S/H) ratios, such as the Phe-B24-Ala mutant, allow for higher conversions at elevated temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephradine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the anaerobic enzymatic synthesis of cephradine can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cephradine meets or exceeds international pharmacopoeial standards, including the critical limits on cephalexin impurities. Our commitment to quality assurance ensures that our clients receive a product that is not only cost-effective but also fully compliant with regulatory requirements for API manufacturing.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their cephradine supply chains through these next-generation technologies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us for specific COA data and route feasibility assessments to understand how our anaerobic processing capabilities can enhance your product portfolio. Let us help you navigate the complexities of beta-lactam production with a partner dedicated to scientific excellence and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
