Advanced One-Step Oxidative Amidation for High-Purity Picolinamide Manufacturing
The global demand for high-purity pyridine carboxamides, particularly nicotinamide and isonicotinamide, continues to surge across the pharmaceutical and nutraceutical sectors, driving an urgent need for more sustainable and efficient synthetic methodologies. Patent CN115108976A introduces a transformative approach to this challenge by detailing a novel one-step oxidative amidation process that utilizes pyridinemethanol or pyridinecarboxaldehyde as direct precursors. This technology represents a significant departure from traditional multi-step syntheses, leveraging a specialized Vanadium-Nitrogen-Carbon (V-N-C) heterogeneous catalyst to facilitate the direct conversion of alcohols or aldehydes into amides using molecular oxygen. For R&D directors and process chemists, this patent offers a compelling blueprint for modernizing production lines, promising not only enhanced safety profiles through the avoidance of hazardous intermediates but also superior atom economy. The core innovation lies in the unique electronic structure of the V-N-C material, which activates molecular oxygen under relatively mild thermal conditions ranging from 60°C to 160°C, thereby enabling a streamlined pathway that bypasses the isolation of unstable or toxic intermediates typically associated with older chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of picolinamide derivatives has relied heavily on the reaction of carboxylic acid derivatives, such as acid chlorides or anhydrides, with ammonia or amines, a route fraught with significant logistical and environmental drawbacks. The preparation of acid chlorides often necessitates the use of thionyl chloride or oxalyl chloride, reagents that generate substantial quantities of corrosive hydrogen chloride gas and require rigorous scrubbing systems to manage emissions safely. Furthermore, these acylation reactions are inherently exothermic and frequently demand the addition of stoichiometric amounts of basic scavengers to neutralize the generated acid, leading to the production of large volumes of salt waste that complicate downstream purification and increase disposal costs. Alternative routes involving the hydrolysis of pyridine nitriles, while avoiding acid chlorides, often suffer from harsh reaction conditions requiring strong acids or bases and elevated temperatures, which can degrade sensitive functional groups on the pyridine ring and limit the scope of applicable substrates. Additionally, existing catalytic methods using manganese oxides have demonstrated a propensity for over-oxidation, resulting in the formation of undesirable nitrile by-products that reduce overall yield and necessitate energy-intensive separation processes to achieve pharmaceutical-grade purity.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN115108976A employs a direct oxidative amidation strategy that converts pyridinemethanol or pyridinecarboxaldehyde directly into the target amide in a single liquid-phase operation. This novel approach utilizes molecular oxygen, either as pure oxygen or air, as the terminal oxidant, which is not only cost-effective but also generates water as the primary by-product, drastically reducing the environmental footprint of the manufacturing process. The use of the V-N-C heterogeneous catalyst is pivotal, as it provides active sites that selectively promote the formation of the amide bond while suppressing the dehydration pathway that leads to nitrile formation, a common side reaction in oxidative amidations. By operating at moderate temperatures between 60°C and 160°C and oxygen pressures of 0.1 to 2 MPa, the process ensures safe and controllable reaction kinetics that are highly amenable to standard stainless steel reactor infrastructure found in most fine chemical facilities. The versatility of this method is further evidenced by its compatibility with a wide range of nitrogen sources, including urea, ammonia water, and various ammonium salts, allowing manufacturers to optimize raw material costs based on regional availability without compromising reaction efficiency.
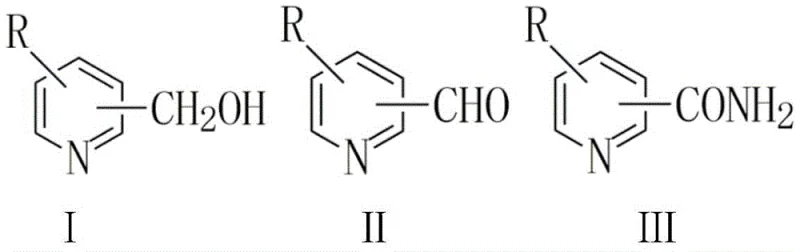
Mechanistic Insights into V-N-C Catalyzed Oxidative Amidation
The efficacy of this synthetic route is fundamentally rooted in the sophisticated design of the V-N-C catalyst, which is synthesized through the high-temperature calcination of inorganic vanadium precursors mixed with organic amines such as melamine or polyaniline. During the calcination process at temperatures ranging from 300°C to 1000°C under an inert atmosphere, the organic amine decomposes to form a nitrogen-doped carbon matrix that effectively anchors vanadium species, preventing their aggregation and leaching during the reaction. This structural integration creates a synergistic effect where the nitrogen dopants modify the electronic environment of the vanadium centers, enhancing their ability to activate molecular oxygen and facilitating the redox cycles necessary for the oxidation of the alcohol or aldehyde substrate. The mechanism likely involves the initial adsorption of the pyridine substrate onto the catalyst surface, followed by the activation of oxygen to form reactive oxygen species that abstract hydrogen atoms from the hydroxymethyl or formyl group, generating an acyl intermediate that subsequently reacts with the nitrogen source to form the amide bond. Crucially, the specific coordination environment provided by the V-N-C lattice appears to stabilize the transition state for amidation over dehydration, thereby explaining the high selectivity observed in the experimental data where nitrile by-products are minimized even under oxidative conditions.
From an impurity control perspective, the heterogeneous nature of the catalyst plays a vital role in maintaining product purity, as the solid catalyst can be physically separated from the reaction mixture via simple filtration once the conversion is complete. This physical separation prevents the catalyst from participating in any subsequent degradation reactions during the work-up phase, a common issue with homogeneous metal catalysts that can contaminate the final API intermediate with trace metals. Furthermore, the mild reaction conditions prevent the thermal decomposition of the pyridine ring or the hydrolysis of the newly formed amide bond, ensuring that the impurity profile remains clean and manageable for downstream crystallization steps. The ability to tune the catalyst composition by varying the ratio of vanadium to nitrogen precursors allows for precise optimization of the active site density, which can be leveraged to maximize turnover numbers and minimize the residence time required for full conversion, ultimately leading to a more robust and reproducible manufacturing process that meets stringent regulatory standards for pharmaceutical intermediates.
How to Synthesize Picolinamide Efficiently
The implementation of this oxidative amidation protocol requires careful attention to the preparation of the V-N-C catalyst and the selection of reaction parameters to ensure optimal performance. The process begins with the mechanical mixing of vanadium sources like ammonium metavanadate or vanadium pentoxide with organic amines, followed by a controlled calcination step to generate the active black powder catalyst. Once the catalyst is prepared, the synthesis involves charging a pressure reactor with the pyridine substrate, the chosen nitrogen source, a compatible solvent such as acetonitrile or toluene, and the catalyst, followed by pressurization with oxygen and heating. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature ramping rates derived from the patent examples, are provided in the guide below to assist technical teams in replicating these results.
- Prepare the V-N-C heterogeneous catalyst by mixing inorganic vanadium compounds with organic amines and calcining at 300-1000°C under inert atmosphere.
- Charge the reactor with pyridinemethanol or pyridinecarboxaldehyde substrate, the V-N-C catalyst, a nitrogen-containing compound (e.g., urea or ammonium salt), and a suitable solvent.
- Pressurize with molecular oxygen (0.1-2 MPa), heat to 60-160°C for up to 12 hours, then filter the catalyst to isolate the high-purity picolinamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this V-N-C catalytic technology presents a strategic opportunity to de-risk the supply of critical pyridine intermediates while simultaneously driving down the total cost of ownership for manufacturing operations. The shift away from acid chloride-based chemistry eliminates the dependency on volatile and often supply-constrained chlorinating agents, replacing them with abundant and inexpensive molecular oxygen or air, which significantly stabilizes the raw material supply chain against market fluctuations. Moreover, the use of readily available pyridinemethanol or pyridinecarboxaldehyde starting materials, which are often produced in large volumes for other applications, ensures a reliable feedstock supply that is less susceptible to the bottlenecks associated with specialized nitrile precursors. The simplified downstream processing, enabled by the ease of catalyst filtration and the absence of salt waste, translates directly into reduced utility consumption and lower waste treatment fees, contributing to substantial long-term operational savings without the need for capital-intensive new equipment installations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling reagents and acid scavengers fundamentally alters the cost structure of picolinamide production by removing expensive consumables from the bill of materials. By utilizing molecular oxygen as the oxidant, the process avoids the purchase of hazardous chemicals like thionyl chloride, leading to direct savings on raw material expenditures while also reducing the costs associated with handling and storing dangerous goods. The heterogeneous nature of the catalyst further enhances economic efficiency by potentially allowing for catalyst recovery and reuse, which lowers the effective catalyst cost per kilogram of product and minimizes the loss of valuable vanadium resources. Additionally, the reduction in waste generation decreases the financial burden of effluent treatment and disposal, creating a leaner and more cost-competitive manufacturing model that improves overall profit margins for high-volume commodity chemicals.
- Enhanced Supply Chain Reliability: Diversifying the synthetic route to include oxidative amidation reduces reliance on single-source suppliers of niche intermediates like pyridine nitriles, which can be subject to production outages or geopolitical trade restrictions. The flexibility to use various ammonium salts or urea as nitrogen sources provides procurement teams with the agility to switch suppliers based on price and availability, ensuring continuous production even if one vendor faces disruptions. Furthermore, the mild reaction conditions and use of common organic solvents mean that the process can be easily transferred between different manufacturing sites globally without requiring specialized infrastructure, thereby enhancing the resilience of the supply network against regional instabilities. This robustness is critical for maintaining consistent delivery schedules to downstream pharmaceutical customers who depend on just-in-time inventory models for their own production lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard liquid-phase oxidation technology that is well-understood in the fine chemical industry, allowing for seamless transition from pilot plant to multi-ton commercial production. The use of molecular oxygen and the generation of water as a by-product align perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal. By adopting this greener methodology, manufacturers can proactively meet sustainability goals and reduce their carbon footprint, which is becoming a key differentiator in supplier selection criteria for major multinational corporations. The safety profile is also markedly improved by avoiding high-pressure hydrogenation or the use of pyrophoric reagents, reducing insurance premiums and operational risks associated with plant safety incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative amidation technology, drawing directly from the experimental data and claims presented in the patent documentation. These insights are intended to clarify the operational feasibility and strategic benefits of adopting the V-N-C catalytic system for the production of pyridine carboxamides. Understanding these nuances is essential for stakeholders evaluating the potential integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of the V-N-C catalytic system over traditional acid chloride methods?
A: The V-N-C catalytic system eliminates the need for hazardous acid chlorides and stoichiometric base scavengers, utilizing molecular oxygen as a green oxidant. This results in significantly reduced waste generation and safer operating conditions compared to conventional acylation routes.
Q: How does this process control the formation of nitrile by-products?
A: Unlike some manganese-based oxide systems that prone to over-oxidation to nitriles, the specific V-N-C material composition and reaction conditions described in the patent effectively suppress nitrile formation, ensuring high selectivity for the desired amide product.
Q: Is the catalyst reusable for industrial scale-up?
A: Yes, the V-N-C material functions as a heterogeneous catalyst, meaning it exists in a different phase than the reaction mixture. This allows for simple filtration and potential recovery, which is critical for reducing operational costs in commercial scale-up scenarios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Picolinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the V-N-C oxidative amidation process in reshaping the landscape of fine chemical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications capable of handling complex heterocyclic syntheses, guaranteeing that every batch of picolinamide or related intermediate meets the highest quality standards required by the global pharmaceutical industry. We are committed to leveraging our technical expertise to optimize these green chemistry routes, delivering products that are not only cost-effective but also environmentally sustainable.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your supply chain context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development and manufacturing programs.
