Advanced Photochemical Route for High-Purity Aminoalcohol Intermediates and Commercial Scale-Up
Introduction to Next-Generation Aminoalcohol Synthesis
The synthesis of functionalized aminoalcohol compounds represents a cornerstone in modern organic chemistry, particularly for the development of bioactive pharmaceutical ingredients and fine chemical intermediates. Patent CN111499524B introduces a groundbreaking methodology that leverages photochemical activation to construct these valuable scaffolds with exceptional efficiency. This technology addresses critical bottlenecks in traditional synthesis by utilizing a unique combination of cyclic diacyl peroxides and halide salts under light irradiation. The core innovation lies in the in situ generation of active oxygen-halogen bonds which undergo homolytic cleavage to drive radical cyclization. 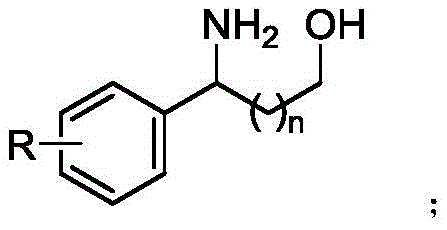 This approach not only streamlines the synthetic route but also aligns perfectly with the principles of green chemistry by minimizing hazardous waste. For R&D directors and procurement specialists seeking a reliable aminoalcohol supplier, understanding this mechanistic shift is crucial for securing a sustainable supply chain for high-purity pharmaceutical intermediates.
This approach not only streamlines the synthetic route but also aligns perfectly with the principles of green chemistry by minimizing hazardous waste. For R&D directors and procurement specialists seeking a reliable aminoalcohol supplier, understanding this mechanistic shift is crucial for securing a sustainable supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aminoalcohol frameworks via halogenated intermediates has relied heavily on hypervalent iodine reagents, as pioneered by researchers like Professor Nagib. While effective, these conventional protocols suffer from significant drawbacks that hinder large-scale manufacturing. The primary limitation is the requirement for stoichiometric or even excess amounts (often 3 equivalents) of inorganic iodine salts, which drastically reduces atom economy and inflates raw material costs. Furthermore, these reactions typically generate stoichiometric amounts of iodobenzene as a byproduct. Iodobenzene is not only an organic waste burden that requires costly disposal but also possesses high reactivity that can lead to unwanted side reactions with the substrate. Additionally, traditional halogen sources often involve the release of toxic halogen molecules, posing safety risks and environmental compliance challenges that modern chemical plants strive to eliminate.
The Novel Approach
In stark contrast, the method disclosed in CN111499524B offers a paradigm shift by replacing harsh oxidants with a photochemically driven radical system. By employing cyclic diacyl peroxides (such as MPO) in conjunction with simple halide salts like cesium iodide, the process generates the necessary reactive species under mild light conditions. This eliminates the need for expensive hypervalent iodine oxidants and avoids the formation of iodobenzene waste entirely. The reaction proceeds through a highly selective radical pathway that functionalizes the beta-position of the hydroxyl group with remarkable precision. This novel approach significantly simplifies the workup procedure, as the byproducts are generally easier to separate than the complex mixtures formed in traditional oxidation reactions. Consequently, this translates to cost reduction in pharmaceutical intermediates manufacturing by lowering both material input costs and waste treatment expenses.
Mechanistic Insights into Photochemical Radical Cyclization
The elegance of this synthesis lies in its detailed radical mechanism, which ensures high regioselectivity and yield. The process initiates when the cyclic diacyl peroxide reacts with the halide salt under light irradiation to form an unstable oxygen-halogen bond. This bond is prone to homolytic cleavage, generating a highly active halogen radical species. This radical then abstracts a hydrogen atom from the substrate via a specific 1,5-hydrogen migration process, creating a carbon-centered radical at the precise location needed for cyclization. 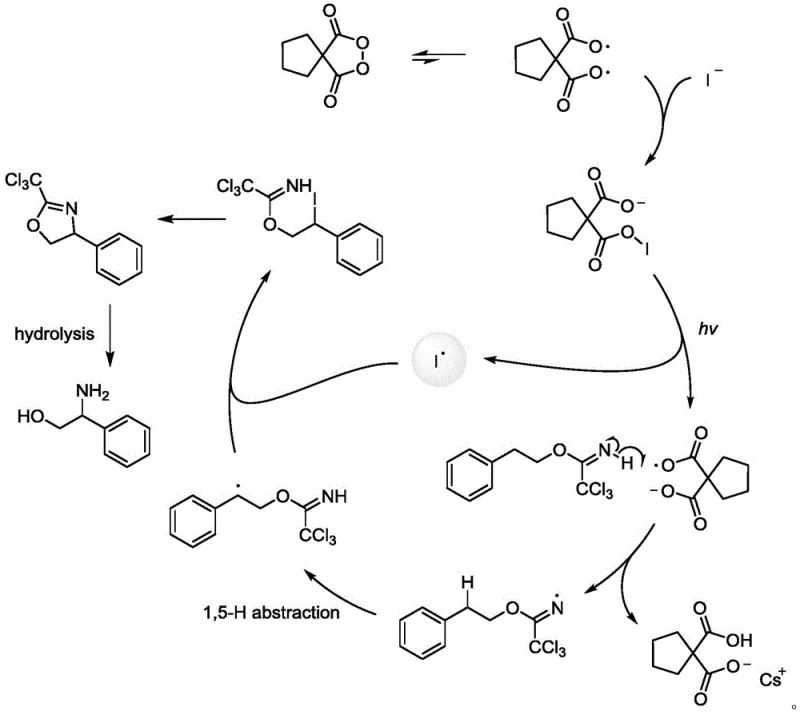 Subsequent intramolecular trapping of this radical by the nitrogen functionality leads to the formation of the oxazoline intermediate. This mechanistic pathway is superior because it avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal contamination in the final API. The control over the radical lifecycle ensures that side reactions are minimized, resulting in a cleaner impurity profile which is a critical parameter for regulatory approval in drug substance manufacturing.
Subsequent intramolecular trapping of this radical by the nitrogen functionality leads to the formation of the oxazoline intermediate. This mechanistic pathway is superior because it avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal contamination in the final API. The control over the radical lifecycle ensures that side reactions are minimized, resulting in a cleaner impurity profile which is a critical parameter for regulatory approval in drug substance manufacturing.
Furthermore, the mechanism explains the observed difference in reactivity between different chain lengths. Experimental data indicates that the formation of five-membered rings (leading to beta-aminoalcohols where n=0) occurs at a faster rate than six-membered rings (gamma-aminoalcohols where n=1). This kinetic preference is attributed to the favorable entropy and enthalpy factors associated with the 1,5-hydrogen abstraction and subsequent cyclization steps in the five-membered transition state. Understanding this nuance allows process chemists to optimize reaction times and temperatures specifically for the desired chain length, ensuring maximum throughput. The ability to tune the reaction outcome based on fundamental mechanistic understanding provides a robust framework for developing diverse libraries of aminoalcohol derivatives for drug discovery programs.
How to Synthesize Aminoalcohol Compounds Efficiently
Implementing this photochemical protocol requires careful attention to reagent ratios and solvent selection to achieve the reported high yields. The standard procedure involves dissolving the trichloroacetimidate precursor in a chlorinated solvent, followed by the addition of the peroxide and iodide source. The reaction is then exposed to light, driving the radical cascade to completion. Following the cyclization, a two-step hydrolysis sequence involving acid and base treatment liberates the free aminoalcohol. This streamlined workflow minimizes unit operations compared to multi-step classical syntheses, making it an attractive option for process intensification.
- Dissolve the trichloroacetimidate substrate in a suitable solvent such as dichloromethane, then add cyclic diacyl peroxide (e.g., MPO) and a halide salt (e.g., Cesium Iodide).
- Irradiate the reaction mixture with natural or artificial light to facilitate the formation of the oxygen-halogen bond and subsequent radical cyclization to form the oxazoline intermediate.
- Hydrolyze the resulting oxazoline salt using inorganic acid followed by alkaline treatment to yield the final high-purity aminoalcohol compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts and stoichiometric hypervalent iodine reagents directly impacts the cost structure of the supply chain. By utilizing commodity chemicals like cesium iodide and commercially available peroxides, the dependency on volatile specialty reagent markets is reduced. This stability in raw material sourcing enhances supply chain reliability, ensuring consistent production schedules without the risk of bottlenecks associated with scarce oxidants. Moreover, the simplified purification process reduces the consumption of chromatography media and solvents during downstream processing, contributing to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of expensive hypervalent iodine oxidants and the reduction of halide salt usage from 3 equivalents to near-stoichiometric levels significantly lowers the bill of materials. Additionally, the avoidance of heavy metal catalysts removes the need for costly scavenging steps and rigorous metal testing, which are often required for pharmaceutical grade materials. The overall process efficiency means less energy and time are spent on purification, leading to a lower cost of goods sold (COGS) for the final aminoalcohol intermediates.
- Enhanced Supply Chain Reliability: The reagents used in this photochemical method, such as dichloromethane, methanol, and cesium iodide, are widely available bulk chemicals with stable global supply chains. Unlike specialized ligands or custom-synthesized oxidants that may have long lead times, these inputs can be sourced from multiple vendors, mitigating the risk of supply disruption. This diversification of the supply base ensures that production of high-purity aminoalcohols can continue uninterrupted, supporting just-in-time manufacturing models for downstream API production.
- Scalability and Environmental Compliance: The reaction operates under mild conditions using light as a traceless reagent, which simplifies reactor design and safety protocols. The absence of toxic halogen gas release and the reduction of organic waste (iodobenzene) align with increasingly stringent environmental regulations. This green chemistry profile facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates and reduces the environmental footprint of the manufacturing site, appealing to sustainability-focused stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and optimization parameters. Understanding these details helps technical teams evaluate the feasibility of integrating this method into existing production lines.
Q: What are the advantages of this photochemical method over traditional hypervalent iodine oxidation?
A: Unlike traditional methods that require stoichiometric amounts of expensive hypervalent iodine oxidants and generate iodobenzene waste, this novel approach utilizes cyclic diacyl peroxides and halide salts under light. This significantly improves atom economy, reduces hazardous organic waste, and simplifies the purification process by avoiding heavy metal or complex oxidant residues.
Q: Can this method be scaled for industrial production of beta-aminoalcohols?
A: Yes, the process demonstrates excellent scalability potential. The reaction conditions are mild, utilizing natural light or standard LED sources, and the reagents like MPO and Cesium Iodide are commercially available. The patent reports high yields (up to 96%) for beta-aminoalcohols (n=0), indicating a robust pathway suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What solvents are optimal for maximizing yield in this radical cyclization?
A: According to the experimental data, dichloromethane is the preferred solvent, delivering the highest yields (96%). Tetrahydrofuran and acetonitrile are also effective alternatives with yields close to 90%. Non-polar or aromatic solvents like toluene result in negligible conversion, highlighting the importance of solvent polarity in stabilizing the radical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoalcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical halogenation technology described in CN111499524B for the production of high-value aminoalcohol intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facilities are equipped with advanced photochemical reactors and stringent purity specifications to handle sensitive radical reactions safely and efficiently. With our rigorous QC labs, we guarantee that every batch of aminoalcohol meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-effective supply chain for your critical aminoalcohol building blocks, driving your drug development programs forward with confidence.
