Advanced Green Synthesis of 2,3,5-Trichloropyridine for Scalable Agrochemical Manufacturing
Advanced Green Synthesis of 2,3,5-Trichloropyridine for Scalable Agrochemical Manufacturing
The global demand for high-performance herbicides and insecticides continues to drive the need for robust, scalable supply chains of key heterocyclic intermediates. Among these, 2,3,5-trichloropyridine stands out as a critical building block for synthesizing major agrochemicals such as clodinafop-propargyl and oxazapyr. A pivotal advancement in this domain is detailed in Chinese Patent CN108912043B, which discloses a novel, environmentally benign synthetic route that addresses longstanding inefficiencies in traditional manufacturing. This patent introduces a streamlined two-step process utilizing 2,3,5,6-tetrachloropyridine as the starting material, leveraging hydrazinolysis followed by oxidative deamination. For R&D directors and procurement strategists, this methodology represents a significant leap forward, offering a pathway to high-purity products while drastically reducing the environmental footprint associated with heavy metal waste and complex purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,3,5-trichloropyridine has been plagued by processes that are either economically inefficient or environmentally hazardous. One prominent prior art method, disclosed in patent WO0155-11, relies on the dechlorination of pentachloropyridine using excessive amounts of zinc powder. While chemically feasible, this approach suffers from severe drawbacks, including poor atom economy and the generation of substantial quantities of zinc-containing waste, which necessitates costly and complex wastewater treatment protocols. Another existing route involves a multi-step sequence starting from 2-chloropyridine, requiring sequential alkoxylation and chlorination; this pathway is operationally cumbersome, generates significant industrial three wastes, and often results in lower overall yields due to the accumulation of impurities at each stage. Furthermore, catalytic ring-closure methods using trichloroacetaldehyde and acrylonitrile often demand harsh reaction conditions and large amounts of metal salts, leading to difficult process control and suboptimal product quality that fails to meet the stringent specifications required for modern pharmaceutical and agrochemical applications.
The Novel Approach
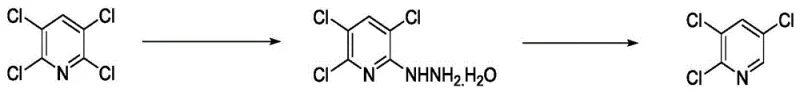
In stark contrast to these legacy methods, the technology outlined in CN108912043B offers a refined, green chemistry solution that prioritizes both efficiency and sustainability. The core innovation lies in the strategic use of 2,3,5,6-tetrachloropyridine, a readily available and cost-effective feedstock, which undergoes a selective nucleophilic substitution with hydrazine hydrate. This initial step is conducted under mild thermal conditions, typically between 60°C and 65°C, ensuring high conversion rates without the need for extreme pressures or exotic catalysts. The resulting intermediate, 2,3,5-trichloro-6-hydrazinopyridine hydrate, is then subjected to a controlled oxidative deamination using sodium hypochlorite in an alkaline medium. This second transformation cleanly removes the hydrazino group to yield the target 2,3,5-trichloropyridine with exceptional purity. By eliminating the reliance on heavy metal reductants like zinc and avoiding complex multi-step functional group manipulations, this novel approach significantly simplifies the operational workflow and reduces the burden on downstream purification units.
Mechanistic Insights into Hydrazinolysis and Oxidative Deamination
The success of this synthetic route hinges on the precise electronic activation of the pyridine ring and the careful control of reaction kinetics. In the first step, the presence of four electron-withdrawing chlorine atoms on the pyridine ring significantly activates the C-6 position towards nucleophilic attack. When hydrazine hydrate is introduced in a polar protic solvent like methanol, it acts as a potent nucleophile, displacing the chlorine atom at the 6-position through an addition-elimination mechanism. The reaction temperature is maintained at 60-65°C to provide sufficient activation energy for the substitution while preventing potential side reactions such as over-hydrazination or solvent degradation. The stoichiometry is carefully balanced, with a molar ratio of tetrachloropyridine to hydrazine hydrate optimized between 1:1.1 and 1:1.15, ensuring complete consumption of the starting material while minimizing excess reagent waste. This step yields the stable hydrazino-intermediate in high purity, setting the stage for the final transformation.
The subsequent oxidative deamination step is equally critical for achieving the desired product profile. The hydrazino-intermediate is treated with an aqueous solution of sodium hypochlorite under alkaline conditions, typically using sodium hydroxide to maintain a pH that facilitates the oxidation. The mechanism likely involves the formation of a diazene or similar nitrogen-rich transient species which rapidly decomposes to release nitrogen gas, leaving behind the aromatic trichloropyridine system. The reaction is exothermic and is conducted at 70-75°C to ensure rapid completion within approximately one hour. Crucially, the use of sodium hypochlorite as the oxidant is advantageous from a safety and cost perspective compared to heavier oxidants like chromates or permanganates. The alkaline environment prevents the formation of acidic byproducts that could corrode equipment or complicate isolation. Following the reaction, simple cooling and filtration allow for the separation of the crude product, which is then purified via reduced pressure distillation to remove trace solvents and minor impurities, resulting in a final product with content exceeding 98%.
How to Synthesize 2,3,5-Trichloropyridine Efficiently
Implementing this synthesis protocol requires strict adherence to the specified reaction parameters to maximize yield and safety. The process begins with the preparation of the reaction vessel, typically a glass-lined or stainless steel reactor equipped with efficient stirring and temperature control systems. Methanol is charged as the solvent, followed by the addition of 2,3,5,6-tetrachloropyridine and hydrazine hydrate. The mixture is heated to the target range of 60-65°C and held for two hours to ensure full conversion to the hydrazino-intermediate. Upon completion, the mixture is cooled to near freezing (0-5°C) to precipitate the intermediate, which is collected by filtration. This solid is then redissolved or suspended in water containing sodium hydroxide, and sodium hypochlorite is added dropwise while maintaining the temperature at 70-75°C. After the oxidation is complete, the mixture is cooled again to crystallize the crude 2,3,5-trichloropyridine. For a comprehensive breakdown of the exact reagent quantities, stirring speeds, and distillation parameters, please refer to the standardized operating procedure below.
- React 2,3,5,6-tetrachloropyridine with hydrazine hydrate in methanol at 60-65°C to form 2,3,5-trichloro-6-hydrazinopyridine hydrate.
- Treat the hydrazino-intermediate with sodium hypochlorite in an alkaline environment at 70-75°C to effect oxidative deamination.
- Cool the reaction mixture, filter the crude product, and purify via reduced pressure distillation to obtain high-purity 2,3,5-trichloropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the waste management profile. By replacing zinc powder with hydrazine and hypochlorite, the process eliminates the generation of heavy metal sludge, which is notoriously expensive to treat and dispose of in compliance with increasingly stringent environmental regulations. This shift not only reduces direct disposal costs but also mitigates the risk of regulatory shutdowns or fines associated with heavy metal discharge. Furthermore, the raw materials employed—2,3,5,6-tetrachloropyridine, hydrazine hydrate, and sodium hypochlorite—are commodity chemicals with stable, global supply chains. This ensures a reliable [agrochemical intermediate] supplier status, as the production is not dependent on scarce or geopolitically sensitive reagents. The robustness of the process also means fewer batch failures and more consistent output, which is crucial for maintaining just-in-time inventory levels for downstream pesticide manufacturers.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and difficult-to-handle reagents. Traditional methods utilizing zinc powder require significant capital expenditure for metal recovery systems or waste neutralization, which adds a hidden cost to every kilogram of product produced. In contrast, the hydrazine-based route utilizes inexpensive oxidants and solvents that can often be recovered and recycled. The high atom economy of the substitution reaction means that less raw material is wasted as byproducts, directly lowering the cost of goods sold (COGS). Additionally, the mild reaction temperatures reduce energy consumption for heating and cooling cycles compared to high-temperature ring-closure methods. These factors combine to create a manufacturing process that is inherently leaner and more cost-effective, allowing for competitive pricing in the global market for [high-purity agrochemical intermediates].
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the fine chemical industry, where disruptions can halt production lines for weeks. This synthesis method enhances reliability by relying on a short, two-step sequence that minimizes the number of unit operations and potential failure points. The starting material, 2,3,5,6-tetrachloropyridine, is a stable solid that is easy to transport and store, unlike some gaseous or highly reactive precursors used in alternative routes. The reaction times are relatively short, typically around two hours per step, which increases the throughput capacity of existing reactor trains. This efficiency allows manufacturers to respond more agilely to fluctuations in market demand. By reducing the complexity of the synthesis, the risk of batch-to-batch variability is minimized, ensuring that customers receive a consistent product specification every time, thereby strengthening long-term supplier-buyer relationships.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, but this route is designed with scalability in mind. The use of heterogeneous filtration for intermediate isolation and simple distillation for final purification are unit operations that scale linearly and predictably. There are no exothermic runaways that are difficult to manage at large scales, nor are there requirements for specialized high-pressure autoclaves. From an environmental standpoint, the process aligns perfectly with green chemistry principles. The absence of heavy metals and the use of aqueous workups significantly reduce the toxicity of the effluent. This makes it easier for facilities to obtain and maintain environmental permits, ensuring uninterrupted [commercial scale-up of complex agrochemical intermediates]. The reduced waste load also lowers the carbon footprint of the manufacturing process, a key metric for multinational corporations aiming to meet their sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application of the patent data, focusing on yield consistency, safety profiles, and downstream compatibility. These insights are derived directly from the experimental examples and comparative data provided in the source documentation, offering a transparent view of the technology's capabilities.
Q: Why is the hydrazine-based route preferred over zinc powder dechlorination for 2,3,5-trichloropyridine?
A: Traditional zinc powder methods generate significant heavy metal waste and suffer from poor atom economy. The hydrazine route described in CN108912043B eliminates toxic metal residues, simplifies wastewater treatment, and utilizes readily available raw materials for a more sustainable process.
Q: What are the typical yield and purity specifications for this synthesis method?
A: According to the patent examples, the process consistently achieves yields exceeding 92% with product content reaching 98%. The mild reaction conditions minimize byproduct formation, ensuring a clean impurity profile suitable for downstream pesticide synthesis.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process uses common solvents like methanol and standard reagents like sodium hypochlorite. The reaction temperatures are moderate (60-75°C), and the workup involves simple filtration and distillation, making it highly adaptable for commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN108912043B are realized in our own manufacturing facilities. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs, which utilize advanced analytical techniques to verify the identity and purity of every batch of 2,3,5-trichloropyridine. We understand that for our partners in the agrochemical and pharmaceutical sectors, consistency is key, and our state-of-the-art infrastructure is designed to deliver exactly that, batch after batch.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Whether you are looking to secure a long-term supply of this critical intermediate or need assistance in optimizing your own production processes, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this green synthesis method can drive value and efficiency for your organization.
