Advanced Synthesis of 2,3,5-Trichloropyridine: A Cost-Effective Route for Global Agrochemical Manufacturing
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of key pyridine intermediates. Patent CN111909080B introduces a transformative preparation method for 2,3,5-trichloropyridine, a critical building block for herbicides such as pyriminobac-methyl and clodinafop-propargyl. This technology addresses long-standing inefficiencies in traditional manufacturing by replacing hazardous phosphonate solvents with a benign methanol-water system. By leveraging selective zinc powder reduction, the process achieves exceptional yields of 90% and HPLC purities exceeding 98%, setting a new benchmark for environmental compliance and operational simplicity in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5-trichloropyridine has relied heavily on processes disclosed in patents such as U.S. Pat. No. 4,581,194, which utilize dimethyl or monomethyl methylphosphonate as the reaction medium. These conventional routes suffer from significant economic and environmental drawbacks, primarily due to the high cost and scarcity of alkyl phosphonate solvents. Furthermore, the removal of phosphorus-containing residues creates immense pressure on three-waste treatment facilities, often requiring complex downstream purification steps that erode profit margins. The reported yields in these legacy processes typically hover between 76% and 78.9%, with product purity struggling to consistently exceed 97.6%, necessitating additional recrystallization cycles that further impact throughput and efficiency.
The Novel Approach
The innovative methodology described in CN111909080B fundamentally reengineers the reduction landscape by employing a mixed solvent system of methanol and water. This strategic shift eliminates the dependency on expensive phosphonates, drastically reducing raw material expenditures while simplifying the waste stream to manageable aqueous and organic phases. The process utilizes zinc powder as a robust reducing agent in the presence of ammonium chloride, facilitating a highly selective dechlorination at the 4-position of the pyridine ring. Optimization studies reveal that maintaining a specific mass ratio of organic solvent to water (preferably 1:1) and controlling the zinc equivalence to 1.5 moles per mole of substrate maximizes conversion. This approach not only elevates the molar yield to an impressive 90% but also ensures the final product meets stringent purity specifications required for downstream pharmaceutical and agrochemical applications.
Mechanistic Insights into Zinc-Mediated Selective Dechlorination
The core of this synthesis lies in the precise control of electron transfer during the reductive dechlorination of 2,3,4,5-tetrachloropyridine. Zinc powder serves as the electron donor, initiating a single-electron transfer mechanism that targets the chlorine atom at the 4-position, which is electronically activated by the adjacent nitrogen and chlorine substituents. The presence of ammonium chloride plays a pivotal role as a proton source and buffer, stabilizing the intermediate radical species and preventing the formation of unwanted byproducts. The solvent environment, specifically the methanol-water mixture, is critical for modulating the solubility of the starting material versus the product; methanol ensures adequate dissolution of the tetrachloro-species, while water facilitates the precipitation of zinc salts and aids in the subsequent crystallization of the trichloro-product. This delicate balance prevents over-reduction to 2,5-dichloropyridine, a common side reaction that plagues less optimized systems.
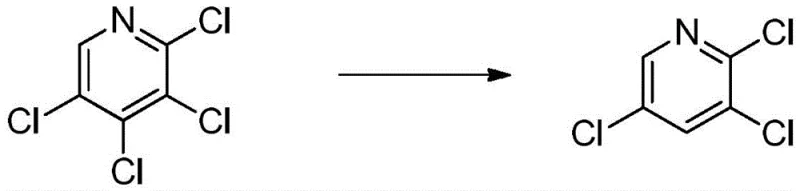
Impurity control is achieved through rigorous optimization of reaction parameters, particularly temperature and time. Data indicates that extending the reaction beyond 2 hours at 72°C offers diminishing returns, as the equilibrium shifts slightly towards over-reduced species without significant gains in starting material conversion. The HPLC profiles demonstrate that under optimal conditions, the residual 2,3,4,5-tetrachloropyridine is minimized to approximately 4.33%, while the over-reduced 2,5-dichloropyridine is kept below 13.42%. The post-reaction workup leverages the differential solubility of the components; hot filtration removes insoluble zinc chloride and excess zinc, while cooling the filtrate to 0-5°C induces the selective crystallization of the target 2,3,5-trichloropyridine. Washing the filter cake with dilute hydrochloric acid further removes basic impurities and residual zinc salts, ensuring the final isolate achieves the requisite 98% purity for commercial use.
How to Synthesize 2,3,5-Trichloropyridine Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing multipurpose reactors, requiring no specialized high-pressure equipment. The process begins with the charging of 2,3,4,5-tetrachloropyridine into a reactor followed by the addition of the methanol-water solvent mixture, ensuring a homogeneous phase before the introduction of the reducing agent. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and crystallization kinetics are essential for reproducibility. For the complete standardized synthesis steps and quality control checkpoints, please refer to the technical guide below.
- Charge 2,3,4,5-tetrachloropyridine into a reactor with a mixed solvent of methanol and water (mass ratio 1: 1 to 1:1.5) and stir to dissolve.
- Add zinc powder (1.2 to 1.5 molar equivalents) and ammonium chloride, then heat the mixture to 72°C and maintain for 2 hours.
- Filter hot to remove zinc salts, cool the filtrate to 0-5°C for crystallization, and wash the resulting cake with hydrochloric acid to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this zinc-mediated reduction pathway offers substantial strategic benefits beyond mere technical feasibility. The elimination of alkyl phosphonates removes a volatile cost component from the bill of materials, insulating the production budget from fluctuations in specialty solvent pricing. Additionally, the simplified waste profile—consisting primarily of aqueous zinc salts and recoverable methanol—significantly lowers the operational expenditure associated with environmental compliance and hazardous waste disposal. This streamlined process enhances the overall reliability of the supply chain by reducing the number of unit operations and minimizing the risk of batch failures due to complex solvent handling issues.
- Cost Reduction in Manufacturing: The substitution of expensive phosphonate solvents with commodity-grade methanol and water results in a drastic reduction in raw material costs. Furthermore, the ability to recycle the mother liquor back into the reaction cycle minimizes solvent loss, creating a closed-loop system that maximizes resource efficiency. The high molar yield of 90% ensures that feedstock utilization is optimized, directly translating to a lower cost per kilogram of the active intermediate compared to legacy methods.
- Enhanced Supply Chain Reliability: By relying on widely available reagents such as zinc powder and methanol, the process mitigates the risk of supply disruptions often associated with niche specialty chemicals. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising purity, ensures consistent batch-to-batch quality. This stability allows for more accurate production planning and shorter lead times, enabling suppliers to respond agilely to market demands for agrochemical intermediates.
- Scalability and Environmental Compliance: The absence of phosphorus-containing waste streams simplifies the effluent treatment process, making it easier to meet increasingly stringent environmental regulations. The exothermic nature of the zinc reduction is easily manageable in large-scale reactors through controlled dosing, ensuring safe scale-up from pilot to commercial tonnage. This environmental compatibility not only reduces regulatory burden but also aligns with the sustainability goals of modern chemical enterprises, enhancing the brand value of the final supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature, providing a clear understanding of the process capabilities. Understanding these nuances is vital for R&D teams evaluating technology transfer and for procurement specialists assessing long-term viability.
Q: Why is the methanol-water solvent system superior to phosphonate solvents?
A: Traditional methods use expensive alkyl phosphonates which complicate waste treatment. The methanol-water system significantly reduces raw material costs, simplifies three-waste processing, and allows for solvent recycling, enhancing overall process sustainability.
Q: What is the critical factor for controlling the 2,5-dichloropyridine impurity?
A: The molar ratio of zinc powder is critical. Using approximately 1.5 equivalents of zinc optimizes the selectivity for the 4-position dechlorination while minimizing over-reduction to 2,5-dichloropyridine, achieving HPLC purity up to 98%.
Q: Can the mother liquor be recycled in this process?
A: Yes, the second filtrate obtained after crystallization can be recycled and reused as part of the mixed solvent in the reduction step. This maximizes industrial application value by reducing solvent consumption and production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical and pharmaceutical sectors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 2,3,5-trichloropyridine with stringent purity specifications, supported by our rigorous QC labs that employ advanced HPLC and GC-MS methodologies to guarantee every batch meets international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to our manufacturing platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a secure and efficient supply of this vital chemical building block.
