Optimizing Lansoprazole Production: A Technical Breakthrough in One-Pot Synthesis and Commercial Scalability
Introduction to Advanced Lansoprazole Manufacturing Technology
The pharmaceutical industry continuously demands more efficient, environmentally benign, and cost-effective pathways for the production of critical Active Pharmaceutical Ingredients (APIs) and their intermediates. In this context, the synthesis method disclosed in patent CN112707889A represents a significant technological leap forward for the manufacturing of Lansoprazole, a widely prescribed proton pump inhibitor. This patent details a robust one-pot methodology that integrates condensation and oxidation steps under alkaline conditions, fundamentally altering the traditional production landscape. By leveraging aqueous solvents and phase transfer catalysis, this process addresses long-standing industry pain points regarding yield, purity, and environmental impact. The technical data presented demonstrates a total molar yield exceeding 92% and an HPLC purity greater than 99.9%, setting a new benchmark for quality in the supply chain. For R&D directors and procurement strategists, understanding the nuances of this patented route is essential for securing a competitive edge in the global market for gastric therapeutics.
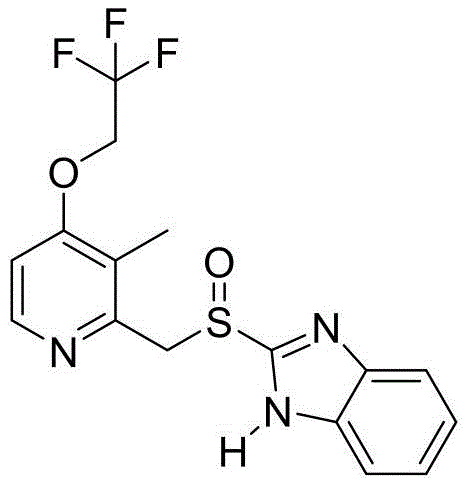
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Lansoprazole has been plagued by several critical inefficiencies that hinder scalable and cost-effective production. Traditional routes often rely on multi-step processes involving expensive and toxic catalysts, such as vanadium oxytrichloride, which introduce significant risks of heavy metal contamination in the final API. Furthermore, existing methodologies frequently utilize organic solvent systems, such as ethanol-water mixtures, which are notoriously difficult to recover and separate, leading to excessive solvent consumption and high volumes of hazardous waste. Previous patents, such as CN104530006, describe processes where the lack of a dedicated refining step results in low content products that are prone to discoloration and instability during the drying phase. The rapid growth of peroxidized sulfone impurities in these conventional routes necessitates repeated purification cycles, driving up operational costs and extending lead times. Additionally, the total molar yields in many legacy processes hover around 68% or lower, representing a substantial loss of valuable raw materials and limiting the overall throughput of manufacturing facilities.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach outlined in the referenced patent introduces a streamlined one-pot synthesis that drastically simplifies the operational workflow. By conducting both condensation and oxidation in a single reactor using water as the primary solvent, this method eliminates the need for complex solvent exchange and extraction procedures. The introduction of a phase transfer catalyst allows for efficient oxidation in an aqueous environment, bypassing the need for toxic vanadium reagents entirely. This shift not only mitigates the risk of metal impurities but also significantly lowers the raw material costs associated with catalyst procurement. The process is designed to operate under mild reaction conditions, typically between -10°C and 25°C during the oxidation phase, which enhances safety and reduces energy consumption for heating or cooling. Most importantly, the integration of a specific refining step using weak bases protects the sensitive sulfinyl linkage, ensuring that the final product maintains exceptional stability and purity profiles suitable for direct pharmaceutical application.
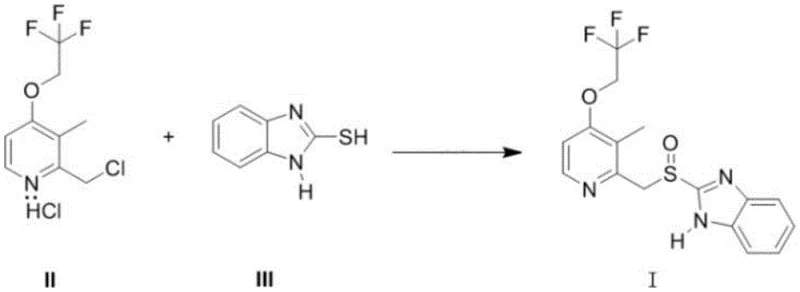
Mechanistic Insights into Phase Transfer Catalyzed Oxidation
The core innovation of this synthesis lies in the sophisticated application of phase transfer catalysis (PTC) to drive the oxidation of the sulfide intermediate to the desired sulfoxide. In a typical aqueous-organic biphasic system, the oxidant (sodium hypochlorite) resides in the aqueous phase, while the organic sulfide substrate may have limited solubility or reactivity at the interface. The addition of quaternary ammonium salts, such as tetrabutylammonium bromide or trimethyl benzyl ammonium chloride, acts as a molecular shuttle, transporting the hypochlorite anion into the organic phase or facilitating the reaction at the interface with high efficiency. This mechanistic advantage ensures that the oxidation proceeds rapidly and selectively, minimizing the over-oxidation to the corresponding sulfone, which is a common and difficult-to-remove impurity in proton pump inhibitor synthesis. The precise control of stoichiometry and temperature during this phase is critical; the patent specifies maintaining the reaction between -10°C and 25°C to balance reaction kinetics with selectivity. By optimizing these parameters, the process achieves a conversion rate where the residual thioether is less than 2.0%, demonstrating superior control over the reaction pathway compared to non-catalyzed or metal-catalyzed alternatives.
Furthermore, the stability of the Lansoprazole molecule is meticulously managed through the strategic use of weak bases during the purification stage. Lansoprazole is known to be acid-labile and susceptible to degradation, particularly under thermal stress during drying. The novel process incorporates a refining step where the crude product is dissolved in ketone or alcohol solvents in the presence of weak bases like triethylamine or ammonia. This creates a buffered micro-environment with a pH maintained between 7 and 7.8, which neutralizes any trace acidic byproducts that could catalyze decomposition. Following crystallization, the product is dried under a nitrogen atmosphere, effectively excluding oxygen and moisture that could otherwise promote oxidative degradation or hydrolysis. This dual-protection strategy—chemical buffering and inert atmosphere drying—is the key mechanism behind achieving the reported HPLC purity of >99.9% and ensuring the physical appearance of the dry product remains white and free from discoloration, a common defect in inferior manufacturing processes.
How to Synthesize Lansoprazole Efficiently
Implementing this advanced synthesis route requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the condensation of 2-mercaptobenzimidazole and the chloromethyl pyridine derivative in an aqueous alkaline medium, followed immediately by the oxidative step without isolating the intermediate sulfide. This telescoping of steps is crucial for maintaining process efficiency and minimizing material handling losses. Operators must carefully monitor the temperature profile, particularly during the exothermic addition of the oxidant, to prevent thermal runaway which could compromise selectivity. The subsequent work-up involves a simple solid-liquid separation, avoiding the need for large-scale solvent extractions. For the final purification, the choice of solvent system (acetone/isopropanol) and the precise addition of the weak base are critical control points that determine the final crystal habit and impurity profile. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are essential for technology transfer.
- Condense 2-mercaptobenzimidazole and 2-chloromethyl-3-methyl-4-(2,2,2-trifluoroethoxy)pyridine hydrochloride in aqueous alkaline solution at 10-60°C.
- Add alkali, phase transfer catalyst, and oxidant (sodium hypochlorite) to the system at -10 to 25°C for oxidation.
- Refine the crude product using ketone/alcohol solvents with weak base protection under nitrogen atmosphere to ensure stability.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies for Lansoprazole intermediates. The most immediate benefit is the drastic reduction in manufacturing costs driven by the elimination of expensive transition metal catalysts and the simplification of the solvent system. By replacing organic solvents with water for the main reaction sequence, the process significantly lowers the expenditure on raw materials and reduces the complexity of solvent recovery infrastructure. This shift translates directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to improve their margins or offer more affordable treatments to patients. Moreover, the high total molar yield of over 92% means that less starting material is required to produce the same amount of finished goods, further enhancing the economic efficiency of the supply chain. These factors combined create a compelling value proposition for partners looking to secure a reliable lansoprazole intermediate supplier with a focus on cost leadership.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplification of unit operations and the use of commodity-grade reagents. The removal of vanadium-based catalysts eliminates the costly downstream processing steps required to reduce metal residues to ppm levels, which is a regulatory necessity for oral medications. Additionally, the one-pot nature of the reaction reduces labor hours and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities. The use of water as a solvent not only cuts material costs but also reduces the fire hazard classification of the plant, potentially lowering insurance and safety compliance expenses. Qualitative analysis suggests that these cumulative efficiencies result in substantial cost savings compared to traditional multi-step organic syntheses, making the final product highly price-competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable raw materials. 2-Mercaptobenzimidazole and the trifluoroethoxy pyridine derivative are established commodities with robust global supply networks, reducing the risk of bottlenecks associated with exotic or specialized reagents. The robustness of the aqueous reaction system also makes the process less sensitive to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. Furthermore, the high yield and purity reduce the likelihood of batch failures or the need for re-processing, which are common causes of supply delays. By partnering with a manufacturer utilizing this technology, buyers can expect more predictable lead times and a steady flow of high-quality intermediates, crucial for maintaining uninterrupted production schedules for finished dosage forms.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify globally, the green chemistry attributes of this process provide a significant strategic advantage. The reduction of three wastes, specifically the decrease in wastewater generation from 17.3g/g to 4.8g/g of product, dramatically lowers the burden on effluent treatment plants and reduces disposal costs. The absence of heavy metals and the minimization of volatile organic compounds (VOCs) simplify the permitting process for new manufacturing lines and ensure compliance with stringent environmental standards like REACH or local EPA regulations. This environmental compatibility facilitates easier commercial scale-up, as the process can be expanded to 100 MT annual capacity without requiring massive investments in waste management infrastructure. For supply chain heads, this means a future-proof supply source that aligns with corporate sustainability goals and regulatory mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific Lansoprazole synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific production needs. The answers highlight the balance between chemical efficiency, product quality, and operational practicality that defines this modern manufacturing approach.
Q: How does the new synthesis method improve product stability compared to conventional routes?
A: Conventional methods often suffer from product deterioration during drying and rapid growth of peroxidized sulfone impurities. The novel approach utilizes a weak base during the refining step to protect the unstable sulfinyl group and employs a nitrogen atmosphere during drying, ensuring the final HPLC purity exceeds 99.9%.
Q: What are the environmental advantages of this one-pot process?
A: By utilizing water as the primary solvent and avoiding complex solvent extraction steps found in older patents, this method significantly reduces three wastes. Data indicates a reduction in wastewater generation from 17.3g/g to 4.8g/g of finished product, making it highly suitable for green industrial manufacturing.
Q: Why is the phase transfer catalyst critical in this oxidation step?
A: The phase transfer catalyst facilitates the oxidation reaction within the aqueous medium, effectively reducing the residue of lansoprazole thioether and suppressing the formation of sulfone impurities. This ensures a total molar yield of over 92% without the need for expensive vanadium catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lansoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112707889A are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous phase transfer reactions and inert atmosphere drying, guaranteeing that every batch meets stringent purity specifications. With rigorous QC labs employing advanced analytical techniques, we validate that our Lansoprazole intermediates consistently achieve the >99.9% purity benchmarks required by global pharmacopoeias. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your supply chain security and product quality.
We invite you to engage with our technical team to explore how this advanced synthesis route can be tailored to your specific project requirements. Whether you are looking to optimize an existing supply chain or develop a new generic formulation, our experts are ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your organization. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us collaborate to bring high-quality, cost-effective gastric therapies to the market faster and more efficiently.
