Revolutionizing Quinazoline Production: Green Catalysis for Commercial Scale-up
Revolutionizing Quinazoline Production: Green Catalysis for Commercial Scale-up
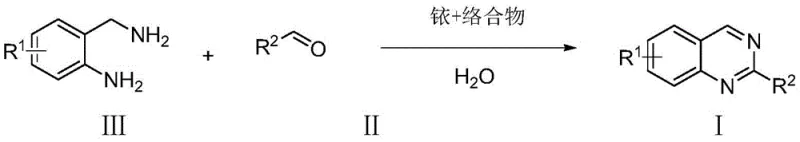
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for constructing privileged heterocyclic scaffolds. Patent CN108264489B, published in June 2021, introduces a groundbreaking methodology for the synthesis of quinazoline derivatives that fundamentally alters the traditional manufacturing landscape. This innovation leverages water-soluble iridium or ruthenium complex catalysts to drive the condensation of aldehydes and o-aminobenzylamines directly in an aqueous medium. Unlike conventional routes that rely on stoichiometric amounts of toxic oxidants and organic solvents, this novel approach operates via an acceptorless dehydrogenative coupling mechanism, releasing environmentally benign hydrogen gas as the sole byproduct. For R&D directors and procurement managers alike, this technology represents a significant leap forward in atom economy and process safety, offering a viable route for the commercial scale-up of complex pharmaceutical intermediates while drastically reducing the environmental burden associated with legacy synthetic methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazoline core has heavily depended on oxidative cyclization strategies that pose substantial challenges for large-scale manufacturing. Traditional protocols typically require the use of harsh, stoichiometric oxidizing agents such as 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ), chloranil, manganese dioxide, or sodium hypochlorite to drive the aromatization step. These reagents are not only expensive and hazardous to handle but also generate massive quantities of toxic waste streams that require complex and costly disposal procedures. Furthermore, these reactions are predominantly conducted in volatile organic solvents, exacerbating safety risks related to flammability and worker exposure. The reliance on hydrogen acceptors, such as unsaturated olefins, further complicates the reaction mixture, often leading to difficult purification processes and lower overall yields due to side reactions. As regulatory pressures regarding waste discharge and solvent emissions intensify globally, these legacy methods are becoming increasingly unsustainable for the production of high-purity pharmaceutical intermediates.
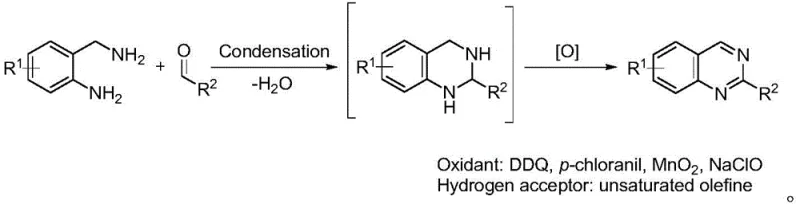
The Novel Approach
In stark contrast to the oxidative burden of prior art, the method disclosed in CN108264489B utilizes a catalytic dehydrogenation strategy that operates cleanly in water. By employing specialized water-soluble organometallic catalysts, the reaction proceeds through a direct condensation followed by intramolecular cyclization and subsequent dehydrogenation without the need for any external oxidant. This "borrowing hydrogen" or acceptorless dehydrogenation pathway ensures that the only byproduct is molecular hydrogen, which can be safely vented or potentially captured for energy recovery. The use of water as the exclusive solvent not only eliminates the cost and hazard of organic solvents but also simplifies the isolation of the product, as many organic quinazolines precipitate or can be easily extracted from the aqueous phase. This paradigm shift enables cost reduction in pharmaceutical manufacturing by streamlining the workup procedure and removing the need for expensive oxidizing reagents, thereby creating a more robust and economically attractive supply chain for critical drug substances.
Mechanistic Insights into Water-Soluble Metal Complex Catalysis
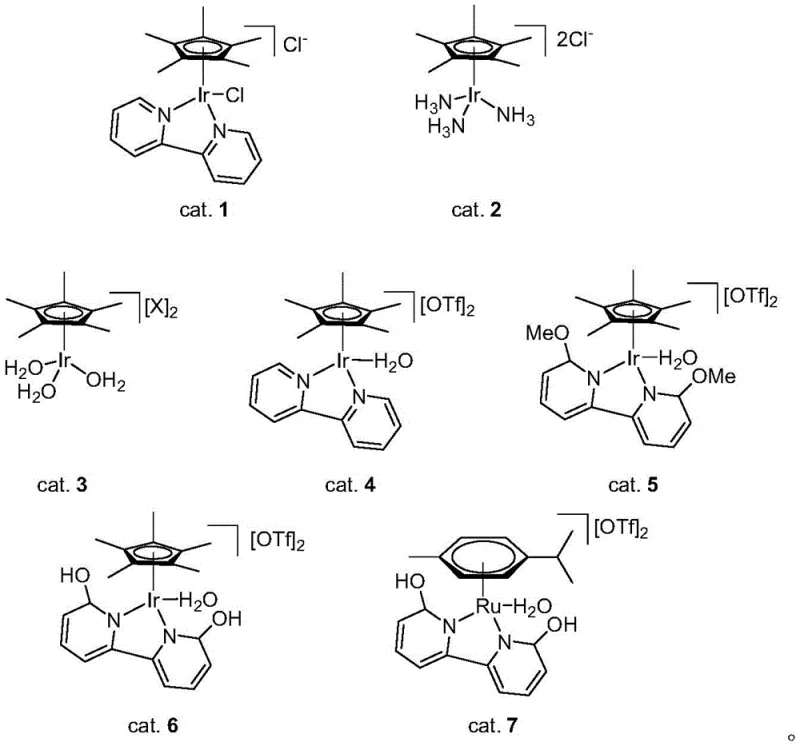
The core of this technological breakthrough lies in the unique capability of half-sandwich iridium and ruthenium complexes to facilitate reversible dehydrogenation in polar media. The catalytic cycle initiates with the coordination of the o-aminobenzylamine and the aldehyde to the metal center, promoting the formation of a hemiaminal intermediate. Subsequently, the metal complex mediates the beta-hydride elimination steps required to aromatize the dihydroquinazoline ring, effectively shuttling hydrogen atoms from the substrate to form H2 gas. The presence of hydrophilic ligands, such as sulfonated or hydroxylated bipyridines, ensures that the catalyst remains dissolved and active in the aqueous phase, preventing precipitation and maintaining high turnover numbers. This mechanistic elegance allows for exceptional functional group tolerance, accommodating a wide range of substituents on both the benzene ring and the aldehyde moiety without compromising reaction efficiency.
Furthermore, the specific design of the catalyst ligands plays a pivotal role in controlling impurity profiles and maximizing yield. The patent details a library of catalysts, including Cp*Ir and p-cymene Ru derivatives, where subtle modifications to the ligand environment (such as the introduction of methoxy or hydroxy groups) can significantly enhance catalytic activity and stability under hydrothermal conditions. For instance, catalysts bearing 6,6'-dihydroxy-2,2'-bipyridine ligands have shown superior performance, likely due to enhanced hydrogen bonding interactions with the solvent or substrate that stabilize key transition states. This level of mechanistic control is crucial for R&D teams aiming to minimize trace metal residues and organic impurities in the final API. By understanding these structure-activity relationships, manufacturers can optimize the process to meet stringent purity specifications required by global regulatory bodies, ensuring the consistent quality of high-purity quinazoline derivatives.
How to Synthesize 2-Substituted Quinazolines Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible for both laboratory optimization and industrial production. The process involves simply combining the aldehyde substrate, o-aminobenzylamine, and a catalytic amount (typically 1-2 mol%) of the water-soluble metal complex in a reaction vessel charged with water. The mixture is then heated, often to temperatures around 130°C, and maintained for a period sufficient to drive the reaction to completion, typically around 12 hours. Following the reaction, the system is cooled, and the water is removed via rotary evaporation, leaving a crude residue that can be purified by standard column chromatography. This operational simplicity reduces the technical barrier for adoption and facilitates rapid scale-up. For detailed standardized operating procedures and specific parameter optimizations, please refer to the guide below.
- Charge a reaction vessel with aldehyde, o-aminobenzylamine, water-soluble metal complex catalyst (1-2 mol%), and water as the solvent.
- Heat the reaction mixture to approximately 130°C and maintain for several hours (typically 12 hours) to allow dehydrogenative cyclization.
- Cool to room temperature, remove water via rotary evaporation, and purify the crude residue by column chromatography to isolate the target quinazoline.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition to this aqueous catalytic method offers profound benefits for supply chain resilience and cost management. The elimination of stoichiometric oxidants like DDQ and manganese dioxide removes a significant cost driver from the bill of materials, as these reagents are often pricey and subject to supply volatility. Moreover, the removal of hazardous waste streams associated with these oxidants drastically lowers the environmental compliance costs and the logistical burden of waste disposal. For procurement managers, this translates into a more predictable cost structure and reduced risk of production stoppages due to regulatory non-compliance or reagent shortages. The ability to source simple, commodity-grade starting materials like benzaldehydes and amines further enhances the economic viability of the process.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the replacement of expensive, single-use oxidants with a reusable catalytic system. By shifting from a stoichiometric oxidative process to a catalytic dehydrogenative one, the consumption of auxiliary chemicals is minimized. Additionally, the use of water as a solvent eliminates the need for purchasing, recovering, or disposing of large volumes of organic solvents, leading to substantial operational expenditure savings. The simplified workup procedure, which avoids complex quenching steps for excess oxidants, also reduces labor and processing time, contributing to overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: Relying on water-soluble catalysts and abundant starting materials mitigates the risk of supply chain disruptions often associated with specialized oxidizing agents. The robustness of the reaction conditions allows for flexible sourcing of raw materials, as the catalyst system tolerates a wide variety of electronic and steric environments on the substrates. This flexibility ensures that production schedules can be maintained even if specific grades of reagents are temporarily unavailable. Furthermore, the green nature of the process aligns with the sustainability goals of major pharmaceutical buyers, potentially securing long-term contracts with partners who prioritize environmentally responsible suppliers.
- Scalability and Environmental Compliance: The inherent safety of running reactions in water at atmospheric pressure (or mild autogenous pressure) makes this process highly scalable. There is no risk of runaway exotherms typical of strong oxidations, and the generation of hydrogen gas can be managed with standard venting systems. From an environmental standpoint, the process generates minimal E-factor (mass of waste per mass of product), primarily consisting of water and small amounts of organic residue. This low waste profile simplifies permitting for new manufacturing lines and ensures compliance with increasingly strict global environmental regulations, future-proofing the supply chain against legislative changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using water as a solvent for quinazoline synthesis?
A: Using water eliminates the need for toxic organic solvents and hazardous oxidants like DDQ or manganese dioxide. This significantly reduces waste disposal costs and aligns with green chemistry principles, enhancing environmental compliance and operational safety.
Q: Which catalysts are most effective for this acceptorless dehydrogenative coupling?
A: Water-soluble iridium and ruthenium complexes, specifically Cp*Ir and p-cymene Ru derivatives bearing bipyridine ligands, demonstrate superior activity. These catalysts facilitate the release of hydrogen gas without requiring external hydrogen acceptors.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the process utilizes inexpensive and readily available starting materials (aldehydes and amines) and operates under relatively mild thermal conditions without high pressure. The absence of stoichiometric oxidants simplifies the workup, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivatives Supplier
As the demand for sustainable and cost-effective synthetic routes grows, NINGBO INNO PHARMCHEM stands ready to leverage this advanced catalytic technology for your project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest industry standards. By integrating green chemistry principles like aqueous catalysis into our portfolio, we offer a competitive edge in both quality and environmental stewardship.
We invite you to discuss how this innovative quinazoline synthesis can optimize your specific supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in pharmaceutical development.
