Advanced Manufacturing of Prasugrel Intermediates: A Green, Scalable Route for Global Supply Chains
Advanced Manufacturing of Prasugrel Intermediates: A Green, Scalable Route for Global Supply Chains
The pharmaceutical industry is constantly seeking robust, environmentally sustainable, and cost-effective pathways for the synthesis of critical antiplatelet agents like prasugrel. Patent CN102219792A, published in October 2011, introduces a transformative methodology that addresses the longstanding inefficiencies and environmental hazards associated with traditional manufacturing processes. This novel approach leverages a sophisticated sequence of Grignard reactions, controlled thermal decompositions, and selective acetylations to construct the complex thienopyridine core with exceptional precision. For R&D directors and supply chain leaders, this patent represents a pivotal shift away from hazardous reagents toward a cleaner, higher-yielding protocol that aligns with modern green chemistry principles. By fundamentally re-engineering the synthetic route, this technology offers a reliable pharmaceutical intermediate supplier with the capability to deliver high-purity materials while drastically reducing the ecological footprint of production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of prasugrel has been plagued by severe operational and environmental drawbacks that hinder efficient cost reduction in pharmaceutical manufacturing. As illustrated in prior art such as Document EP 542411, traditional routes often rely on the use of elemental bromine and tetracol phenixin, reagents known for their extreme toxicity and significant environmental pollution potential. These processes not only impose stringent labor protection requirements but also generate strongly acidic hydrogen bromide as a byproduct, which causes severe corrosion to production equipment, thereby increasing maintenance costs and downtime. Furthermore, the condensation yields in these legacy methods are notoriously poor, often hovering around merely 30%, which renders the entire synthesis economically unviable for large-scale operations. The accumulation of hazardous waste and the inability to scale safely have made these conventional pathways increasingly obsolete in the face of stricter global environmental regulations.
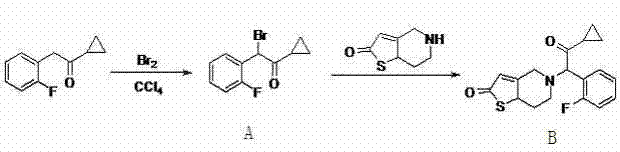
The Novel Approach
In stark contrast, the methodology disclosed in CN102219792A circumvents these pitfalls through a cleverly designed four-step sequence that prioritizes safety and efficiency. The new route initiates with a Grignard reaction under anhydrous conditions, utilizing magnesium powder and tert-butyl perbenzoate to construct the protected intermediate, effectively bypassing the need for hazardous halogenation steps. This is followed by a controlled thermal decomposition under acidic conditions to reveal the core structure, a step that is both high-yielding and operationally simple. The subsequent acetylation and final condensation steps are performed under mild alkaline conditions in polar solvents, ensuring that the process remains gentle on equipment and easy to control. This strategic redesign eliminates the generation of corrosive gases and toxic byproducts, resulting in a process that is not only environmentally friendly but also economically superior due to significantly improved yields and simplified purification protocols.
Mechanistic Insights into Grignard-Mediated Core Construction
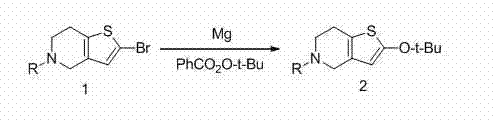
The cornerstone of this innovative synthesis lies in the initial Grignard reaction, which serves as the foundation for building the substituted thienopyridine scaffold. In this critical step, a bromo-substituted precursor (Compound 1) reacts with magnesium powder in an anhydrous solvent such as tetrahydrofuran or toluene to form a reactive organomagnesium species. This intermediate is then quenched with tert-butyl perbenzoate, introducing the necessary oxygen functionality while simultaneously protecting the nitrogen atom with a trityl group. The choice of solvent and temperature control, ranging from -10°C to 70°C, is paramount to ensuring the stability of the Grignard reagent and preventing side reactions that could compromise the integrity of the sensitive heterocyclic ring system. This mechanistic pathway allows for the precise installation of functional groups that are difficult to introduce via direct substitution, thereby enhancing the overall regioselectivity of the synthesis.
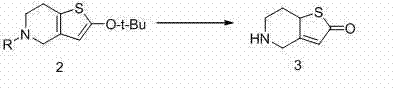
Following the formation of the protected intermediate, the process employs a thermal decomposition strategy to unveil the active ketone functionality required for downstream coupling. Under acidic conditions facilitated by catalysts like p-toluenesulfonic acid or hydrochloric acid, the tert-butoxy and trityl protecting groups are cleaved at elevated temperatures between 120°C and 180°C. This thermal lability is exploited to drive the reaction to completion without the need for harsh chemical reagents that might degrade the product. The result is the formation of 5,6,7,7a-tetrahydro-4H-thieno-[3,2-c]pyridin-2-one (Compound 3) with remarkable purity, often exceeding 99% HPLC purity after simple recrystallization. This high level of impurity control is crucial for meeting the stringent quality standards required for high-purity API intermediates, ensuring that downstream biological testing and final drug formulation are not compromised by trace contaminants.
How to Synthesize Prasugrel Intermediates Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly regarding moisture exclusion during the Grignard formation and precise temperature management during the thermal decomposition phase. Operators must ensure that all solvents are rigorously dried and that the reaction atmosphere is maintained under inert gas protection to prevent the quenching of the organometallic species. The subsequent acetylation and condensation steps benefit from the use of mild bases like diisopropylethylamine or pyridine, which facilitate the reaction without promoting hydrolysis of the sensitive ester linkages. For a detailed breakdown of the specific molar ratios, reaction times, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Perform a Grignard reaction on compound formula 1 using magnesium powder and tert-butyl perbenzoate in an anhydrous solvent to obtain compound formula 2.
- Subject compound formula 2 to thermal decomposition under acidic conditions to yield the key intermediate, compound formula 3.
- Acetylate compound formula 3 using an acetylation reagent to form compound formula 4, followed by condensation with 2-fluoro-alpha-cyclopropyl carbonyl bromobenzyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of highly toxic and corrosive reagents such as elemental bromine and hydrogen bromide significantly lowers the barrier to entry for manufacturing facilities, as it reduces the need for specialized corrosion-resistant equipment and extensive safety infrastructure. This simplification of the production environment directly contributes to cost reduction in pharmaceutical manufacturing by minimizing capital expenditure on reactor linings and waste treatment systems. Furthermore, the use of readily available raw materials ensures a stable supply chain, mitigating the risks associated with sourcing scarce or regulated chemicals that often plague the production of complex heterocyclic drugs.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substantial improvement in reaction yields compared to legacy methods. By avoiding the low-yielding condensation steps characteristic of older routes, the overall material throughput is maximized, meaning less raw material is wasted per kilogram of final product. Additionally, the removal of expensive and hazardous reagents reduces the cost of goods sold (COGS), while the simplified purification steps—often requiring only recrystallization rather than complex chromatography—further drive down processing costs. This efficiency makes the commercial scale-up of complex pharmaceutical intermediates far more attractive from a margin perspective.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available solvents like tetrahydrofuran, toluene, and acetonitrile ensures that production is not bottlenecked by the availability of exotic reagents. This accessibility allows for flexible sourcing strategies and reduces lead times for high-purity pharmaceutical intermediates. Moreover, the robustness of the reaction conditions means that the process is less susceptible to batch failures caused by minor fluctuations in parameters, thereby guaranteeing a consistent and reliable output of material to meet downstream demand schedules without interruption.
- Scalability and Environmental Compliance: From a regulatory standpoint, this route is exceptionally well-positioned for commercial scale-up due to its minimal generation of "three wastes" (wastewater, waste gas, and solid residues). The absence of heavy metal catalysts and halogenated byproducts simplifies waste disposal and lowers the environmental compliance burden, which is increasingly critical in global manufacturing hubs. The straightforward nature of the unit operations—heating, stirring, and filtration—facilitates easy translation from laboratory bench scale to multi-ton industrial reactors, ensuring that supply continuity can be maintained as market demand for antiplatelet therapies grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential partners and licensees.
Q: How does this novel method improve upon conventional prasugrel synthesis?
A: Unlike conventional methods that utilize highly toxic elemental bromine and generate corrosive hydrogen bromide with low yields (around 30%), this novel route employs a Grignard reaction and thermal decomposition. This eliminates severe environmental pollution, reduces equipment corrosion risks, and significantly improves overall yield and operational safety.
Q: What are the purity specifications achievable with this process?
A: The patent data indicates that the intermediate compounds, such as 5,6,7,7a-tetrahydro-4H-thieno-[3,2-c]pyridin-2-one (compound formula 3), can achieve an HPLC purity of 99.2% after recrystallization. The final product also demonstrates high purity levels suitable for pharmaceutical applications.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrialization. It utilizes readily available raw materials, low-toxicity reagents, and simple production operations. The process generates minimal 'three wastes' (wastewater, waste gas, waste residues), making it environmentally compliant and scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prasugrel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of prasugrel intermediate meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific requirements of Grignard chemistry and thermal processing, providing a secure and compliant environment for your most valuable projects.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your production of high-value antiplatelet intermediates.
