Advanced One-Pot Synthesis of Dapagliflozin Intermediate for Commercial Scale-Up
Advanced One-Pot Synthesis of Dapagliflozin Intermediate for Commercial Scale-Up
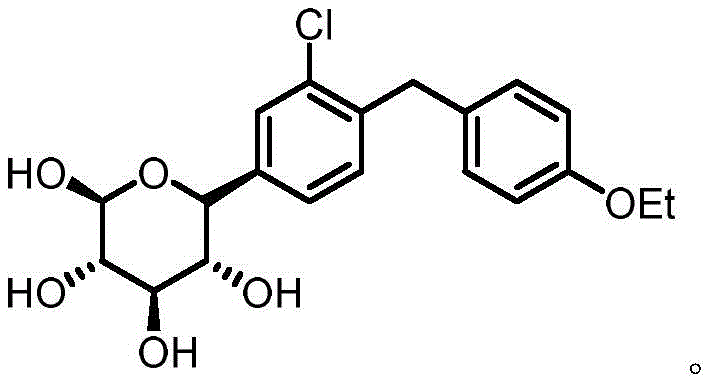
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical Active Pharmaceutical Ingredients (APIs), particularly for high-demand medications like Dapagliflozin, a leading SGLT2 inhibitor used in the management of type 2 diabetes. A significant technological breakthrough in this domain is detailed in patent CN112920030A, which discloses a novel one-pot method for preparing the key intermediate, (5-bromo-2-chlorophenyl)(4-ethoxyphenyl)methanone. This intermediate serves as the foundational building block for the subsequent reduction and glycosylation steps required to synthesize the final drug substance. The structural complexity of Dapagliflozin necessitates precise control over stereochemistry and purity, making the quality of this ketone intermediate paramount for the overall success of the manufacturing campaign. By shifting away from multi-step, hazardous traditional protocols, this new methodology offers a streamlined approach that aligns perfectly with modern green chemistry principles and industrial efficiency standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
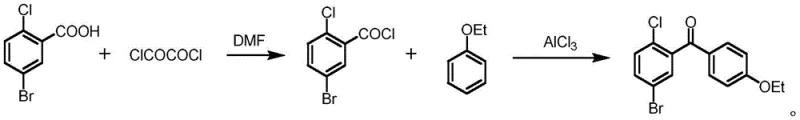
Historically, the synthesis of (5-bromo-2-chlorophenyl)(4-ethoxyphenyl)methanone has relied heavily on classical Friedel-Crafts acylation strategies. As illustrated in the traditional synthetic schemes, this typically involves a two-step sequence where 5-bromo-2-chlorobenzoic acid is first converted into its corresponding acyl chloride using aggressive chlorinating agents such as thionyl chloride or oxalyl chloride. This initial step is fraught with challenges, including the generation of corrosive acidic gases and the requirement for strict anhydrous conditions to prevent hydrolysis. Following the formation of the acyl chloride, a second reaction with phenetole is conducted in the presence of stoichiometric or excess amounts of Lewis acids like aluminum trichloride. This conventional route not only generates substantial quantities of hazardous waste requiring complex neutralization and disposal but also poses significant safety risks due to the exothermic nature of the reactions and the toxicity of the reagents involved. Furthermore, the purification of the resulting ketone often requires extensive chromatographic separation or multiple recrystallizations to remove metal residues and regioisomeric impurities, driving up both production costs and lead times.
The Novel Approach
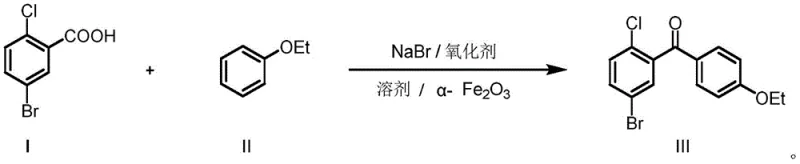
In stark contrast to the cumbersome traditional pathways, the innovative method described in the patent utilizes a direct oxidative coupling strategy that telescopes the activation and coupling steps into a single reactor vessel. This one-pot protocol employs 5-bromo-2-chlorobenzoic acid directly, bypassing the isolation of the unstable acyl chloride intermediate entirely. The reaction system leverages a unique combination of red phosphorus, sodium bromide, and an oxidant to generate the reactive acylating species in situ, which then immediately reacts with phenetole under the influence of a heterogeneous alpha-Fe2O3 catalyst. This approach dramatically simplifies the operational workflow, eliminating the need for separate acyl chlorination equipment and the handling of large volumes of corrosive chlorinating agents. The result is a significantly cleaner reaction profile with fewer by-products, allowing for a much more straightforward isolation process that typically involves simple quenching, phase separation, and recrystallization. This paradigm shift represents a major advancement in process chemistry, offering a robust alternative for reliable dapagliflozin intermediate supplier networks looking to optimize their manufacturing footprint.
Mechanistic Insights into Alpha-Fe2O3 Catalyzed Oxidative Coupling
The core of this technological advancement lies in the sophisticated interplay between the in situ generated acyl bromide and the heterogeneous iron oxide catalyst. Mechanistically, the system operates by utilizing the red phosphorus and sodium bromide in the presence of an oxidant to effectively convert the carboxylic acid group into a highly reactive acyl bromide species within the reaction medium. Unlike the traditional acyl chloride route which relies on harsh chlorinating gases, this bromination pathway is milder and more controllable. The alpha-Fe2O3 catalyst, prepared via a specific coprecipitation and calcination process, plays a pivotal role in activating the aromatic ring of the phenetole and facilitating the electrophilic attack by the acyl bromide. The surface properties of the nanostructured iron oxide likely provide active sites that stabilize the transition state, thereby lowering the activation energy required for the C-C bond formation. This catalytic efficiency allows the reaction to proceed at moderate temperatures, typically between 10°C and 60°C, which is crucial for minimizing thermal degradation and side reactions that often plague high-temperature Friedel-Crafts processes.
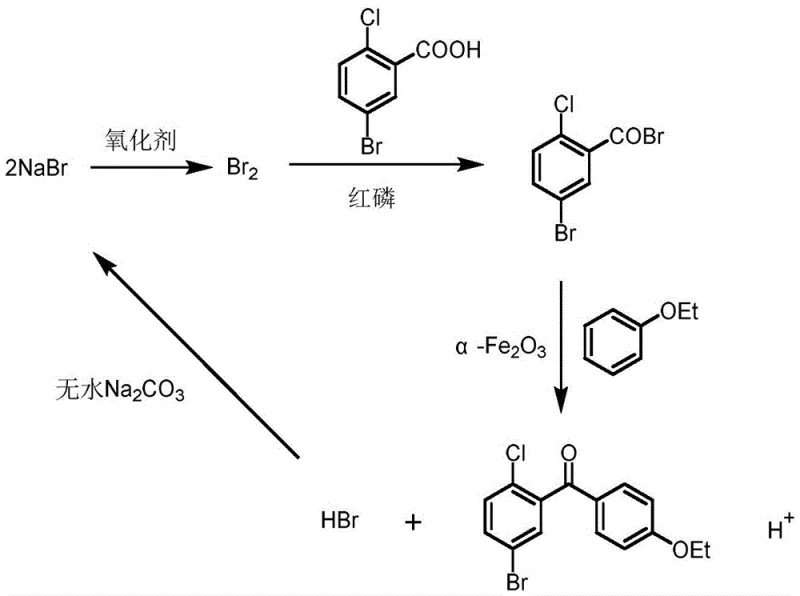
Furthermore, the inclusion of molecular sieves and anhydrous sodium carbonate in the reaction matrix serves a dual purpose of water scavenging and acid neutralization, which is critical for maintaining the integrity of the catalyst and preventing the hydrolysis of the reactive intermediates. The control of impurities is inherently superior in this system because the absence of excessive Lewis acids like AlCl3 prevents the formation of complex coordination by-products that are notoriously difficult to remove. The selectivity for the para-position on the phenetole ring is enhanced by the specific electronic environment created by the catalyst surface, ensuring that the desired (5-bromo-2-chlorophenyl)(4-ethoxyphenyl)methanone is formed with high regioselectivity. This mechanistic elegance translates directly into a product with exceptional purity profiles, often exceeding 99% by HPLC after a single recrystallization step, thereby meeting the stringent quality requirements necessary for high-purity pharmaceutical intermediates intended for global regulatory markets.
How to Synthesize (5-Bromo-2-Chlorophenyl)(4-Ethoxyphenyl)Methanone Efficiently
Implementing this advanced synthesis route requires careful attention to the preparation of the catalytic system and the control of addition rates to manage the exotherm effectively. The process begins with the uniform suspension of the solid components, including the specialized alpha-Fe2O3 catalyst and molecular sieves, in a suitable solvent such as 1,4-dioxane. The oxidant, which can be selected from options like potassium persulfate or tert-butyl hydroperoxide, is introduced to initiate the activation cycle. A critical operational parameter is the dropwise addition of phenetole, which must be controlled precisely to maintain the reaction temperature within the optimal window, ensuring maximum conversion while suppressing potential side reactions. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures validated by experimental data, are outlined below for technical reference.
- Prepare the reaction mixture by combining 5-bromo-2-chlorobenzoic acid, red phosphorus, sodium bromide, anhydrous sodium carbonate, alpha-Fe2O3 catalyst, molecular sieves, and oxidant in a solvent like 1,4-dioxane.
- Add phenetole dropwise to the stirred mixture at a controlled rate while maintaining a constant temperature between 10°C and 60°C.
- After the reaction completes, quench the mixture, separate layers, concentrate, and recrystallize from absolute ethanol to obtain the high-purity ketone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis method presents a compelling value proposition centered around risk mitigation and cost optimization. By eliminating the dependency on hazardous acyl chlorinating agents and stoichiometric Lewis acids, the process inherently reduces the logistical and safety burdens associated with storing and handling dangerous chemicals. This simplification of the raw material portfolio not only enhances workplace safety but also streamlines the supply chain by reducing the number of distinct chemical inputs required for production. The simplified work-up procedure, which avoids complex aqueous quenches of aluminum salts, leads to a significant reduction in wastewater treatment costs and environmental compliance overhead. Consequently, manufacturers can achieve substantial cost savings in pharmaceutical intermediates manufacturing through reduced utility consumption, lower waste disposal fees, and decreased labor hours associated with purification.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like oxalyl chloride and aluminum trichloride directly lowers the bill of materials. Additionally, the telescoping of two reaction steps into one pot reduces energy consumption by removing the need for intermediate isolation, drying, and reactor cleaning cycles. This streamlined workflow translates into a more economically viable production model that maximizes asset utilization and minimizes operational expenditures without compromising on yield or quality.
- Enhanced Supply Chain Reliability: The reliance on stable, commodity-grade starting materials such as 5-bromo-2-chlorobenzoic acid and phenetole ensures a robust supply base that is less susceptible to market volatility compared to specialized acyl chlorides. The robustness of the catalytic system allows for consistent batch-to-batch reproducibility, which is critical for maintaining continuous supply to downstream API manufacturers. This reliability reduces the risk of production delays and ensures that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable strategic goal.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard laboratory equipment that translates well to pilot and commercial plant scales. The reduction in acidic off-gases and heavy metal waste aligns with increasingly strict environmental regulations, facilitating easier permitting and long-term operational sustainability. This eco-friendly profile supports the commercial scale-up of complex pharmaceutical intermediates while adhering to global green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process for stakeholders evaluating this technology for potential adoption or partnership.
Q: What are the primary advantages of this one-pot method over traditional acyl chloride routes?
A: The one-pot method eliminates the need for hazardous acyl chlorination reagents like thionyl chloride and excessive Lewis acids such as aluminum trichloride. This significantly simplifies post-treatment, reduces acidic waste generation, and improves operational safety while maintaining high product purity.
Q: What is the role of alpha-Fe2O3 in this synthesis?
A: Alpha-Fe2O3 acts as a heterogeneous catalyst that facilitates the oxidative coupling reaction. It helps activate the reaction pathway, allowing for milder conditions and better selectivity compared to traditional homogeneous Lewis acid catalysts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It uses readily available raw materials, avoids complex isolation of unstable intermediates, and features a simplified work-up procedure involving standard crystallization, making it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (5-Bromo-2-Chlorophenyl)(4-Ethoxyphenyl)Methanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the competitive landscape of diabetes care therapeutics. Our team of expert chemists has thoroughly analyzed the potential of this one-pot oxidative coupling technology and is fully equipped to translate these laboratory findings into robust commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant with international quality standards. Our state-of-the-art facilities feature rigorous QC labs and stringent purity specifications, guaranteeing that every batch of intermediate we produce meets the exacting requirements necessary for the synthesis of life-saving medications like Dapagliflozin.
We invite forward-thinking pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in process optimization can drive value and security into your supply chain for this essential pharmaceutical building block.
