Advanced Manufacturing of Prohexadione Calcium: A Cost-Effective Agrochemical Intermediate Route
Advanced Manufacturing of Prohexadione Calcium: A Cost-Effective Agrochemical Intermediate Route
The global demand for efficient plant growth regulators continues to drive innovation in agrochemical intermediate manufacturing, specifically for Prohexadione Calcium, a critical compound known chemically as calcium 3-oxido-5-oxo-4-propionyl-cyclohexanecarboxylate. Patent CN100588642C introduces a transformative synthesis methodology that addresses long-standing economic and operational bottlenecks in the production of this high-value active ingredient. By re-engineering the traditional synthetic pathway, this technology replaces costly reagents and hazardous solvents with more sustainable and economically viable alternatives, establishing a new benchmark for industrial scalability. The core innovation lies in the strategic substitution of catalysts and acid-binding agents during the critical acylation and rearrangement phases, which traditionally accounted for a disproportionate share of production expenses. For R&D directors and procurement strategists, understanding this patented route is essential for securing a competitive edge in the supply of high-purity agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prohexadione Calcium, as documented in prior art such as EP 123001, relied heavily on inefficient and cost-prohibitive chemical protocols that hindered large-scale commercialization. The conventional acylation process utilized dichloromethane (DCM) as the primary solvent, necessitating the use of three equivalents of triethylamine as an acid-binding agent, which drastically inflated raw material costs and complicated downstream processing. Furthermore, the subsequent rearrangement step depended on N,N-dimethylaminopyridine (DMAP) at a substantial 25% loading, a catalyst known for its high market price and difficulty in removal, leading to significant impurity profiles in the final product. These legacy methods not only imposed a heavy financial burden due to the consumption of expensive reagents but also created severe environmental compliance challenges associated with the disposal of chlorinated solvents and amine wastes. The cumulative effect of these inefficiencies was a manufacturing process that was fragile, expensive, and difficult to scale without compromising purity or margin.
The Novel Approach
The improved method disclosed in the patent data fundamentally重构 s the synthetic landscape by introducing a robust, cost-optimized pathway that eliminates the reliance on premium-priced catalysts and hazardous solvents. In this novel approach, the acylation reaction is conducted in toluene using inexpensive monovalent or divalent carbonates, such as sodium carbonate, replacing the costly triethylamine and dichloromethane system entirely. This shift not only reduces direct material costs but also simplifies the workup procedure, allowing the crude acylation product to proceed directly to the next stage without rigorous purification. The most significant breakthrough occurs in the rearrangement step, where expensive DMAP is substituted with N-alkyl imidazole derivatives, specifically N-isopropylimidazole, which offers superior catalytic activity at a fraction of the cost. 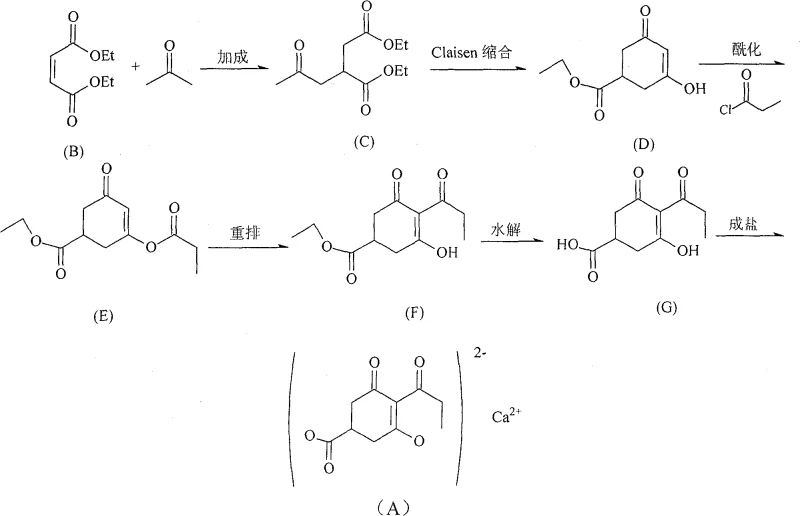 This comprehensive route optimization ensures that the transition from Compound D to the final target Compound A is seamless, efficient, and commercially viable for high-volume production.
This comprehensive route optimization ensures that the transition from Compound D to the final target Compound A is seamless, efficient, and commercially viable for high-volume production.
Mechanistic Insights into Imidazole-Catalyzed Rearrangement
The heart of this technological advancement lies in the mechanistic efficiency of the imidazole-catalyzed rearrangement, which converts the acylated intermediate (Compound E) into the crucial tricarbonyl precursor (Compound F). Unlike the bulky and expensive DMAP molecule, the N-alkyl imidazole catalyst facilitates a smoother nucleophilic attack and subsequent migration of the acyl group, driven by the specific electronic properties of the imidazole ring substituted with C1-C6 alkyl groups. Experimental data indicates that using N-isopropylimidazole in toluene at reflux temperatures promotes high conversion rates while minimizing side reactions that typically plague amine-catalyzed systems. This catalytic system is particularly effective because it maintains stability under the reaction conditions required for the Fries-like rearrangement, ensuring that the delicate cyclohexenone ring structure remains intact while the propionyl group migrates to the thermodynamically favored position. 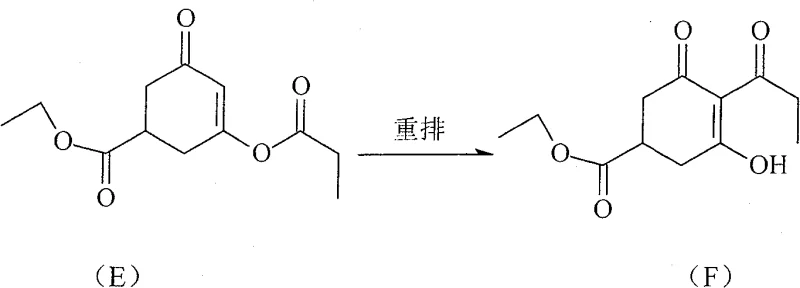 The result is a cleaner reaction profile with fewer byproducts, which directly translates to reduced purification loads in later stages and higher overall yields of the target carboxylic acid intermediate.
The result is a cleaner reaction profile with fewer byproducts, which directly translates to reduced purification loads in later stages and higher overall yields of the target carboxylic acid intermediate.
From an impurity control perspective, the switch to carbonate-based acid binders and imidazole catalysts significantly alters the impurity spectrum, removing complex amine salts that are notoriously difficult to separate from the product matrix. In the conventional process, residual triethylamine and DMAP often formed stable complexes with the acidic intermediates, requiring extensive washing and chromatographic separation that lowered throughput. The new method generates inorganic salts (such as sodium chloride) and neutral organic byproducts that are easily removed via simple aqueous washes or crystallization steps. This purification advantage is critical for meeting the stringent purity specifications required for agrochemical registration, where trace organic impurities can disqualify a batch. By engineering a reaction environment that naturally suppresses the formation of hard-to-remove contaminants, this process ensures that the final Prohexadione Calcium meets rigorous quality standards with minimal downstream intervention.
How to Synthesize Prohexadione Calcium Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly during the high-pressure addition step and the temperature-sensitive rearrangement phase. The process begins with the Michael addition of diethyl maleate and acetone under pressure, followed by a Claisen condensation to form the cyclic core, setting the stage for the critical functionalization steps. Operators must maintain strict temperature controls during the acylation and rearrangement phases to maximize the efficacy of the sodium carbonate and imidazole catalysts respectively. While the general workflow is straightforward, the specific stoichiometry and solvent choices detailed in the patent are vital for reproducing the high yields reported in the examples. For a detailed breakdown of the standardized operating procedures and safety protocols required for execution, please refer to the technical guide below.
- Perform addition reaction between Diethyl Maleate and Acetone using Diethylamine catalyst under pressure to form Compound C.
- Execute Claisen condensation on Compound C using Sodium Ethoxide to yield the cyclic enone Compound D.
- Conduct acylation of Compound D with Propionyl Chloride using Sodium Carbonate as acid binder in Toluene.
- Catalyze the rearrangement of Compound E to Compound F using N-Alkyl Imidazole derivatives.
- Hydrolyze the ester group in Compound F using NaOH followed by acidification to obtain Compound G.
- Form the final calcium salt by reacting Compound G with Calcium Hydroxide in aqueous ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to drastically reduce the total cost of ownership for Prohexadione Calcium manufacturing. By eliminating the dependency on volatile, high-cost reagents like DMAP and dichloromethane, the process insulates the supply chain from price fluctuations associated with specialty amines and chlorinated solvents. The simplification of the workup procedures, specifically the ability to use crude intermediates without purification, translates directly into reduced cycle times and lower labor costs per kilogram of output. Furthermore, the use of common industrial solvents like toluene and ethanol enhances supply security, as these materials are readily available globally, unlike some specialized reagents that may face logistical bottlenecks. This robustness ensures a more reliable supply of high-purity agrochemical intermediates, mitigating the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The replacement of expensive DMAP catalysts and excess triethylamine with low-cost imidazole derivatives and sodium carbonates results in substantial raw material savings. Additionally, the elimination of dichloromethane reduces solvent recovery and disposal costs, while the ability to skip purification steps for intermediate compounds lowers energy consumption and processing time. These cumulative efficiencies drive down the unit cost of production significantly without compromising the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified by relying on commodity chemicals such as toluene, sodium carbonate, and calcium hydroxide, which have stable global supply chains compared to specialty catalysts. The reduced complexity of the synthesis route minimizes the number of critical control points, thereby decreasing the likelihood of batch failures and ensuring consistent delivery schedules for downstream formulators. This reliability is crucial for maintaining continuous production lines in the competitive agrochemical sector.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations capable of handling reflux conditions and aqueous workups without specialized equipment. From an environmental standpoint, the removal of chlorinated solvents and the reduction of amine waste streamline wastewater treatment processes, facilitating easier compliance with increasingly strict environmental regulations. This green chemistry approach not only reduces regulatory risk but also aligns with corporate sustainability goals, enhancing the marketability of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved synthesis method for Prohexadione Calcium. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for commercial manufacturing. The answers below highlight the specific operational benefits and chemical advantages that define this innovative approach.
Q: How does the new imidazole catalyst reduce production costs compared to DMAP?
A: The patented method replaces expensive DMAP (25% loading) with cheaper N-alkyl imidazole derivatives, significantly lowering raw material costs while maintaining high catalytic efficiency for the rearrangement step.
Q: What are the environmental benefits of switching from Dichloromethane to Toluene?
A: Replacing Dichloromethane (DCM) with Toluene eliminates the use of a chlorinated solvent, simplifying waste treatment and reducing environmental compliance burdens associated with halogenated waste disposal.
Q: Can the intermediate products be used without purification in this process?
A: Yes, the optimized acylation step produces Compound E with sufficient purity to be directly used in the subsequent rearrangement reaction without intermediate column chromatography, streamlining the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prohexadione Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes in the agrochemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the imidazole-catalyzed Prohexadione Calcium process are executed with precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for global agrochemical registration. We are committed to leveraging advanced technologies, such as the one described in CN100588642C, to deliver high-purity agrochemical intermediates that empower our clients to bring effective plant growth regulators to market faster.
We invite procurement leaders and R&D innovators to collaborate with us to explore how this optimized synthesis route can enhance your supply chain resilience and cost structure. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can serve as a reliable foundation for your long-term success in the plant growth regulator sector.
