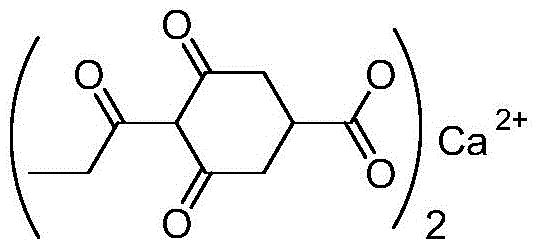
Revolutionizing Plant Growth Regulator Production: A Solvent-Free Catalytic Route for Prohexadione Calcium Intermediates
Revolutionizing Plant Growth Regulator Production: A Solvent-Free Catalytic Route for Prohexadione Calcium Intermediates
The global demand for efficient plant growth regulators continues to surge, driven by the need for high-yield agriculture and crop protection. At the forefront of this sector is Prohexadione Calcium, a potent cyclohexenone-based regulator known for enhancing crop lodging resistance and stress tolerance. However, the manufacturing of its key intermediate, ethyl 3,5-dioxo-4-propionylcyclohexanecarboxylate, has historically been plagued by environmental hazards and complex purification steps. A groundbreaking technical disclosure found in patent CN111187163A introduces a paradigm shift in this synthesis, replacing toxic solvents and harsh reagents with a green, solvent-free Lewis acid catalytic system. This innovation not only addresses critical environmental compliance issues but also offers a robust pathway for cost-effective, large-scale production. By leveraging propionic anhydride as both a reagent and a reaction medium, this method eliminates the need for volatile organic compounds (VOCs) entirely. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic and commercial implications of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
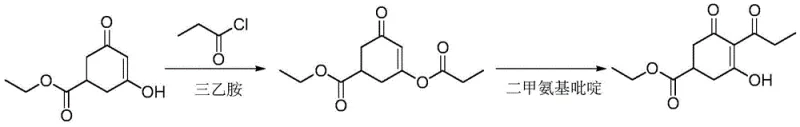
The Limitations of Conventional Methods
Historically, the industrial synthesis of prohexadione calcium intermediates has relied on a multi-step sequence involving hazardous reagents and extensive solvent usage. As detailed in prior art literature, the conventional route typically begins with the acylation of 3-hydroxy-5-ethoxycarbonyl-cyclohex-2-en-1-one using propionyl chloride in the presence of triethylamine. This step generates significant amounts of triethylamine hydrochloride salt, necessitating filtration and washing procedures that complicate the workflow. Furthermore, the subsequent rearrangement step requires the use of 4-Dimethylaminopyridine (DMAP) and solvents such as dichloroethane or toluene under reflux conditions. The reliance on chlorinated solvents poses severe environmental and safety risks, including toxicity to operators and high costs associated with solvent recovery and waste treatment. Moreover, the crude product from these traditional methods often contains difficult-to-remove impurities, forcing manufacturers to employ column chromatography for purification. This reliance on chromatographic separation is a major bottleneck for commercial scale-up of complex agrochemical intermediates, as it drastically reduces throughput and increases operational expenditures, making the process economically unviable for multi-ton production.
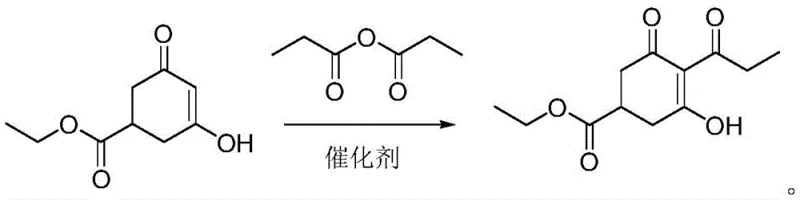
The Novel Approach
In stark contrast to the legacy processes, the methodology disclosed in CN111187163A presents a streamlined, one-pot strategy that fundamentally redesigns the reaction landscape. Instead of using reactive acid chlorides and amine bases, this novel approach utilizes propionic anhydride directly as the acylating agent in the presence of a catalytic amount of a Lewis acid. This substitution is transformative because propionic anhydride serves a dual role: it acts as the reactant and effectively as the solvent, thereby eliminating the need for external organic solvents like toluene. The reaction proceeds smoothly at mild temperatures ranging from 40°C to 50°C, which significantly lowers energy consumption compared to the high-temperature reflux required in traditional methods. By removing the generation of amine salts and avoiding chlorinated solvents, the post-reaction workup is drastically simplified to a basic water wash and crystallization. This shift not only enhances the safety profile of the manufacturing facility but also aligns perfectly with modern green chemistry principles, offering a compelling solution for cost reduction in plant growth regulator manufacturing through simplified unit operations.
Mechanistic Insights into Lewis Acid-Catalyzed Acylation
The core of this technological breakthrough lies in the precise selection and application of Lewis acid catalysts, specifically Aluminum Chloride (AlCl3), Ferric Chloride (FeCl3), or Titanium Tetrachloride (TiCl4). Unlike traditional Brønsted acid catalysts or base-mediated systems, these Lewis acids facilitate the activation of the carbonyl group in propionic anhydride with exceptional efficiency. The mechanism involves the coordination of the Lewis acid to the oxygen atom of the anhydride, increasing the electrophilicity of the carbonyl carbon. This activated species then undergoes nucleophilic attack by the enol form of the 3-hydroxy-5-ethoxycarbonyl-cyclohex-2-en-1-one substrate. What is particularly remarkable is the catalyst loading; the patent specifies a molar ratio of substrate to catalyst between 1:0.001 and 1:0.005. This ultra-low loading indicates a highly active catalytic cycle where the metal center is rapidly regenerated, minimizing metal contamination in the final product. For R&D teams focused on impurity profiles, this is critical, as it reduces the burden of heavy metal removal downstream. The reaction environment, being essentially neat (solvent-free), ensures high local concentrations of reactants, driving the equilibrium towards the desired 3,5-dioxo-4-propionyl product with high selectivity.
Furthermore, the mechanism inherently suppresses the formation of side products that typically plague the traditional triethylamine route. In the conventional method, the basic conditions can lead to polymerization or degradation of the sensitive cyclohexenone ring. The mild, slightly acidic conditions provided by the Lewis acid catalyst preserve the structural integrity of the ring system while promoting the necessary acylation and subsequent tautomerization to the stable triketo form. The absence of strong bases prevents the formation of unwanted salts and emulsions during the workup phase. Instead, upon completion of the reaction, the addition of water serves a dual purpose: it quenches any remaining anhydride to form propionic acid (which is easily recovered) and dissolves the metal catalyst salts into the aqueous phase. This clean phase separation allows the organic product to be isolated simply by cooling and crystallization. The result is a product with HPLC purity exceeding 96%, and in optimized examples up to 98.7%, demonstrating that this mechanistic approach offers superior control over the high-purity agrochemical intermediate specification required by top-tier formulators.
How to Synthesize Ethyl 3,5-dioxo-4-propionylcyclohexanecarboxylate Efficiently
Implementing this synthesis in a pilot or production setting requires careful attention to the stoichiometry and thermal management of the exothermic acylation. The process begins by charging the reactor with the starting ketone and propionic anhydride in a molar ratio ranging from 1:1 to 1:3, ensuring an excess of the anhydride to drive the reaction to completion. Following the addition of the chosen Lewis acid catalyst, the mixture is heated to a moderate temperature window of 40°C to 50°C. Maintaining this temperature for a duration of 10 to 24 hours allows the reaction to reach full conversion without risking thermal degradation of the product. Once the reaction is complete, the unreacted propionic anhydride and the generated propionic acid are recovered via reduced pressure distillation, a step that not only purifies the crude mass but also recycles valuable raw materials back into the process. The detailed standardized operating procedure for this synthesis, including specific quenching and crystallization parameters, is outlined below.
- Charge 3-hydroxy-5-ethoxycarbonyl-cyclohex-2-en-1-one and propionic anhydride (molar ratio 1: 1 to 1:3) into a reactor.
- Add a Lewis acid catalyst (AlCl3, FeCl3, or TiCl4) at a molar ratio of 1: 0.001 to 0.005 relative to the substrate.
- Heat the mixture to 40-50°C for 10-24 hours, then recover excess anhydride under reduced pressure.
- Wash the residue with water to remove the catalyst, separate layers, and freeze-crystallize the organic phase at 0-5°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solvent-free technology represents a significant opportunity to optimize the total cost of ownership for prohexadione calcium intermediates. The elimination of organic solvents such as toluene and dichloroethane removes a major variable cost component, as these solvents are subject to volatile market pricing and stringent transportation regulations. Moreover, the absence of solvents drastically reduces the volume of waste streams requiring treatment, leading to substantial savings in environmental compliance and disposal fees. The ability to recover and reuse propionic anhydride further enhances the atom economy of the process, ensuring that raw material costs are kept to a minimum. From a logistical standpoint, the simplified workflow reduces the number of unit operations, which translates to shorter batch cycles and increased facility throughput. This efficiency is crucial for reducing lead time for high-purity agrochemical intermediates, allowing suppliers to respond more agilely to seasonal demand fluctuations in the agricultural sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive solvent purchase and recovery infrastructure. Traditional methods require large volumes of solvents for reaction and purification, along with the energy-intensive distillation columns needed to recycle them. By operating in a neat system, the capital expenditure for reactor volume is optimized, and operational energy costs are significantly lowered due to the mild reaction temperatures (40-50°C). Additionally, the avoidance of column chromatography, which is prohibitively expensive at scale, is replaced by simple crystallization, a unit operation that is both cheap and highly scalable. These factors combine to create a manufacturing process with a markedly lower cost basis, providing a competitive edge in pricing strategies.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by the dependency on specialized or hazardous reagents. The conventional route relies on propionyl chloride and triethylamine, which are corrosive and require careful handling and storage. The new method utilizes propionic anhydride, a more stable and widely available commodity chemical. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to batch failures caused by moisture sensitivity or temperature excursions. This reliability ensures a consistent flow of material, mitigating the risk of stockouts that can disrupt the production schedules of downstream formulators. The simplified purification also means that quality control testing is faster, accelerating the release of batches into the supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges, particularly when solvents are involved. The solvent-free nature of this invention simplifies scale-up, as the heat capacity of the reaction mass is higher, allowing for better temperature control in large reactors. From an environmental perspective, the process aligns with increasingly strict global regulations regarding VOC emissions and hazardous waste. By generating minimal waste and avoiding chlorinated solvents, manufacturers can maintain their social license to operate and avoid potential regulatory fines. This sustainability profile is increasingly becoming a prerequisite for partnerships with major multinational agrochemical companies who prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN111187163A, providing a factual basis for evaluating the technology's viability. Understanding these details is vital for technical teams assessing the feasibility of adopting this greener manufacturing standard.
Q: What are the primary advantages of the solvent-free method described in CN111187163A?
A: The primary advantage is the elimination of hazardous organic solvents like toluene and dichloroethane, which significantly reduces environmental impact and waste disposal costs. Additionally, the process avoids complex column chromatography purification, relying instead on simple water washing and crystallization to achieve high purity (>96%).
Q: Which catalysts are suitable for this acylation reaction?
A: The patent specifies the use of Lewis acids such as Aluminum Chloride (AlCl3), Ferric Chloride (FeCl3), or Titanium Tetrachloride (TiCl4). These catalysts are effective at very low loadings (0.1% to 0.5% molar ratio), making the process economically viable.
Q: How does this new route compare to traditional methods in terms of yield?
A: Traditional methods involving propionyl chloride and triethylamine often require multi-step purification and yield around 80%. The novel solvent-free route described achieves yields exceeding 90%, with HPLC purity reaching up to 98.7%, demonstrating superior efficiency and product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 3,5-dioxo-4-propionylcyclohexanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies is key to maintaining competitiveness in the global agrochemical market. Our technical team has extensively analyzed the solvent-free Lewis acid catalysis route described in CN111187163A and possesses the expertise to implement this technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for plant growth regulator intermediates, guaranteeing that every batch meets the highest international standards for potency and impurity profiles.
We invite procurement leaders and R&D directors to collaborate with us to leverage this innovative chemistry for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this green synthesis route for your operations. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your production volume requirements. Let us help you secure a sustainable and cost-effective supply of prohexadione calcium intermediates.
