Advanced Purification Technology for High-Purity Prohexadione Calcium Manufacturing
Advanced Purification Technology for High-Purity Prohexadione Calcium Manufacturing
The global demand for efficient and environmentally benign plant growth regulators has driven significant innovation in the synthesis of cyclohexanedione derivatives, specifically Prohexadione Calcium. As detailed in the recent patent literature, specifically CN111087294A, a breakthrough preparation method has been disclosed that addresses the longstanding challenges of purity and scalability in producing this critical agrochemical active ingredient. This novel approach leverages a sophisticated precipitation purification technique that transforms the crude rearrangement product of 3,5-dioxo-4-propionyl cyclohexanecarboxylic acid ethyl ester into a highly purified sodium salt intermediate, thereby bypassing the need for expensive and labor-intensive column chromatography. The structural integrity and functional groups of the target molecule, as illustrated below, are preserved through mild reaction conditions that favor industrial applicability while ensuring the final product meets stringent quality specifications for agricultural use.
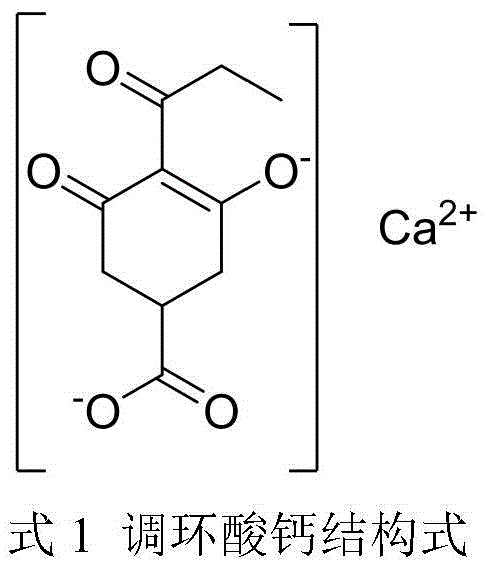
Prohexadione Calcium acts as a potent inhibitor of gibberellin biosynthesis, effectively controlling vegetative growth in cereal crops and enhancing their resistance to lodging and environmental stress. Unlike triazole-based retardants, this compound degrades rapidly into carbon dioxide and water in soil environments, presenting a superior ecological profile that aligns with modern sustainable agriculture mandates. The ability to manufacture this compound with high purity is paramount, as impurities can affect both the biological efficacy and the environmental safety profile of the final formulation. The patented process described herein offers a robust pathway to achieve these high purity standards through a sequence of salt formation, selective precipitation, and controlled hydrolysis, representing a significant leap forward in the manufacturing technology of high-value agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Prohexadione Calcium, such as those referenced in prior art like CN104140368A, often involve a multi-step sequence starting from diethyl maleate, proceeding through Michael addition, Claisen condensation, and acylation. While chemically feasible, these conventional pathways frequently result in crude products laden with complex by-product profiles that are difficult to separate. The reliance on column chromatography for purification in laboratory-scale syntheses creates a severe bottleneck when attempting to scale up to commercial production levels, as chromatographic separation is inherently batch-limited, solvent-intensive, and costly. Furthermore, the presence of residual impurities from incomplete reactions or side-products can compromise the stability of the final calcium salt, leading to inconsistent performance in field applications and potential regulatory hurdles regarding residue limits in food crops.
The Novel Approach
The innovative methodology presented in patent CN111087294A circumvents these bottlenecks by introducing a targeted purification step at the sodium salt stage of the synthesis. By reacting the crude rearrangement product with sodium hydride in specific organic solvents such as methanol or acetone, the process induces the formation of a rearrangement product sodium salt that exhibits low solubility in the reaction medium. This physicochemical property is exploited to drive the precipitation of the desired intermediate while leaving soluble impurities in the supernatant, which are subsequently removed via simple suction filtration. This strategic shift from chromatographic separation to crystallization-based purification drastically simplifies the workflow, reduces solvent consumption, and enhances the overall throughput of the manufacturing process, making it ideally suited for large-scale industrial implementation without compromising on the purity of the final Prohexadione Calcium product.
Mechanistic Insights into Sodium Salt Precipitation and Hydrolysis
The core of this technological advancement lies in the precise control of the acid-base chemistry governing the formation of the intermediate sodium salts. The process initiates with the deprotonation of the enolic hydroxyl group on the cyclohexanedione ring of the crude ester by sodium hydride, generating a resonance-stabilized sodium enolate. As depicted in the reaction scheme below, this sodium salt (Formula I) possesses distinct solubility characteristics in polar aprotic or protic solvents like acetone and methanol, causing it to crash out of the solution as a solid beige precipitate. This precipitation event serves as a powerful purification mechanism, effectively segregating the target molecular scaffold from non-ionic organic impurities and unreacted starting materials that remain dissolved in the mother liquor, thus achieving a high degree of chemical enrichment prior to the final conversion steps.
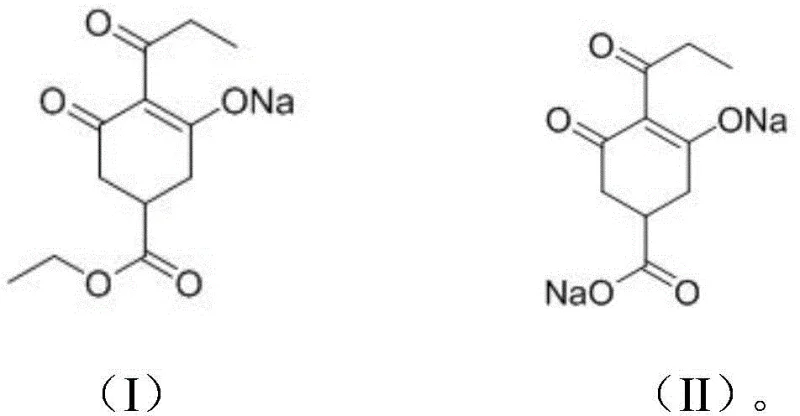
Following the isolation of the purified sodium salt, the mechanism proceeds through an alkaline hydrolysis step where the ethyl ester moiety is cleaved under controlled thermal conditions (50-70°C) in the presence of aqueous sodium hydroxide. This hydrolysis converts the ester into the corresponding carboxylate sodium salt (Formula II), which is then carefully acidified to a pH of 7.5-8.5 to generate the free acid form without inducing decomposition of the sensitive beta-diketone system. The final step involves the metathesis reaction with a calcium salt, such as calcium chloride, where the calcium ion coordinates with the enolate and carboxylate oxygens to form the stable, insoluble Prohexadione Calcium complex. This sequence ensures that the stereochemical and structural integrity of the molecule is maintained throughout the transformation, resulting in a product with consistent biological activity and minimal impurity burden.
How to Synthesize Prohexadione Calcium Efficiently
The synthesis of high-purity Prohexadione Calcium via this patented route requires careful attention to solvent selection and stoichiometric ratios to maximize the efficiency of the precipitation purification. The process begins with the preparation of a sodium hydride solution in an organic solvent, followed by the controlled addition of the crude rearrangement product to induce the formation of the insoluble sodium salt intermediate. Detailed operational parameters, including specific molar ratios of 1.0:1.1 for the ester to sodium hydride and precise temperature controls during hydrolysis, are critical for reproducibility. For a comprehensive, step-by-step guide on executing this synthesis with optimal yield and purity, please refer to the standardized protocol outlined below.
- Dissolve sodium hydride in an organic solvent (methanol or acetone) to prepare a reactive liquid base, then add the crude rearrangement product of 3,5-dioxo-4-propionyl cyclohexanecarboxylic acid ethyl ester to the solvent system.
- React the mixture to form the purified rearrangement product sodium salt (Formula I), which precipitates out of the solution, allowing for the removal of insoluble impurities via suction filtration.
- Hydrolyze the filtered sodium salt under alkaline conditions to generate the cycloregulated acid sodium salt (Formula II), followed by acidification and reaction with a calcium salt to yield the final Prohexadione Calcium product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic advantages by fundamentally altering the cost structure and operational reliability of Prohexadione Calcium production. The elimination of column chromatography, a unit operation known for its high capital expenditure and slow throughput, translates directly into reduced processing times and lower utility costs per kilogram of product. Furthermore, the use of common, commodity-grade solvents like methanol and acetone, combined with the simplicity of filtration-based purification, mitigates supply chain risks associated with specialized reagents or complex separation media, ensuring a more resilient and continuous manufacturing operation capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the replacement of expensive chromatographic purification with a low-cost precipitation and filtration sequence. By removing the need for silica gel columns and large volumes of elution solvents, the process significantly lowers the variable costs associated with raw materials and waste disposal. Additionally, the simplified workflow reduces labor hours and equipment occupancy time, allowing for higher asset utilization rates and a more favorable cost-of-goods-sold profile that enhances competitiveness in the global agrochemical market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which operate effectively at room temperature for the salt formation and moderate temperatures for hydrolysis, reduces the risk of batch failures due to thermal runaway or sensitive catalyst deactivation. The reliance on widely available reagents such as sodium hydride and calcium chloride ensures that raw material sourcing remains stable and unaffected by niche supply shortages. This operational stability allows manufacturers to maintain consistent inventory levels and reliable delivery schedules, which is critical for downstream formulators who depend on just-in-time supply chains for seasonal agricultural applications.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently greener due to the significant reduction in organic solvent waste generated by chromatographic processes. The ability to perform the key purification step via simple filtration makes the technology highly scalable from pilot plants to multi-tonne commercial reactors without the engineering complexities associated with scaling up chromatography columns. This ease of scale-up, combined with the rapid biodegradability of the final product in soil, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals regarding green chemistry and waste minimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical implications of adopting this technology for commercial production. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities.
Q: How does this novel method improve the purity of Prohexadione Calcium compared to traditional routes?
A: The method utilizes a unique precipitation purification strategy where the intermediate sodium salt becomes insoluble in specific organic solvents like methanol or acetone. This physical property allows impurities to remain in the solution while the pure intermediate precipitates, eliminating the need for complex column chromatography separation.
Q: Is column chromatography required for the purification of the rearrangement product?
A: No, one of the primary advantages of this patented process (CN111087294A) is that it completely avoids the need for column chromatography. The purification is achieved through controlled precipitation and simple suction filtration, which significantly lowers operational complexity and cost.
Q: What are the optimal reaction conditions for the hydrolysis step?
A: The hydrolysis of the rearrangement product sodium salt is conducted in an aqueous NaOH solution with a mass concentration of 10-20%. The reaction is typically stirred at a temperature range of 50-70°C for 1 to 3 hours to ensure complete conversion to the cycloregulated acid sodium salt before acidification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prohexadione Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation crop protection solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Prohexadione Calcium we supply meets the exacting standards required for regulatory approval and field efficacy.
We invite you to collaborate with us to leverage this advanced purification technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and reliability for your agrochemical portfolio.
