Scalable Synthesis of Chiral Beta-Amino Acid Esters via Iridium Catalysis for Global Pharma Supply Chains
The pharmaceutical industry continuously demands more efficient routes to chiral building blocks, particularly for the synthesis of carbapenem antibiotics and other bioactive compounds. Patent CN113354554B introduces a groundbreaking methodology for the preparation of (2R,3S)-β′-hydroxy-β-amino acid ester derivatives, utilizing a highly selective Iridium-catalyzed asymmetric hydrogenation strategy. This technology addresses critical bottlenecks in the production of key pharmaceutical intermediates by offering a pathway that combines exceptional stereocontrol with operational simplicity. The process operates under moderate temperatures ranging from 10°C to 100°C and hydrogen pressures between 1.0 MPa and 10.0 MPa, achieving reaction yields as high as 95%. For global supply chain leaders, this represents a significant opportunity to secure a reliable source of high-purity intermediates that meet stringent regulatory standards for impurity profiles and optical purity.
The significance of this patent lies in its ability to produce versatile precursors for unnatural peptides and β-lactam rings, which are foundational structures in modern medicinal chemistry. By leveraging a specific chiral ligand system coupled with an Iridium metal complex, the method ensures consistent quality across batches, a paramount concern for procurement managers overseeing long-term API production contracts. The robustness of the catalytic system allows for flexibility in substrate scope, accommodating various substituted aryl and alkyl groups without compromising selectivity. This adaptability is crucial for pharmaceutical companies looking to diversify their pipeline while maintaining a streamlined manufacturing footprint. Furthermore, the method's compatibility with standard industrial solvents facilitates easier technology transfer from laboratory scale to multi-ton commercial production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β′-hydroxy-β-amino acid ester derivatives has relied heavily on Ruthenium-catalyzed asymmetric hydrogenation or biocatalytic approaches, both of which present distinct challenges for large-scale manufacturing. As illustrated in prior art such as the work by Rimoldi et al., conventional Ruthenium systems often necessitate prolonged reaction times exceeding 60 hours to achieve acceptable conversion, which severely impacts throughput and increases energy consumption. 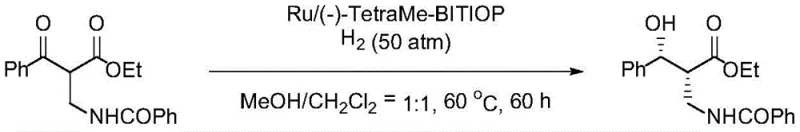 Additionally, alternative methods reported by Zhang et al. using asymmetric hydrogen transfer reactions have demonstrated enantioselectivity around 95.4% ee and diastereoselectivity of 97.8% de, figures that fall short of the rigorous purity requirements for next-generation antibiotic synthesis. Biocatalytic routes, while environmentally friendly, often suffer from lower diastereoselectivity, with some reports indicating de values as low as 70%, necessitating costly and yield-reducing recrystallization steps. These inefficiencies translate directly into higher cost of goods sold (COGS) and extended lead times, creating vulnerabilities in the supply chain for critical medical ingredients.
Additionally, alternative methods reported by Zhang et al. using asymmetric hydrogen transfer reactions have demonstrated enantioselectivity around 95.4% ee and diastereoselectivity of 97.8% de, figures that fall short of the rigorous purity requirements for next-generation antibiotic synthesis. Biocatalytic routes, while environmentally friendly, often suffer from lower diastereoselectivity, with some reports indicating de values as low as 70%, necessitating costly and yield-reducing recrystallization steps. These inefficiencies translate directly into higher cost of goods sold (COGS) and extended lead times, creating vulnerabilities in the supply chain for critical medical ingredients.
The Novel Approach
In stark contrast, the novel approach detailed in CN113354554B utilizes a tailored Iridium catalyst system that drastically accelerates the reaction kinetics while enhancing stereochemical outcomes. The new methodology reduces reaction times to a window of 2 to 24 hours, representing a substantial improvement in process efficiency compared to the multi-day cycles of legacy technologies. By employing a specific chiral ligand (III) in conjunction with Iridium complexes such as [Ir(COD)Cl]2, the process achieves enantioselectivity up to 99% ee and diastereoselectivity up to 99:1 dr. This leap in performance eliminates the need for extensive downstream purification to remove unwanted isomers, thereby simplifying the overall workflow. The use of readily available bases like sodium acetate or potassium tert-butoxide further enhances the practicality of the method, allowing for cost reduction in pharmaceutical intermediate manufacturing by minimizing reagent costs and waste generation.
Mechanistic Insights into Ir-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise interaction between the Iridium metal center and the chiral ferrocenyl phosphine-sulfonamide ligands. The catalytic cycle begins with the formation of an active Iridium-hydride species upon exposure to hydrogen gas, which then coordinates with the β′-carbonyl-β-amino acid ester substrate. The steric bulk and electronic properties of the chiral ligand create a highly defined pocket that directs the facial selectivity of the hydrogen addition, ensuring the exclusive formation of the (2R,3S) configuration. 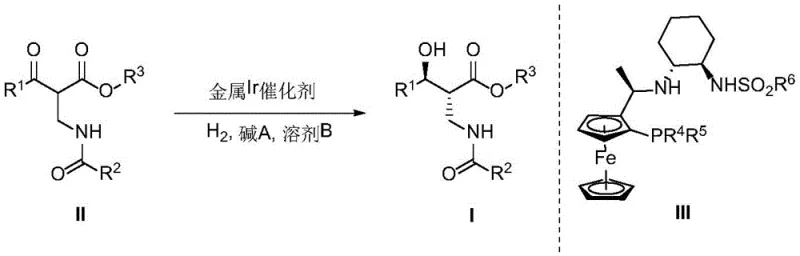 This level of control is critical for R&D directors who must guarantee that every batch of intermediate possesses the correct chirality to prevent the formation of toxic or inactive diastereomers in the final drug product. The mechanism also benefits from the electron-rich nature of the ferrocene backbone, which stabilizes the metal center against decomposition under the elevated pressures required for industrial hydrogenation. Understanding this mechanistic nuance allows process chemists to fine-tune reaction parameters, such as temperature and pressure, to maximize turnover numbers and catalyst longevity.
This level of control is critical for R&D directors who must guarantee that every batch of intermediate possesses the correct chirality to prevent the formation of toxic or inactive diastereomers in the final drug product. The mechanism also benefits from the electron-rich nature of the ferrocene backbone, which stabilizes the metal center against decomposition under the elevated pressures required for industrial hydrogenation. Understanding this mechanistic nuance allows process chemists to fine-tune reaction parameters, such as temperature and pressure, to maximize turnover numbers and catalyst longevity.
Furthermore, the choice of solvent and base plays a pivotal role in modulating the reaction environment to favor the desired transition state. The patent specifies the use of solvents like toluene, ethyl acetate, or tetrahydrofuran, which provide optimal solubility for both the organic substrate and the organometallic catalyst. The addition of weak bases such as sodium acetate helps to neutralize any acidic byproducts that could potentially poison the catalyst or promote racemization of the product. This careful balancing of reaction components ensures that the impurity profile remains exceptionally clean, with minimal formation of over-reduced side products or regioisomers. For quality assurance teams, this means that the resulting high-purity pharmaceutical intermediates consistently meet the strict specifications required for regulatory filings, reducing the risk of batch rejection and ensuring supply continuity.
How to Synthesize (2R,3S)-β′-hydroxy-β-amino acid ester Efficiently
Implementing this synthesis route requires a systematic approach to catalyst preparation and reaction management to fully realize its efficiency benefits. The process begins with the in situ generation of the active catalyst by mixing the Iridium precursor with the chiral ligand in a dry solvent under an inert atmosphere, ensuring that moisture and oxygen do not deactivate the sensitive metal complex. Once the catalyst solution is prepared, it is transferred to a high-pressure reactor containing the substrate, additional solvent, and the appropriate base. This standardized protocol allows for reproducible results across different scales, from gram-level optimization to multi-kilogram production runs, providing a clear roadmap for process engineers.
- Prepare the metal Ir catalyst solution by reacting metal Ir complex with chiral ligand (III) in solvent A under argon at 0-60°C for 0.5-6 hours.
- Load the β′-carbonyl-β-amino acid ester derivative, prepared catalyst, solvent B, and base A into a high-pressure reactor under nitrogen protection.
- Conduct asymmetric hydrogenation at 10-100°C and 1.0-10.0 MPa H2 pressure for 2-24 hours, followed by concentration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Ir-catalyzed technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The drastic reduction in reaction time from days to hours significantly enhances asset utilization, allowing manufacturing facilities to produce more batches within the same timeframe without requiring additional capital investment in reactors. This increased throughput capability directly contributes to reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations and emergency orders. Moreover, the high selectivity of the process minimizes the consumption of raw materials and the generation of chemical waste, aligning with corporate sustainability goals and reducing disposal costs associated with hazardous byproducts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like Ruthenium in favor of more efficient Iridium systems, combined with the removal of lengthy purification steps, leads to substantial cost savings. By achieving near-quantitative conversion and high selectivity, the process reduces the loss of valuable starting materials and lowers the overall cost per kilogram of the final intermediate. The use of common industrial solvents and bases further drives down operational expenses, making the commercial scale-up of complex chiral intermediates economically viable even in competitive markets.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent performance across multiple batches, mitigating the risk of production delays caused by failed reactions or off-spec material. This reliability is essential for maintaining a steady flow of materials to downstream API manufacturers, preventing bottlenecks that could disrupt the entire drug production schedule. Additionally, the scalability of the method means that suppliers can easily ramp up production volumes to meet surging demand without compromising on quality, providing a secure and dependable source for critical healthcare ingredients.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, featuring high atom economy and minimal waste generation, which simplifies compliance with increasingly stringent environmental regulations. The ability to operate at moderate temperatures and pressures reduces energy consumption, contributing to a lower carbon footprint for the manufacturing site. These environmental advantages not only reduce regulatory burdens but also enhance the brand reputation of the supplier as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis method, we have compiled answers based on the specific data and claims found within the patent documentation. These insights are intended to clarify the operational parameters and expected outcomes for partners considering this technology for their supply networks. Understanding these details helps stakeholders make informed decisions about integrating this advanced intermediate into their production workflows.
Q: What are the stereochemical outcomes of this Ir-catalyzed hydrogenation method?
A: The method achieves exceptional stereoselectivity, with enantioselectivity up to 99% ee and diastereoselectivity up to 99:1 dr, ensuring high optical purity for downstream API synthesis.
Q: How does this process compare to conventional Ruthenium-catalyzed methods?
A: Unlike prior art Ru-catalyzed methods that often require over 60 hours, this Ir-catalyzed process significantly reduces reaction time to 2-24 hours while improving yield to 95% and simplifying purification.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and common solvents like toluene or ethyl acetate, featuring high atomic economy and low green pollution, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,3S)-β′-hydroxy-β-amino acid ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality chiral intermediates for the development of life-saving medications. Our team of expert process chemists has extensively evaluated the technology described in CN113354554B and possesses the technical capability to implement this Ir-catalyzed hydrogenation route at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every shipment of (2R,3S)-β′-hydroxy-β-amino acid ester derivatives meets the highest industry standards for optical purity and chemical integrity.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in advanced catalytic processes can become a strategic advantage for your organization.
