Advanced Electrodialysis Purification for High-Purity Cilastatin Manufacturing
The pharmaceutical industry continuously seeks robust purification technologies that balance high yield with stringent purity requirements, particularly for critical antibiotic adjuvants like cilastatin. Patent CN102675175B introduces a transformative approach to this challenge by leveraging electrodialysis technology for the separation and purification of cilastatin. This method addresses the longstanding issue of removing inorganic salts, specifically sodium chloride, generated during the synthesis of cilastatin from its precursors. Unlike traditional resin-based methods that suffer from complex regeneration cycles and potential product degradation, this electrodialytic process offers a streamlined pathway to high-purity products with low ignition residue. For R&D directors and process engineers, this represents a significant leap forward in downstream processing efficiency, ensuring that the final active pharmaceutical ingredient (API) intermediate meets the rigorous quality standards required for combination therapies with carbapenem antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of cilastatin has been plagued by the difficulties associated with removing the substantial quantities of inorganic salts produced during the neutralization and reaction steps. Conventional techniques often rely heavily on ion exchange resins, such as Zeo-karb, which necessitate elution with ammonia water. This approach inherently converts the product into an ammonium salt form, requiring a subsequent acidification step with hydrochloric acid to recover the free acid form of cilastatin. Unfortunately, this secondary acidification regenerates inorganic ammonium chloride salts, creating a cyclical purification problem that complicates the workflow. Furthermore, alternative methods utilizing macroporous adsorbent resins, while reducing some exothermic risks, introduce significant operational complexity and high material costs due to the expense of the polymeric adsorbents. These traditional pathways are often tedious, difficult to control on a large industrial scale, and prone to yielding products with unacceptable levels of ignition residue, thereby failing to meet the strict specifications for parenteral formulations.
The Novel Approach
The methodology disclosed in CN102675175B circumvents these pitfalls by integrating electrodialysis directly into the purification workflow following the initial synthesis reaction. The process begins with the reaction of Compound (V), a chloro-heptenoic acid derivative, with cysteine hydrochloride in an alkaline aqueous solution, typically using sodium hydroxide. This reaction generates the crude cilastatin along with significant amounts of sodium chloride as a byproduct. Instead of resorting to resin columns or complex solvent extractions that suffer from low partition coefficients, the novel approach utilizes an electric field to drive the migration of ions through selective membranes.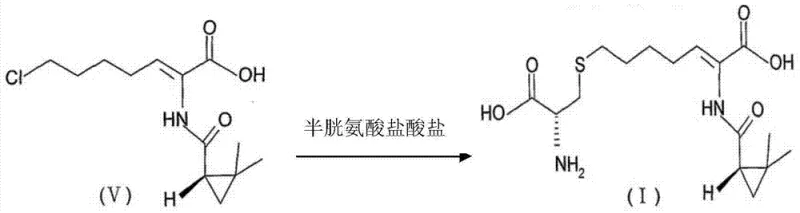
By applying a direct current voltage between 10 and 100V, sodium cations and chloride anions are selectively transported across ion-exchange membranes, effectively desalting the solution while the larger, zwitterionic cilastatin molecules are retained. This physical separation mechanism avoids the chemical transformations and equilibrium limitations associated with resin binding, resulting in a much simpler operation that is highly amenable to continuous industrial production. The result is a purified solution ready for concentration and crystallization, boasting significantly improved yield and purity profiles compared to the prior art.
Mechanistic Insights into Electrodialytic Desalting
The core innovation lies in the application of electrodialysis, a membrane separation process that utilizes electrical potential to drive the transport of ions. In this specific application, the apparatus comprises anode and cathode compartments separated by ion-exchange membranes that exhibit selectivity based on charge. The anode membrane permits the passage of positively charged ions (cations), while the cathode membrane allows negatively charged ions (anions) to pass. When the crude reaction mixture containing cilastatin and sodium chloride is subjected to a DC electric field, the sodium ions migrate towards the cathode and the chloride ions migrate towards the anode. Crucially, cilastatin, being a larger organic molecule with complex stereochemistry ([R-[R*, S* (Z)]]-7-[(2-amino-2-carboxy ethyl) sulphur]-2-[[(2,2-dimethyl cyclopropyl) carbonyl] amino]-2-heptenoic acid), does not readily permeate these membranes under the operating conditions. This size and charge-based selectivity ensures that the product remains in the diluate compartment while the salt impurities are removed into the concentrate compartments.
From an impurity control perspective, this mechanism is superior because it targets the root cause of high ignition residue—inorganic salts—without introducing new organic impurities or degrading the sensitive beta-lactam-stabilizing structure of cilastatin. The process is monitored by measuring the specific conductivity of the solution, with the operation terminating once conductivity drops to the range of 600 to 800 μS/cm. This precise endpoint control ensures that salt removal is maximized without over-processing the batch. Furthermore, the subsequent crystallization step, performed using solvents like acetone at controlled low temperatures (-15 to -5°C), leverages the already desalted state of the solution to promote the formation of high-quality crystals. This dual-stage purification (electrodialysis followed by crystallization) effectively minimizes the presence of related substances and residual solvents, delivering a product that is chemically stable and suitable for formulation with imipenem.
How to Synthesize Cilastatin Efficiently
The synthesis and purification protocol outlined in the patent provides a clear roadmap for manufacturing high-quality cilastatin suitable for commercial distribution. The process integrates reaction, desalting, and crystallization into a cohesive workflow that minimizes unit operations and maximizes throughput. By adhering to the specific parameters regarding pH control, voltage application, and solvent ratios, manufacturers can consistently achieve yields exceeding 90%, a marked improvement over the sub-80% yields typical of older resin-based methods. The detailed standardized synthesis steps below outline the critical operational windows necessary to replicate this success in a GMP environment.
- React Compound (V) with cysteine hydrochloride in an alkaline aqueous solution (preferably NaOH) at 45-55°C to form crude cilastatin.
- Acidify the reaction mixture to pH 2-3 using mineral acid, then subject the solution to electrodialysis at 10-100V until conductivity reaches 600-800 μS/cm to remove sodium chloride.
- Concentrate the desalted solution under reduced pressure, dissolve in organic solvent (e.g., acetone), decolorize with activated carbon, and crystallize at -15 to -5°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrodialysis-based purification method translates directly into tangible operational efficiencies and cost optimizations. The elimination of expensive ion-exchange resins and macroporous adsorbents removes a significant variable cost from the manufacturing bill of materials. Additionally, the simplified workflow reduces the labor hours and equipment occupancy time required for purification, allowing for faster batch turnover. The robustness of the electrodialysis process also enhances supply chain reliability by reducing the risk of batch failures due to resin exhaustion or breakthrough, ensuring a more consistent supply of this critical antibiotic adjuvant to the global market.
- Cost Reduction in Manufacturing: The transition away from consumable resin columns significantly lowers the direct material costs associated with purification. Traditional resin methods require frequent regeneration or replacement, adding both material costs and downtime; electrodialysis membranes have longer lifespans and do not require chemical regeneration cycles. Furthermore, the substantial increase in yield—from roughly 76% in comparative examples to over 92% in the patented method—means that less raw material is required to produce the same amount of finished goods, driving down the cost per kilogram of the final API intermediate significantly.
- Enhanced Supply Chain Reliability: The scalability of electrodialysis is a major advantage for securing long-term supply contracts. Unlike batch-wise resin columns which can become bottlenecks, electrodialysis stacks can be easily scaled by adding more membrane pairs or running in continuous mode. This flexibility allows manufacturers to respond rapidly to spikes in demand for imipenem/cilastatin combinations without the lead time associated with sourcing and qualifying new batches of specialized adsorbent resins. The process stability ensures that delivery schedules are met with greater consistency.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method offers a cleaner production profile. The reduction in chemical usage for resin regeneration decreases the volume of hazardous waste streams requiring treatment. Moreover, the high purity of the final product reduces the burden on downstream quality control labs and minimizes the risk of rejection due to failed ignition residue tests. The ability to recycle solvents like acetone in the crystallization step further aligns the process with green chemistry principles, facilitating easier regulatory approval in environmentally stringent markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrodialysis purification technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on how this method outperforms legacy techniques in terms of yield, purity, and operational simplicity.
Q: Why is electrodialysis superior to ion exchange resins for cilastatin purification?
A: Unlike ion exchange resins which can produce ammonium salts requiring further acidification (generating new impurities) or cause exothermic degradation, electrodialysis selectively removes inorganic salts like NaCl without altering the chemical structure of the cilastatin molecule, resulting in higher purity and yield.
Q: What are the critical parameters for the electrodialysis step in this process?
A: The process requires applying a voltage between 10 to 100V across ion-exchange membranes. The operation continues until the specific conductivity of the solution drops to a range of 600 to 800 μS/cm, ensuring effective removal of sodium and chloride ions while retaining the larger cilastatin molecules.
Q: How does this method improve the ignition residue specifications?
A: Traditional methods often leave behind significant inorganic salt residues. By utilizing electrodialysis to physically separate ions from the organic product before crystallization, the final cilastatin sterling exhibits significantly lower ignition residue, meeting stringent pharmacopeial standards for parenteral antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cilastatin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the efficacy and safety of life-saving antibiotic therapies. Our technical team has extensively analyzed the advancements presented in CN102675175B and possesses the expertise to implement this electrodialysis purification strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including low ignition residue and precise stereochemical integrity, guaranteeing that every batch of cilastatin meets the highest international standards.
We invite pharmaceutical partners to collaborate with us to leverage these process improvements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this purification route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of imipenem/cilastatin sodium remains competitive, compliant, and uninterrupted.
