Advanced Green Synthesis of 7-ANCA: Enhancing Purity and Scalability for Global Cephalosporin Production
Advanced Green Synthesis of 7-ANCA: Enhancing Purity and Scalability for Global Cephalosporin Production
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical antibiotic intermediates. Patent CN101357927A introduces a groundbreaking preparation method for 7-amino-3-non-3-cephalosporin-4-carboxylic acid, commonly known as 7-ANCA, which serves as the essential parent nucleus for third-generation cephalosporins like Ceftizoxime and Ceftibuten. This innovative technology addresses long-standing challenges in beta-lactam synthesis by replacing hazardous reagents with greener alternatives, specifically utilizing metal borohydride reduction followed by enzymatic deprotection. For R&D directors and procurement specialists, this patent represents a significant leap forward in process chemistry, offering a route that not only improves product appearance and purity but also drastically simplifies waste management protocols. By shifting away from traditional phosphorus-based deprotection strategies, this method aligns perfectly with modern green chemistry principles while ensuring the economic viability required for large-scale API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
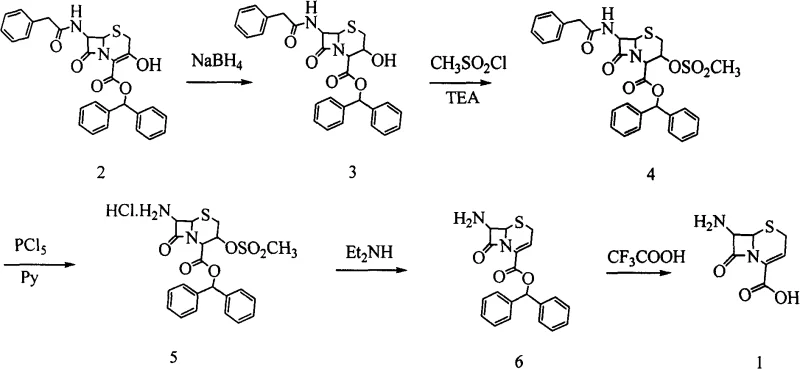
Historically, the synthesis of 7-ANCA has relied on harsh chemical conditions that pose significant operational and environmental burdens. As illustrated in the traditional synthetic routes, earlier methods frequently employed phosphorus pentachloride (PCl5) for the removal of protecting groups on the 7-amino position, coupled with trifluoroacetic acid (TFA) for deprotecting the 4-carboxyl group. These conventional approaches, while chemically feasible, suffer from severe drawbacks including the generation of substantial quantities of phosphorus-containing waste and the requirement for expensive, corrosive reagents. Furthermore, the industrial yields of these legacy processes are often inconsistent, and the resulting product frequently exhibits poor physical appearance, necessitating additional purification steps that erode profit margins. The reliance on heavy metal catalysts or toxic organotin compounds in some variations further complicates the regulatory landscape, making these routes increasingly unattractive for modern, compliance-focused manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated sequence of reduction, esterification, and elimination reactions that preserve the structural integrity of the cephem nucleus. The process begins with the selective reduction of the 3,4-double bond using metal borohydrides, followed by sulfonate esterification and subsequent elimination to restore unsaturation. Crucially, the final deprotection steps employ catalytic hydrogenation to remove the p-nitrobenzyl ester and immobilized acylase enzymes to cleave the 7-acyl group. This strategic shift eliminates the need for PCl5 and TFA entirely, resulting in a cleaner reaction profile and a product with superior optical properties. The use of enzymatic catalysis in the final step is particularly noteworthy, as it operates under mild physiological pH conditions, minimizing the risk of beta-lactam ring opening and ensuring high-purity 7-ANCA suitable for downstream coupling reactions in antibiotic synthesis.
Mechanistic Insights into Borohydride Reduction and Enzymatic Deprotection
The core of this technological advancement lies in the precise control of oxidation states and protecting group orthogonality. In the initial step, the use of potassium borohydride or sodium borohydride at cryogenic temperatures (ranging from -70°C to -30°C) allows for the selective reduction of the delta-3 double bond without affecting the sensitive beta-lactam carbonyl or the p-nitrobenzyl ester moiety. This selectivity is paramount; if the ester were reduced to an alcohol, the subsequent steps would fail. Following reduction, the 3-hydroxyl group is activated via mesylation or tosylation using sulfonyl chlorides in the presence of organic bases like triethylamine. The subsequent elimination reaction, triggered by alkali treatment, regenerates the double bond with high stereochemical fidelity. This reduction-oxidation sequence effectively 'resets' the stereochemistry at the C-3 position, ensuring the correct geometry required for biological activity in the final antibiotic.
Furthermore, the impurity control mechanism is robust due to the mild nature of the final deprotection steps. Traditional acid hydrolysis often leads to the formation of delta-2 isomers and ring-opened byproducts, which are difficult to separate. By employing catalytic hydrogenation (using Pd/C or Pt/C) for the carboxyl deprotection and immobilized acylase (such as PGK-400) for the amino deprotection, the process avoids strong acids and bases in the final stages. The enzymatic step, operating at a controlled pH of 6-9 and temperatures around 30-33°C, exhibits high specificity for the phenylacetyl side chain, leaving the cephem core untouched. This specificity significantly reduces the complexity of the impurity profile, facilitating easier crystallization and drying of the final white solid powder, which meets stringent pharmacopeial standards for cephalosporin intermediates.
How to Synthesize 7-ANCA Efficiently
The synthesis of 7-amino-3-non-3-cephalosporin-4-carboxylic acid described in this patent offers a reproducible framework for laboratory and pilot-scale production. The procedure involves dissolving the starting 3-hydroxy-cepham compound in solvents such as tetrahydrofuran or dichloromethane and carefully controlling the temperature during the borohydride addition to prevent exothermic runaway. Detailed standardized operating procedures regarding stoichiometry, solvent recycling, and catalyst loading are critical for achieving the reported molar yields of up to 80%. For process chemists looking to implement this route, attention to the pH control during the enzymatic step is vital to maintain enzyme activity and product stability. The detailed standardized synthesis steps see the guide below.
- Reduce the 3,4-double bond of 7-phenylacetamido-3-hydroxy-3-cephem-4-carboxylic acid-p-nitrobenzyl ester using potassium borohydride or sodium borohydride at low temperatures (-70°C to -30°C).
- Esterify the 3-hydroxyl group with a sulfonyl chloride (e.g., methanesulfonyl chloride) in the presence of an organic base, followed by elimination with alkali to restore the double bond.
- Remove the p-nitrobenzyl protecting group via catalytic hydrogenation, followed by enzymatic removal of the 7-acyl group using immobilized acylase to yield the final 7-ANCA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By eliminating the consumption of trifluoroacetic acid and phosphorus pentachloride, manufacturers can achieve significant cost reduction in API manufacturing through the removal of expensive raw material line items and the associated costs of hazardous waste disposal. The process replaces these costly inputs with more commodity-grade reagents like borohydrides and sulfonyl chlorides, which are widely available and subject to less volatile pricing fluctuations. Additionally, the ability to recover and reuse noble metal catalysts (such as palladium on carbon) and immobilized enzymes further enhances the cost-efficiency profile, turning what were once consumable expenses into manageable capital assets with extended lifecycles.
- Cost Reduction in Manufacturing: The elimination of trifluoroacetic acid and phosphorus pentachloride removes two of the most expensive and hazardous reagents from the bill of materials. This substitution not only lowers direct material costs but also reduces the burden on waste treatment facilities, as the process no longer generates large volumes of phosphorus-containing sludge or fluoride-rich wastewater. The overall simplified workflow reduces utility consumption and labor hours associated with handling corrosive materials, leading to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like sodium borohydride and methanesulfonyl chloride mitigates the risk of supply disruptions often associated with specialized or regulated reagents. The robustness of the enzymatic step ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, ensuring a steady flow of high-purity intermediates to downstream antibiotic formulation units without the bottleneck of complex purification protocols.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as hydrogenation and filtration that are common in existing pharmaceutical infrastructure. From an environmental perspective, the drastic reduction in toxic waste generation simplifies regulatory compliance and lowers the carbon footprint of the manufacturing site. This alignment with green chemistry principles future-proofs the supply chain against tightening environmental regulations, ensuring long-term operational continuity and enhancing the corporate sustainability profile for stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 7-ANCA synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and equipment requirements. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the total cost of ownership.
Q: How does this new 7-ANCA synthesis method improve environmental compliance compared to traditional routes?
A: The novel process eliminates the use of phosphorus pentachloride (PCl5) and trifluoroacetic acid (TFA), thereby avoiding the generation of large amounts of phosphorus-containing waste and expensive acidic effluents associated with conventional methods.
Q: What are the key advantages of using enzymatic deprotection in the final step of 7-ANCA production?
A: Using immobilized acylase for the removal of the 7-acyl protecting group offers high regioselectivity under mild pH conditions (6-9), which preserves the integrity of the beta-lactam ring and reduces the formation of degradation impurities often seen with harsh chemical hydrolysis.
Q: Can this synthesis route be scaled for commercial production of third-generation cephalosporins?
A: Yes, the process utilizes readily available starting materials and standard unit operations such as catalytic hydrogenation and filtration, making it highly suitable for commercial scale-up while maintaining high molar yields ranging from 45% to over 80% depending on specific optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-ANCA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global fight against bacterial infections. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the advanced synthesis methods described in CN101357927A can be seamlessly translated from the laboratory to full-scale manufacturing. We are committed to delivering high-purity 7-ANCA that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to process excellence ensures that every batch not only meets chemical standards but also adheres to the highest levels of safety and environmental stewardship.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthesis technology for their cephalosporin portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your antibiotic supply chain.
