Industrial Scale-Up of Tolterodine: A Safer Borohydride-Based Synthetic Route
The pharmaceutical landscape for treating overactive bladder has long relied on the efficacy of Tolterodine, a potent muscarinic receptor antagonist. However, the manufacturing processes for this critical active pharmaceutical ingredient (API) have historically been plagued by safety hazards and high operational costs. Patent CN1903833A introduces a transformative synthetic methodology that addresses these industrial bottlenecks by replacing dangerous hydride reagents with a safer, more economical borohydride-based system. This innovation not only streamlines the production of racemic Tolterodine but also ensures a robust pathway to its biologically active L-tartrate salt. For stakeholders in the fine chemical sector, this patent represents a pivotal shift towards greener, more scalable chemistry that aligns with modern regulatory and economic demands.
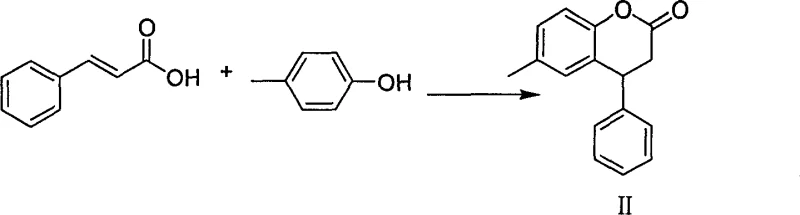
By leveraging common starting materials such as p-cresol and cinnamic acid, the disclosed route minimizes supply chain volatility while maximizing yield efficiency. The core breakthrough lies in the strategic ring-opening reduction of a dihydrocoumarin intermediate, a step that traditionally required pyrophoric reagents. This document provides a comprehensive technical analysis of this novel pathway, offering R&D directors and procurement managers deep insights into how this chemistry can be leveraged for cost reduction in API manufacturing and enhanced supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tolterodine and its intermediates has depended heavily on aggressive reducing agents such as lithium aluminum hydride (LiAlH4) or diisobutylaluminum hydride (DIBAL-H). These reagents, while effective in laboratory settings, pose severe challenges for commercial scale-up due to their extreme reactivity with moisture and air, necessitating specialized equipment and rigorous safety protocols. Furthermore, the use of these reagents often generates significant amounts of aluminum-containing waste, complicating downstream purification and environmental compliance. Previous methods also frequently involved harsh methylation and demethylation steps using reagents like boron tribromide, which are corrosive and difficult to handle on a multi-ton scale. These factors collectively drive up the cost of goods sold (COGS) and introduce substantial risk into the production schedule, making traditional routes less attractive for high-volume manufacturing.
The Novel Approach
The methodology outlined in CN1903833A circumvents these issues by employing a catalytic system comprising sodium borohydride or potassium borohydride alongside a Lewis acid catalyst. This combination allows for the selective reduction of the lactone ring in 6-methyl-4-phenyl-3,4-dihydrocoumarin under much milder conditions. The process utilizes readily available solvents like ethanol or methanol, eliminating the need for anhydrous ether environments typically required for aluminum hydrides. By shifting to this chemistry, manufacturers can achieve high yields, often exceeding 90%, while drastically simplifying the workup procedure. This approach not only enhances operator safety but also reduces the burden on waste treatment facilities, offering a clear path toward sustainable and cost-effective production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid Catalyzed Borohydride Reduction
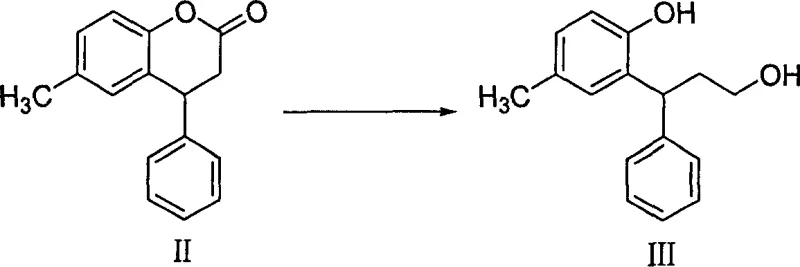
The heart of this synthetic innovation is the activation of the lactone carbonyl by a Lewis acid, which facilitates nucleophilic attack by the borohydride anion. In the absence of a catalyst, sodium borohydride is generally too mild to reduce esters or lactones efficiently at ambient temperatures. However, the addition of catalysts such as iodine, zinc chloride, or aluminum trichloride generates an activated species, likely an acyloxyborane or a coordinated complex, which is highly susceptible to hydride transfer. This mechanistic nuance allows the reaction to proceed smoothly in protic solvents like ethanol, where the solvent itself may participate in stabilizing the transition state. The result is a clean conversion to the corresponding diol or hydroxy-alcohol intermediate without the over-reduction or side reactions often seen with more aggressive hydrides.
From an impurity control perspective, this mechanism offers distinct advantages. The mildness of the borohydride system minimizes the formation of degradation products that can arise from the harsh thermal conditions required by alternative methods. Furthermore, the byproducts of the reaction, primarily borate salts, are water-soluble and easily removed during the aqueous workup, leading to a crude product of exceptional purity. This high level of cleanliness is crucial for subsequent steps, particularly the halogenation and amination reactions, where trace impurities can act as poisons or lead to difficult-to-remove byproducts. The ability to maintain high chemical fidelity throughout the synthesis ensures that the final API meets stringent pharmacopeial standards with minimal recrystallization efforts.
How to Synthesize Tolterodine Efficiently
The synthesis of Tolterodine via this route involves a logical sequence of condensation, reduction, functional group interconversion, and resolution. The process begins with the acid-catalyzed cyclization of p-cresol and cinnamic acid, followed by the critical borohydride reduction step discussed previously. Subsequent transformations involve converting the alcohol to a leaving group, protecting the phenolic hydroxyl to prevent side reactions during amination, and finally coupling with diisopropylamine. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures for each stage, are provided in the guide below to ensure reproducibility and safety.
- Condense p-cresol and cinnamic acid under sulfuric acid catalysis to form 6-methyl-4-phenyl-3,4-dihydrocoumarin.
- Perform a ring-opening reduction of the coumarin intermediate using sodium borohydride and a Lewis acid catalyst like iodine or zinc chloride.
- Convert the resulting propanol to a halide, protect the phenol, couple with diisopropylamine, deprotect, and resolve with L-tartaric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The substitution of exotic, high-cost reagents with commodity chemicals fundamentally alters the cost structure of the manufacturing process. Sodium borohydride is produced on a massive global scale for various industries, ensuring a stable supply and predictable pricing, unlike the specialized organometallic reagents used in legacy processes. This shift mitigates the risk of supply disruptions and allows for more accurate long-term budgeting. Additionally, the simplified safety profile reduces the need for expensive containment infrastructure and specialized training, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The elimination of lithium aluminum hydride and DIBAL-H removes a significant cost driver from the bill of materials. These traditional reagents are not only expensive to purchase but also require costly disposal methods for their hazardous waste. By switching to a borohydride-based system, manufacturers can achieve substantial cost savings through reduced raw material expenses and lower waste management fees. The higher yields reported in the patent examples also contribute directly to improved material efficiency, ensuring that more product is obtained per unit of starting material, which is a key metric for profitability in competitive API markets.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals like sodium borohydride, iodine, and common solvents ensures that the supply chain is robust and resilient. Unlike proprietary catalysts or niche reagents that may have single-source suppliers, the inputs for this process are widely available from multiple vendors globally. This diversification reduces the risk of production stoppages due to vendor shortages or logistics delays. Furthermore, the stability of the intermediates allows for flexible scheduling and potential storage, providing supply chain planners with greater agility to respond to market demand fluctuations without compromising quality.
- Scalability and Environmental Compliance: The safety profile of this process makes it inherently more scalable. The absence of pyrophoric reagents means that reactions can be run in standard stainless steel reactors without the need for inert gas blanketing to the same extreme degree required for aluminum hydrides. This ease of scale-up accelerates the timeline from pilot plant to commercial production. Moreover, the generation of less hazardous waste aligns with increasingly strict environmental regulations, reducing the regulatory burden and potential liability associated with chemical manufacturing. This compliance advantage is critical for maintaining uninterrupted operations in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why is the borohydride reduction method superior to lithium aluminum hydride for Tolterodine?
A: The borohydride method eliminates the need for hazardous and expensive reagents like LiAlH4 or DIBAL-H. It operates under milder conditions, significantly improving operational safety and reducing waste treatment costs in industrial settings.
Q: What is the role of the Lewis acid catalyst in the reduction step?
A: The Lewis acid catalyst, such as iodine or zinc chloride, activates the lactone carbonyl group, enabling the milder sodium borohydride to effectively reduce the ester to the corresponding alcohol without requiring extreme temperatures.
Q: How is the chiral purity of L-Tolterodine Tartrate achieved in this process?
A: Chiral purity is achieved through a classical resolution step in the final stage, where racemic Tolterodine is reacted with L-(+)-tartaric acid to selectively crystallize the desired L-(+)-tartrate-R-tolterodine salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolterodine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Tolterodine meets the highest international standards. We are committed to delivering consistent quality and reliability for your pharmaceutical supply chain.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this method. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Let us help you secure a competitive edge in the market with a supply solution that is both economically and technically superior.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
