Scalable Manufacturing of Tolterodine Intermediates via Novel Reductive Ring-Opening Technology
Scalable Manufacturing of Tolterodine Intermediates via Novel Reductive Ring-Opening Technology
The global demand for effective treatments for Overactive Bladder (OAB) syndrome continues to drive the pharmaceutical industry's need for reliable, high-quality active pharmaceutical ingredients (APIs) and their precursors. Patent CN100500642C introduces a transformative synthetic methodology for the preparation of Tolterodine and its L-tartrate salt, addressing critical bottlenecks associated with traditional manufacturing routes. This technical insight report analyzes the strategic value of this novel pathway, which replaces hazardous, high-cost reagents with safer, more economical alternatives while maintaining high product purity and yield. By leveraging a sophisticated reductive ring-opening strategy involving borohydride salts and Lewis acid catalysts, this technology offers a compelling value proposition for procurement managers and supply chain directors seeking to optimize their API sourcing strategies. The following analysis details the mechanistic advantages, commercial implications, and scalability potential of this innovative process, positioning it as a superior choice for modern pharmaceutical manufacturing.
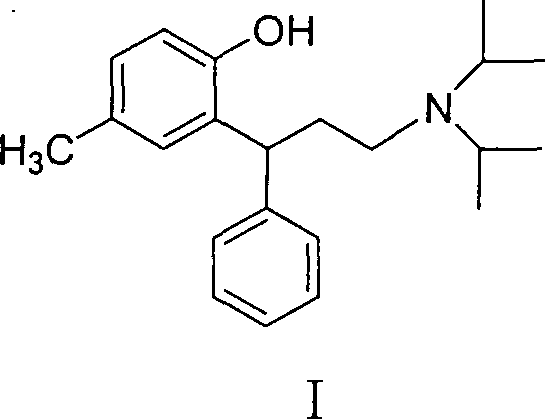
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tolterodine has relied heavily on methodologies described in earlier patents, such as U.S. Patent No. 5,382,600 and 5,922,514, which utilize aggressive and hazardous reducing agents like Lithium Aluminium Hydride (LiAlH4) or Diisobutylaluminium Hydride (DIBAL-H). These conventional approaches present significant operational challenges, including the requirement for strictly anhydrous conditions, the use of expensive solvents like ether, and the inherent safety risks associated with handling pyrophoric materials on a multi-ton scale. Furthermore, the quenching of these reactive aluminum species generates substantial volumes of hazardous waste, complicating environmental compliance and increasing disposal costs. Alternative routes employing palladium-carbon catalyzed reductive amination also suffer from the high cost of noble metal catalysts and the complexity of removing trace metal impurities to meet stringent regulatory standards for pharmaceutical products. These factors collectively contribute to elevated production costs, extended lead times, and increased supply chain vulnerability for manufacturers relying on legacy synthetic technologies.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN100500642C pioneers a streamlined synthetic route that fundamentally reimagines the reduction of the coumarin intermediate. By employing sodium borohydride or potassium borohydride in conjunction with accessible Lewis acid catalysts such as iodine, zinc chloride, or boron trifluoride diethyl etherate, this novel approach achieves efficient reductive ring-opening under significantly milder conditions. This strategic shift eliminates the need for dangerous pyrophoric reagents and allows for the use of protic solvents like ethanol and methanol, which are cheaper, safer, and easier to recover than the ether solvents required by traditional methods. The result is a process that not only enhances operator safety but also drastically simplifies the workup procedure, leading to higher overall yields and reduced waste generation. This innovation represents a paradigm shift in Tolterodine manufacturing, offering a robust platform for cost reduction in pharmaceutical intermediates manufacturing while ensuring consistent product quality.
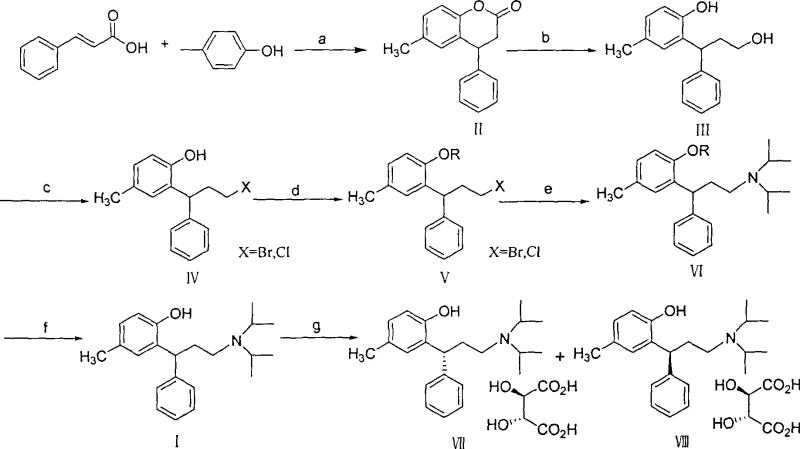
Mechanistic Insights into Borohydride-Mediated Reductive Ring-Opening
The core innovation of this patent lies in the mechanistic elegance of the reductive ring-opening of 6-methyl-4-phenyl-3,4-coumarin (Compound II) to form 3-(2-hydroxy-5-methylphenyl)-3-phenylpropanol (Compound III). In this transformation, the Lewis acid catalyst plays a pivotal role by coordinating with the carbonyl oxygen of the lactone ring, thereby increasing the electrophilicity of the carbonyl carbon and facilitating nucleophilic attack by the hydride species generated from the borohydride salt. This activation allows the reduction to proceed efficiently at reflux temperatures in alcoholic solvents, a condition under which sodium borohydride alone would typically be unreactive towards esters or lactones. The reaction mechanism avoids the formation of unstable aluminum alkoxide intermediates characteristic of LiAlH4 reductions, instead proceeding through a more manageable borate ester pathway that is easily hydrolyzed during the aqueous workup. This mechanistic distinction is crucial for R&D directors, as it implies a cleaner reaction profile with fewer side products and a simplified purification process, directly impacting the final impurity profile of the API.
Furthermore, the control of impurities in this novel route is inherently superior due to the selectivity of the borohydride-Lewis acid system. Unlike strong reducing agents that may indiscriminately reduce other functional groups or cause over-reduction, this catalytic system is highly chemoselective for the lactone moiety in the presence of the aromatic rings and the phenolic hydroxyl group (which is transiently involved in the ring structure). The subsequent steps, involving halogenation with agents like phosphorus tribromide or thionyl chloride followed by protection and amination, are standard high-yielding transformations that benefit from the high purity of the propanol intermediate. The ability to utilize various protecting groups (esters, silyl ethers, benzyl) provides additional flexibility for process optimization, allowing manufacturers to tailor the synthesis to their specific equipment and solvent recovery capabilities. This level of mechanistic control ensures that the final Tolterodine product meets rigorous quality specifications with minimal effort, reducing the burden on analytical quality control laboratories.
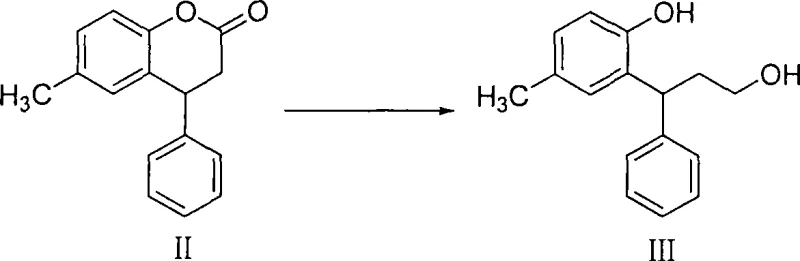
How to Synthesize Tolterodine Intermediates Efficiently
The synthesis of Tolterodine intermediates via this patented route involves a sequence of well-defined chemical transformations that begin with the acid-catalyzed condensation of p-cresol and cinnamic acid. This initial step forms the coumarin scaffold, which serves as the key precursor for the subsequent reductive ring-opening. The detailed standardized synthesis steps involve precise control of reaction temperatures, stoichiometry of the Lewis acid catalyst, and careful management of the exothermic nature of the borohydride addition. For a comprehensive understanding of the specific operating parameters, solvent choices, and workup procedures required to replicate this high-yielding process, please refer to the technical guide below.
- Condense p-cresol and cinnamic acid under sulfuric acid catalysis to form 6-methyl-4-phenyl-3,4-coumarin.
- Perform reductive ring-opening of the coumarin using sodium borohydride and a Lewis acid catalyst (e.g., iodine or zinc chloride) to yield the propanol intermediate.
- Convert the propanol to a halogenated propane, protect the phenolic hydroxyl group, couple with diisopropylamine, and finally deprotect to obtain racemic Tolterodine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical efficiency. The elimination of expensive and hazardous reagents like LiAlH4 and DIBAL-H translates directly into significant cost savings on raw materials and waste disposal, while the use of common solvents like ethanol reduces dependency on volatile specialty solvent markets. Moreover, the improved safety profile of the process minimizes the risk of production shutdowns due to safety incidents, thereby enhancing supply chain reliability and ensuring consistent delivery schedules for downstream API manufacturers. The robustness of the chemistry also facilitates easier technology transfer and scale-up, allowing suppliers to respond more agilely to fluctuations in market demand without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The replacement of high-cost reducing agents and noble metal catalysts with inexpensive borohydride salts and Lewis acids creates a fundamentally more economical cost structure for Tolterodine production. By avoiding the complex quenching and filtration steps associated with aluminum and palladium residues, manufacturers can reduce processing time and utility consumption, leading to substantial operational expenditure savings. Additionally, the higher yields reported in the patent embodiments mean that less raw material is required to produce the same amount of finished product, further driving down the cost of goods sold and improving margin potential for the entire supply chain.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium borohydride, iodine, and ethanol ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialized reagent markets. This accessibility allows for greater flexibility in sourcing and inventory management, enabling manufacturers to maintain optimal stock levels without the fear of supply disruption. The simplified process flow also reduces the number of critical process parameters that need tight control, making the manufacturing process more robust against minor variations in raw material quality and thus ensuring a more consistent and reliable supply of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The benign nature of the reagents and solvents used in this route significantly lowers the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulations on industrial emissions and waste. The absence of heavy metal catalysts simplifies the validation process for API registration, as there is no need for extensive testing and validation of metal clearance steps. This environmental and regulatory advantage facilitates faster scale-up from pilot plant to commercial production, allowing companies to bring products to market more quickly and efficiently while maintaining a strong commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Tolterodine synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on safety, cost, and scalability concerns that are critical for decision-makers in the pharmaceutical industry.
Q: What are the primary safety advantages of this Tolterodine synthesis route compared to conventional methods?
A: This novel route eliminates the use of hazardous reagents such as Lithium Aluminium Hydride (LiAlH4) and DIBAL-H, which are pyrophoric and require strict anhydrous conditions. By utilizing sodium borohydride or potassium borohydride in the presence of Lewis acid catalysts, the process significantly reduces operational risks, simplifies quenching procedures, and lowers the barrier for safe industrial scale-up.
Q: How does the new reductive ring-opening method impact production costs?
A: The substitution of expensive and difficult-to-handle reducing agents with cost-effective borohydride salts leads to substantial raw material savings. Furthermore, the milder reaction conditions reduce energy consumption and minimize the need for specialized corrosion-resistant equipment, resulting in a more economically viable manufacturing process for high-purity Tolterodine intermediates.
Q: Is this synthetic pathway suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the method is designed for industrial suitability. The use of common solvents like ethanol and methanol, combined with high-yield reactions and simplified workup procedures (such as avoiding complex metal removal steps associated with palladium catalysts), ensures that the process is robust, reproducible, and capable of meeting the demands of commercial-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolterodine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the route disclosed in CN100500642C and confirmed its potential for delivering high-purity Tolterodine intermediates with exceptional efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling, guaranteeing that every batch of Tolterodine intermediate we produce adheres to the highest international standards for safety and efficacy.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cost-effective and scalable synthetic route for their Tolterodine projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and enhance your product's market position. Let us be your trusted partner in navigating the complexities of API intermediate manufacturing with precision and reliability.
