Scalable Manufacturing of High-Purity Butylphthalide for Global Pharmaceutical Applications
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their critical intermediates, particularly for drugs treating serious conditions like cerebral ischemia. A significant technological breakthrough in this domain is detailed in patent CN111961018B, which outlines a robust preparation method for high-purity butylphthalide. This compound, chemically known as 3-n-butylphthalide, is a racemate originally extracted from celery seeds and serves as a vital therapeutic agent for reducing infarction focus and improving microcirculation in ischemic areas. The disclosed innovation addresses longstanding challenges in synthetic organic chemistry by introducing a milder initiation system for Grignard reagents and a safer reduction protocol. By shifting away from hazardous solid-feeding modes and complex chromatographic separations, this methodology offers a pathway to achieve purity levels reaching 100.00%. For global procurement teams and R&D directors, understanding this process is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering material that meets stringent injectable-grade specifications without the baggage of toxic residual solvents or heavy metal contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of butylphthalide has been plagued by safety hazards and scalability issues that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Prior art, such as the methods disclosed in CN 101962374A and CN 105130934A, often relied on the use of iodine as an initiator for Grignard reagent preparation, which inadvertently introduced new impurities into the reaction matrix, limiting final product purity to merely 97-98%. Furthermore, traditional routes frequently employed volatile ether solvents under high-temperature reflux conditions (up to 300°C in some anhydrous sodium acetate methods), creating significant explosion risks and requiring expensive, specialized containment infrastructure. The reliance on column chromatography for purification in small-batch processes, as seen in CN 105859670A, presents another bottleneck; while effective for laboratory scales, chromatography is economically unviable for multi-ton production due to high solvent consumption, slow throughput, and difficult waste disposal. These legacy methods also suffered from long reflux times and high-temperature ring-closing reactions that promoted the generation of unwanted byproducts, complicating the downstream purification landscape.
The Novel Approach
The novel approach presented in the patent data fundamentally re-engineers the synthesis workflow to prioritize safety and scalability. Instead of hazardous solid feeding, the process utilizes a sodium borohydride aqueous solution mode for the critical carbonyl reduction step, effectively eliminating the risk of explosion associated with exothermic solid-liquid interactions. The refinement strategy abandons complex chromatography in favor of a streamlined reduced pressure distillation technique, which is not only simpler to operate but also highly effective at removing trace impurities. By controlling the distillation temperature between 140-150°C under a vacuum of 100-450 Pa, the process achieves a purity exceeding 99.5% with single impurities below 0.05%. This shift allows for commercial scale-up of complex pharmaceutical intermediates by utilizing standard industrial distillation equipment rather than bespoke purification columns. The mild reaction conditions, particularly the Grignard initiation at just 48-52°C, further reduce energy consumption and thermal stress on the reactor systems, aligning perfectly with modern green chemistry principles and operational safety standards.
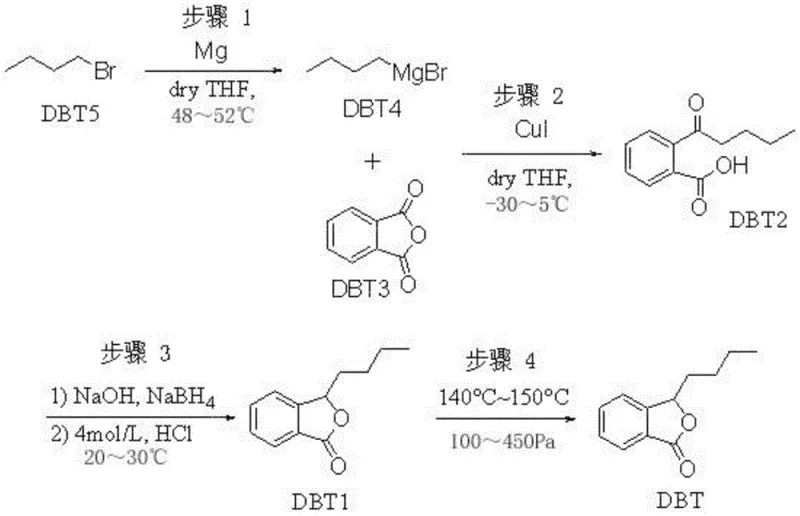
Mechanistic Insights into CuI-Catalyzed Grignard Addition and Cyclization
The core of this synthetic route lies in the precise control of organometallic reactions and subsequent cyclization mechanisms. The process begins with the formation of n-butyl magnesium bromide, where the use of pre-formed n-butyl magnesium bromide as an initiator ensures a mild and controllable start to the exothermic Grignard formation, avoiding the violent onset often seen with iodine initiation. This Grignard reagent then reacts with phthalic anhydride in the presence of a cuprous iodide (CuI) catalyst at low temperatures ranging from -30 to 5°C. The copper catalyst plays a pivotal role in facilitating the nucleophilic attack of the Grignard reagent on the anhydride, directing the formation of the key intermediate, 2-valeryl benzoic acid (DBT2), while minimizing side reactions such as over-addition or ester formation. Following the isolation of this keto-acid intermediate, the mechanism shifts to a reduction-cyclization sequence. The ketone group is selectively reduced using sodium borohydride in an alkaline aqueous environment, generating a hydroxy-acid species in situ.
Subsequent acidification triggers an intramolecular lactonization, closing the ring to form the phthalide structure. A unique aspect of this mechanism is the intermediate purification step involving ring-opening and re-closing. The crude product is treated with potassium hydroxide in methanol to open the lactone ring, forming a water-soluble potassium salt. This step is critical for impurity control, as it allows for the washing away of non-acidic organic impurities that do not form salts. Upon re-acidification, the pure hydroxy-acid precipitates or separates, and upon heating, it spontaneously re-closes to form the lactone ring of butylphthalide. This 'dissolution-recrystallization-cyclization' loop acts as a powerful chemical filter, ensuring that the final crude material entering the distillation column is already of exceptionally high quality, thereby reducing the load on the final purification unit operation.
How to Synthesize Butylphthalide Efficiently
The implementation of this synthesis route requires careful attention to temperature gradients and phase separations to maximize yield and purity. The process is divided into four distinct operational stages: Grignard reagent preparation, intermediate synthesis, reduction cyclization, and final refining. Each stage builds upon the purity of the previous one, emphasizing the importance of rigorous in-process controls. For R&D teams looking to replicate or license this technology, the key lies in maintaining the specific molar ratios, such as the 1.1 to 1.2:1 ratio of Grignard reagent to phthalic anhydride, and adhering strictly to the low-temperature protocols during the copper-catalyzed step. The following guide outlines the standardized operational flow derived from the patent examples, serving as a foundational reference for process engineers aiming to establish a robust production line for high-purity OLED material precursors or pharmaceutical actives.
- Preparation of n-butyl magnesium bromide Grignard reagent using mild initiation conditions at 48-52°C.
- Synthesis of 2-valeryl benzoic acid via CuI-catalyzed reaction with phthalic anhydride at low temperatures (-30 to 5°C).
- Reduction and cyclization using aqueous sodium borohydride followed by acidification to form crude butylphthalide.
- Purification of the final product through reduced pressure distillation at 140-150°C under 100-450 Pa vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology represents a strategic opportunity to optimize the supply base for critical neurological drug intermediates. The elimination of hazardous reagents and the simplification of purification steps directly translate to a more resilient and cost-effective supply chain. By removing the dependency on column chromatography, manufacturers can significantly increase batch sizes and throughput without proportional increases in capital expenditure or operating costs. The use of common solvents like tetrahydrofuran and methyl tert-butyl ether, coupled with aqueous workups, simplifies solvent recovery and waste treatment, further driving down the total cost of ownership. This process stability ensures that suppliers can maintain consistent delivery schedules, reducing lead time for high-purity pharmaceutical intermediates even during periods of high market demand.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and labor-intensive purification techniques with efficient vacuum distillation. Traditional methods relying on chromatography consume vast quantities of silica gel and solvents, creating a heavy burden on waste management budgets. By contrast, the distillation-based refinement described here allows for continuous or large-batch processing with minimal consumable usage. Additionally, the avoidance of solid sodium borohydride feeding reduces the need for specialized explosion-proof feeding equipment and lowers insurance premiums associated with hazardous operations. The mild reaction temperatures also result in lower energy consumption for heating and cooling cycles, contributing to substantial operational savings over the lifecycle of the plant.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes that are prone to failure or variability. This method enhances reliability by utilizing robust, well-understood chemical transformations that are less sensitive to minor fluctuations in raw material quality. The use of n-butyl magnesium bromide as a self-initiator for the Grignard reaction removes the variability associated with iodine activation, leading to more consistent reaction starts and completion times. Furthermore, the ability to produce material with purity levels suitable for direct use in injection drug preparation reduces the risk of batch rejection due to impurity profiles. This consistency allows supply chain planners to operate with leaner safety stocks, knowing that the production process is stable and predictable.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden bottlenecks, particularly in heat transfer and mixing. This synthesis route is designed with scalability in mind, utilizing reaction conditions that are easily managed in standard stainless steel reactors. The exothermic nature of the Grignard formation is controlled through dropwise addition and moderate temperatures (48-52°C), preventing thermal runaways that can shut down production lines. From an environmental perspective, the process minimizes the use of chlorinated solvents like methylene chloride, which are increasingly regulated and costly to dispose of. The shift towards aqueous workups and alcohol-based recrystallizations aligns with stricter environmental regulations, ensuring long-term compliance and reducing the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of butylphthalide using this advanced methodology. These insights are derived directly from the comparative data and experimental results provided in the patent documentation, offering clarity on how this process outperforms legacy technologies. Understanding these nuances is essential for technical buyers evaluating potential partners for long-term supply agreements.
Q: How does this method improve safety compared to traditional Grignard processes?
A: Traditional methods often use solid sodium borohydride feeding which poses explosion risks. This patent utilizes an aqueous sodium borohydride solution mode for carbonyl reduction, significantly mitigating explosion hazards and ensuring production safety during the synthesis of butylphthalide.
Q: What purity levels can be achieved with this vacuum distillation technique?
A: The refined butylphthalide obtained through the described reduced pressure distillation process reaches a purity of up to 100.00%, with maximum single impurity content controlled below 0.05%, making it suitable for high-grade pharmaceutical applications including injectables.
Q: Why is column chromatography avoided in this large-scale process?
A: Column chromatography is difficult to implement in large-scale industrial production due to high equipment maintenance costs and separation difficulties. This method replaces it with a feasible reduced pressure distillation step, which is simpler, more economical, and better suited for batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butylphthalide Supplier
As the global demand for neurological therapeutics continues to rise, securing a supply of high-quality intermediates like butylphthalide is paramount for pharmaceutical manufacturers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies to deliver products that meet the most rigorous quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch conforms to the required impurity profiles, including the control of single impurities below 0.05%.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to bring safer, more effective treatments to patients worldwide through superior chemical manufacturing.
