Scalable Solvent-Free Hydrothermal Synthesis of Bis-indolylmethane Derivatives for Industrial Applications
Scalable Solvent-Free Hydrothermal Synthesis of Bis-indolylmethane Derivatives for Industrial Applications
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN111606838B, which discloses a novel preparation method for bis-indolylmethane (BIM) compounds. These derivatives serve as critical building blocks in the synthesis of various bioactive molecules, including potential anticancer agents and enzyme inhibitors. The patent introduces a paradigm shift by replacing traditional toxic Lewis acid catalysts and surfactant-heavy systems with a clean, hydrothermal approach. By utilizing trace amounts of water under sealed, elevated temperature conditions, this method achieves high conversion rates while drastically simplifying the downstream purification process. For R&D directors and procurement managers alike, this technology represents a tangible opportunity to enhance the sustainability profile of their supply chain while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-indolylmethane derivatives has relied heavily on alkylation reactions between indoles and aldehydes or ketones in the presence of strong acid catalysts. Conventional protocols frequently employ protonic acids, Lewis acids, or Lewis acid-surfactant combined catalysts (LASCs) in solvents such as dichloroethane or aqueous surfactant systems. While effective on a small laboratory scale, these methods suffer from severe drawbacks when translated to industrial manufacturing. The use of LASCs, for instance, often necessitates the use of sodium lauryl sulfate, which generates substantial volumes of toxic wastewater that is notoriously difficult to treat due to emulsification issues. Furthermore, the recovery and recycling of homogeneous metal catalysts are technically challenging and economically inefficient, leading to increased production costs and environmental liabilities. The atom economy of these traditional processes is often poor, resulting in significant waste generation that conflicts with modern green chemistry principles and regulatory compliance requirements.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111606838B offers a streamlined, catalyst-free alternative that leverages the unique properties of water under hydrothermal conditions. The process involves charging aromatic aldehydes and indoles into a pressure-resistant reactor with only trace amounts of water, acting as a thermal catalyst rather than a stoichiometric reagent. By heating the mixture to temperatures between 110°C and 180°C in a sealed environment, the reactants undergo liquefaction and gasification, creating a self-solvent system that promotes efficient mass transfer. This eliminates the need for large volumes of external organic solvents during the reaction phase and completely avoids the introduction of toxic metal contaminants. The result is a cleaner reaction profile where the target bis-indolylmethane compounds can be isolated through simple solid-liquid separation and recrystallization, significantly reducing the complexity of post-reaction processing and waste management.
Mechanistic Insights into Hydrothermal Catalysis
The core innovation of this technology lies in the exploitation of the "self-solvent thermodynamic catalytic effect." Under standard conditions, water is a poor solvent for organic reactants like indoles and aromatic aldehydes. However, within a sealed reactor heated above 100°C, the physical properties of water change dramatically, enhancing its ability to dissolve organic species and facilitate proton transfer. The patent data indicates that at temperatures around 160°C, the trace water molecules participate in the activation of the carbonyl group of the aldehyde, promoting the electrophilic attack by the indole nucleophile. This hydrothermal environment mimics the efficiency of acid catalysis without the corrosive and contaminating effects of strong mineral acids. The sealed system maintains a pressure of 1-10 atmospheres, ensuring that the volatile components remain in the liquid or supercritical phase, thereby maximizing collision frequency and reaction kinetics. This mechanism allows for rapid conversion times, often completing within 20 to 180 minutes, which is highly advantageous for high-throughput manufacturing environments.
From an impurity control perspective, this solvent-free, metal-free approach inherently reduces the complexity of the impurity profile. Traditional metal-catalyzed routes often leave behind trace metal residues that require expensive scavenging steps to meet pharmaceutical purity specifications (e.g., ICH Q3D guidelines). By eliminating the metal catalyst entirely, the risk of heavy metal contamination is removed at the source. Additionally, the absence of surfactants prevents the formation of stable emulsions during the workup phase, allowing for sharp phase separation between the organic product and the aqueous byproduct. The patent examples demonstrate that products obtained via this method can achieve purities greater than 99.9% after simple recrystallization, indicating a highly selective reaction pathway that minimizes side reactions such as polymerization or over-alkylation.
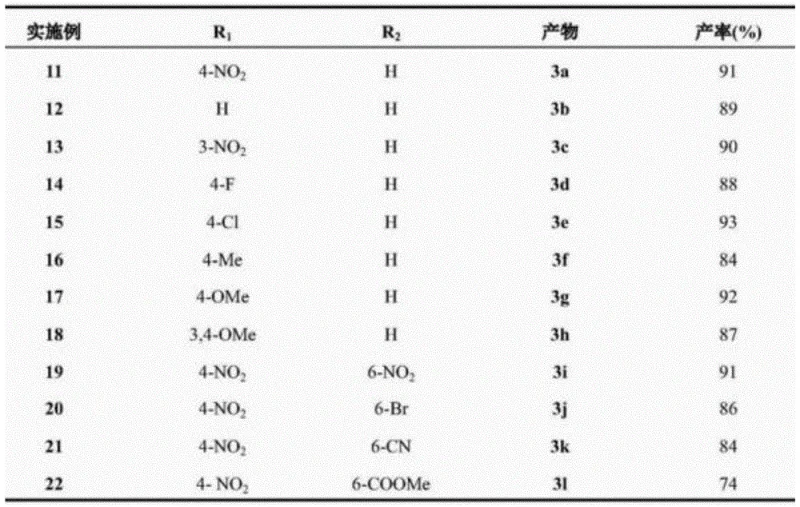
How to Synthesize Bis-indolylmethane Efficiently
Implementing this hydrothermal synthesis route requires precise control over reaction parameters to ensure reproducibility and safety. The process begins with the accurate weighing of aromatic aldehydes and indoles in a 1:2 molar ratio, along with a calculated amount of deionized water ranging from 0.185% to 1.85% molar relative to the substrate. These materials are loaded into a pressure-rated reactor, which is then sealed and heated using an external heating mantle or oil bath. Temperature monitoring is critical, with infrared sensors recommended to ensure uniform heating up to the target setpoint of 160°C. Once the reaction time is complete, the system must be cooled safely before opening to prevent solvent flash-off. The crude solid is then washed with a polar organic solvent, such as dichloromethane, to remove unreacted starting materials, followed by filtration and drying. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Charge aromatic aldehydes, indoles, and trace water (0.185%-1.85% mol) into a pressure-resistant reactor.
- Heat the sealed system to 110-180°C (optimally 160°C) for 20-180 minutes to induce hydrothermal catalysis.
- Cool to room temperature, extract with organic solvent, and purify via recrystallization to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrothermal technology translates directly into operational resilience and cost optimization. The most significant advantage is the drastic reduction in raw material costs associated with catalysts and solvents. By removing the requirement for expensive Lewis acids and surfactants, the bill of materials is simplified, and the dependency on volatile specialty chemical markets is reduced. Furthermore, the elimination of toxic metal catalysts removes the need for costly downstream purification steps, such as column chromatography or metal scavenging resin treatments, which are often bottlenecks in large-scale production. This streamlined workflow not only accelerates the manufacturing cycle but also lowers the overall energy consumption per kilogram of product produced, contributing to a lower carbon footprint for the final API intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the unit operations. Since the reaction proceeds without external solvents or catalysts, the volume of waste generated is significantly minimized. This leads to substantial savings in waste disposal fees, which are a major cost center in chemical manufacturing. Additionally, the ability to recycle the mother liquor solvent further enhances the atom economy, ensuring that raw materials are utilized to their maximum potential. The reduction in processing steps also means less labor and equipment time is required, effectively increasing the capacity utilization of existing manufacturing assets without the need for capital expenditure on new reactors.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents. Traditional methods relying on specific Lewis acids or surfactants can face disruptions if suppliers encounter production issues. In contrast, the reagents required for this hydrothermal method—aromatic aldehydes, indoles, and water—are commodity chemicals with robust, multi-source global supply chains. This commoditization of inputs reduces the risk of supply shortages and price volatility. Moreover, the simplicity of the process makes it easier to transfer between different manufacturing sites or contract manufacturing organizations (CMOs), providing greater flexibility in sourcing strategies and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates environmental challenges, particularly regarding heat transfer and waste management. This solvent-free hydrothermal method is inherently scalable because it relies on the intrinsic properties of the reactants rather than complex mixing regimes required for heterogeneous catalysis. The closed-system nature of the reaction contains volatile organic compounds (VOCs), ensuring compliance with strict environmental regulations regarding air emissions. The absence of heavy metals and surfactants in the effluent simplifies wastewater treatment, making it easier for manufacturing facilities to meet local discharge standards and avoid regulatory fines, thereby securing the long-term viability of the production license.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111606838B, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these details is crucial for R&D teams planning pilot runs and for procurement teams assessing the total cost of ownership.
Q: How does this method eliminate toxic wastewater compared to traditional LASCs catalysts?
A: Traditional methods use Lewis acid-surfactant combined catalysts (LASCs) which generate difficult-to-treat wastewater containing sodium lauryl sulfate and toxic metals. This patented hydrothermal method uses only trace water as a catalyst, completely eliminating heavy metal contamination and surfactant waste streams.
Q: What are the typical reaction conditions for scaling this synthesis?
A: The process operates in a sealed pressure vessel at temperatures between 110°C and 180°C, typically optimized at 160°C. The reaction time ranges from 20 to 180 minutes depending on the specific substrate substituents, allowing for rapid throughput in batch reactors.
Q: Is the solvent recyclable in this green synthesis process?
A: Yes, the process design includes a solvent recovery step. After the reaction, organic solvents like dichloromethane are used for extraction and washing. The mother liquor is collected, evaporated to recover the solvent, and reused, significantly reducing raw material consumption and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-indolylmethane Supplier
The technological potential of solvent-free hydrothermal synthesis aligns perfectly with NINGBO INNO PHARMCHEM's commitment to sustainable and efficient manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with state-of-the-art pressure reactors and rigorous QC labs capable of verifying stringent purity specifications, including residual solvent and heavy metal analysis. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems guarantee that every batch of bis-indolylmethane intermediate meets the highest international standards.
We invite you to explore how this innovative synthesis route can optimize your project economics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals. Let us collaborate to bring high-quality, eco-friendly chemical intermediates to your pipeline efficiently and reliably.
