Revolutionizing Bis-Indolylmethane Production: A Green Ionic Liquid Catalysis Strategy for Commercial Scale-Up
Introduction to Advanced Bis-Indolylmethane Synthesis
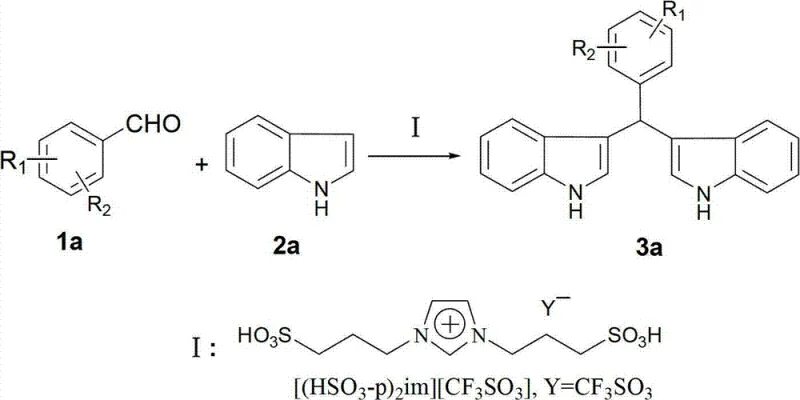
The pharmaceutical industry continuously seeks robust and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical intermediates for oncology therapeutics. Patent CN102766081A introduces a groundbreaking methodology for the synthesis of bis-indolylmethane derivatives, a class of compounds renowned for their potent biological activities including anti-cancer properties and hormone regulation capabilities. This innovation leverages a dual alkyl sulfonic acid type ionic liquid, specifically [(HSO3-p)2im][CF3SO3], to catalyze the condensation of aromatic aldehydes with indole under remarkably mild conditions. By eliminating the need for volatile organic solvents and operating effectively at room temperature, this technology addresses the pressing demand for greener manufacturing processes in the fine chemical sector. The strategic implementation of this ionic liquid catalyst not only streamlines the reaction workflow but also ensures high atom economy and superior product isolation profiles. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards sustainable and cost-effective production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-indolylmethane derivatives has relied heavily on traditional catalytic systems that present significant operational and environmental challenges for large-scale manufacturers. Conventional protocols often utilize solid acid catalysts like montmorillonite K-10 or Lewis acids such as ferric chloride, which necessitate the use of substantial quantities of volatile organic solvents like ethanol, ether, or ethyl acetate to facilitate the reaction medium. These legacy methods suffer from inherent inefficiencies, including prolonged reaction times that can extend for several hours, thereby reducing throughput capacity and increasing energy consumption in industrial reactors. Furthermore, the separation of these heterogeneous catalysts from the reaction mixture is notoriously difficult, often requiring extensive solvent elution and filtration steps that generate considerable chemical waste streams. The reliance on corrosive protonic acids also poses safety risks and equipment corrosion issues, while the difficulty in recycling these catalysts leads to elevated raw material costs and inconsistent batch-to-batch quality. Consequently, the industry has long struggled with a trade-off between reaction efficiency and environmental compliance when employing these outdated synthetic routes.
The Novel Approach
In stark contrast to these cumbersome traditional methods, the novel approach detailed in the patent utilizes a task-specific ionic liquid that functions as both the catalyst and the reaction medium, fundamentally redefining the process economics. This innovative strategy operates under solvent-free conditions at ambient temperatures, typically between 20°C and 30°C, which drastically reduces the thermal energy input required for production. The unique physicochemical properties of the [(HSO3-p)2im][CF3SO3] ionic liquid allow it to activate the aromatic aldehyde efficiently while remaining immiscible with the organic product, facilitating an exceptionally simple workup procedure. Upon completion of the reaction, the addition of deionized water dissolves the ionic liquid catalyst, allowing the solid product to be isolated via simple filtration without the need for complex extraction or chromatography. This seamless separation capability not only accelerates the manufacturing cycle but also enables the direct recovery and reuse of the catalyst from the aqueous phase after concentration, creating a closed-loop system that minimizes waste generation. Such a streamlined process offers a compelling value proposition for supply chain managers seeking to reduce lead times and enhance the sustainability profile of their chemical sourcing.
Mechanistic Insights into Ionic Liquid Catalyzed Condensation
The efficacy of this synthesis relies on the dual Brønsted acidic nature of the ionic liquid catalyst, which possesses sulfonic acid groups capable of strongly activating the carbonyl group of the aromatic aldehyde. Upon interaction with the catalyst, the electrophilicity of the aldehyde carbon is significantly enhanced, promoting a rapid nucleophilic attack by the electron-rich C3 position of the indole molecule. This initial addition forms an intermediate alcohol species, which subsequently undergoes acid-catalyzed dehydration to generate a reactive indolyl-methyl cation. This cationic intermediate is then attacked by a second equivalent of indole, leading to the formation of the stable bis-indolylmethane skeleton. The mild acidity provided by the ionic liquid is sufficient to drive this cascade to completion within minutes, yet gentle enough to prevent the polymerization or degradation of the sensitive indole ring system, which is a common side reaction in stronger acidic media. This precise control over reaction kinetics ensures a clean impurity profile, reducing the burden on downstream purification units and ensuring high-quality output suitable for pharmaceutical applications.
Furthermore, the structural integrity of the ionic liquid contributes to superior impurity control mechanisms throughout the synthesis lifecycle. Unlike homogeneous mineral acids that can cause over-reaction or charring of organic substrates, the organized ionic environment stabilizes the transition states and intermediates, favoring the desired bis-substitution pathway. The absence of organic solvents eliminates the risk of solvent-derived impurities or azeotropic complications during isolation, resulting in a crude product of exceptional purity. The water-solubility of the catalyst post-reaction acts as a built-in purification step, as any residual catalyst is washed away into the aqueous layer, leaving the organic product free from acidic contamination. This inherent selectivity and ease of purification are critical for maintaining stringent quality standards required for API intermediates, where trace metal or acid residues can compromise the safety and efficacy of the final drug product. Understanding these mechanistic advantages allows R&D teams to confidently adopt this technology for the synthesis of diverse derivative libraries with predictable outcomes.
How to Synthesize Bis-Indolylmethane Derivatives Efficiently
The practical implementation of this synthesis route is designed for simplicity and scalability, making it accessible for both laboratory optimization and industrial manufacturing environments. The process begins with the precise metering of aromatic aldehyde and indole substrates into a reaction vessel, followed by the addition of the ionic liquid catalyst in a specific molar ratio optimized for maximum turnover. The mixture is then subjected to mechanical stirring at room temperature, where the reaction proceeds rapidly to completion, monitored by standard analytical techniques to ensure full conversion. Following the reaction, a straightforward aqueous workup is performed to separate the catalyst from the product, followed by recrystallization to achieve the final purity specifications. For detailed operational parameters, stoichiometry, and specific handling instructions, please refer to the standardized synthesis guide below.
- Mix aromatic aldehyde and indole in a molar ratio of 1: 2.0~2.2 with the ionic liquid catalyst (1:50~150 ratio) in a reaction vessel.
- Stir the mixture at room temperature (20-30°C) for 5 to 15 minutes to complete the reaction.
- Add deionized water to dissolve the ionic liquid, filter the solid crude product, and recrystallize from ethanol to obtain pure bis-indolylmethane.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this ionic liquid catalyzed synthesis offers profound commercial benefits that directly address the core concerns of procurement managers and supply chain executives regarding cost stability and operational reliability. The elimination of volatile organic solvents removes a major variable cost component and mitigates the regulatory burdens associated with solvent storage, handling, and disposal, leading to substantial overhead reductions. Additionally, the ability to recycle the catalyst multiple times without significant loss of activity transforms a consumable expense into a durable asset, further driving down the cost of goods sold over the lifecycle of the product. The simplified workup procedure, which replaces complex extractions with simple filtration and washing, significantly reduces labor hours and equipment occupancy time, thereby increasing overall plant throughput and capacity utilization. These factors combine to create a resilient supply chain model that is less susceptible to raw material price fluctuations and regulatory changes, ensuring a steady and economical flow of critical intermediates.
- Cost Reduction in Manufacturing: The transition to a solvent-free system inherently lowers the raw material expenditure by removing the need for purchasing, recovering, or disposing of large volumes of organic solvents. Moreover, the reusability of the ionic liquid catalyst means that the effective cost per kilogram of catalyst consumed is drastically reduced compared to single-use mineral acids or expensive metal complexes. The energy savings derived from running the reaction at room temperature rather than under reflux conditions also contribute to a lower utility bill, enhancing the overall margin profile of the manufacturing process. By minimizing the number of unit operations required for purification, the process reduces the consumption of auxiliary materials and the wear and tear on processing equipment, leading to long-term capital preservation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent production schedules, as the short reaction times and simple workup minimize the risk of batch failures or delays caused by complex purification bottlenecks. The use of commercially available and stable starting materials, combined with a catalyst that can be stockpiled and reused, reduces dependency on fragile supply chains for specialized reagents. This reliability is crucial for maintaining continuous production lines for downstream API manufacturing, preventing costly shutdowns or inventory shortages. Furthermore, the environmental friendliness of the process simplifies permitting and compliance procedures, reducing the administrative lead time required to bring new production capacity online or to expand existing facilities.
- Scalability and Environmental Compliance: The solvent-free nature of this reaction makes it inherently safer and easier to scale up, as there are no flammable vapors to manage and heat transfer is more efficient in the absence of diluent solvents. The process aligns perfectly with green chemistry principles, generating minimal waste and avoiding the release of hazardous VOCs, which helps companies meet increasingly stringent environmental regulations and corporate sustainability goals. The aqueous waste stream containing the dissolved ionic liquid can be treated and concentrated for reuse, closing the loop on material usage and minimizing the environmental footprint of the operation. This alignment with eco-friendly standards not only future-proofs the supply chain against regulatory tightening but also enhances the brand reputation of the manufacturer among environmentally conscious partners and consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis for bis-indolylmethane derivatives. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the advantages of using [(HSO3-p)2im][CF3SO3] ionic liquid over traditional catalysts?
A: Unlike traditional protonic or Lewis acids which require harsh conditions and generate heavy metal waste, this ionic liquid operates at room temperature without solvent, is water-soluble for easy separation, and can be recycled multiple times without loss of activity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to its solvent-free nature, short reaction time (5-15 minutes), and simple workup procedure involving only water washing and filtration, which significantly reduces operational complexity and environmental impact.
Q: What is the typical yield and purity achievable with this method?
A: The patent data indicates high yields ranging from 86% to 96% across various substituted aromatic aldehydes, with high purity achieved through simple ethanol recrystallization, making it ideal for pharmaceutical intermediate standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Indolylmethane Derivatives Supplier
As the global demand for high-purity oncology intermediates continues to rise, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the pharmaceutical market. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced catalytic technologies like the ionic liquid method described in CN102766081A to deliver superior quality bis-indolylmethane derivatives. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality or delivery timelines. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of these innovative synthetic routes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our green chemistry solutions can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our bis-indolylmethane portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can build a more sustainable and efficient future for pharmaceutical manufacturing.
