Revolutionizing Ipratropium Bromide Production: A Green One-Pot Strategy for Industrial Scale-Up
Introduction to Next-Generation Ipratropium Bromide Synthesis
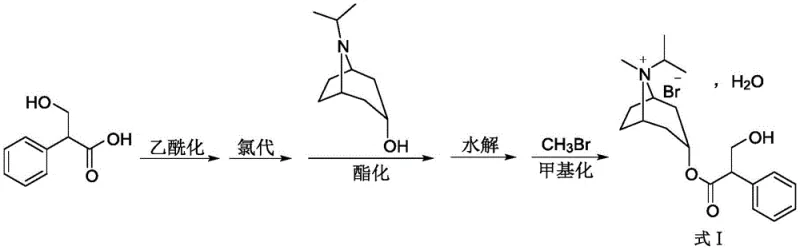
The pharmaceutical landscape for respiratory treatments continues to evolve, demanding higher purity standards and more sustainable manufacturing processes for critical active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in Chinese Patent CN115572290A, which discloses a novel one-pot method for synthesizing Ipratropium Bromide, a potent anticholinergic agent widely used for managing Chronic Obstructive Pulmonary Disease (COPD) and asthma. This innovative approach addresses longstanding inefficiencies in legacy production methods by integrating acetylation, chlorination, esterification, hydrolysis, and methylation into a streamlined workflow. By avoiding the isolation of unstable intermediates and eliminating the need for highly toxic catalysts like titanium tetrachloride, this technology offers a robust pathway for producing high-purity pharmaceutical intermediates. For global supply chains, this represents a pivotal shift towards greener chemistry that does not compromise on yield or quality, ensuring a reliable supply of this essential bronchodilator.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ipratropium Bromide has been fraught with significant chemical and operational challenges that hinder efficient commercial scale-up of complex quaternary ammonium salts. Prior art, such as the route described in US3505337A, relies heavily on the use of titanium tetrachloride (TiCl4) for formylation reactions. TiCl4 is not only highly toxic and corrosive, posing severe safety risks to personnel and equipment, but it also generates unstable intermediates prone to oxidation and decomposition. Furthermore, conventional esterification steps often require solvent-free conditions at elevated temperatures, which inadvertently promote the formation of degradation impurities and transesterification byproducts. These factors collectively result in lower overall yields, typically struggling to exceed modest benchmarks, and necessitate rigorous, costly purification protocols to meet pharmacopeial standards. The cumulative effect is a manufacturing process that is environmentally burdensome, operationally complex, and economically inefficient for modern API production.
The Novel Approach
In stark contrast, the methodology outlined in CN115572290A introduces a paradigm shift by employing a continuous one-pot strategy that fundamentally redesigns the reaction sequence. Instead of isolating sensitive intermediates, the process performs acetylation and chlorination sequentially in the same vessel, utilizing vacuum distillation to remove excess reagents which can then be recovered and recycled. The esterification step is revolutionized by the introduction of 4-Dimethylaminopyridine (DMAP) as a highly efficient catalyst in a chloroform solvent system, allowing the reaction to proceed at mild temperatures between 35°C and 55°C. This温和 condition effectively suppresses the thermal degradation pathways seen in older methods. Finally, the methylation is conducted at low temperatures (-5°C to 5°C) using a pre-prepared solution of methyl bromide, which drastically reduces the formation of enantiomeric impurities. This holistic approach not only simplifies the operational workflow but also ensures a superior impurity profile, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into DMAP-Catalyzed Esterification and Low-Temp Quaternization
The core chemical innovation driving the success of this synthesis lies in the precise control of reaction kinetics during the esterification and methylation phases. In the esterification step, the addition of DMAP acts as a nucleophilic catalyst that significantly accelerates the acylation of isopropyltropine alcohol. Unlike traditional thermal methods that rely on brute force heat to drive the equilibrium, DMAP lowers the activation energy, allowing the reaction to reach completion rapidly at 45°C. This mechanistic advantage is critical because it prevents the thermal decomposition of the tropane ring structure and minimizes the formation of elimination byproducts. The subsequent hydrolysis step is carefully managed by adjusting the pH to a specific alkaline range (9.5-10.5) using inorganic bases like potassium carbonate, ensuring selective extraction of the desired ester while leaving acidic impurities in the aqueous phase. This level of selectivity is paramount for achieving the reported HPLC purity of 99.99%.
Furthermore, the control of stereochemical integrity is addressed through the low-temperature methylation protocol. Quaternization reactions are notoriously sensitive to temperature; excessive heat can lead to racemization or the formation of unwanted N-oxide impurities. By maintaining the reaction environment between -5°C and 5°C during the addition of methyl bromide, the process kinetically favors the formation of the desired quaternary ammonium salt while suppressing side reactions. The use of a saturated dichloromethane solution of methyl bromide ensures a consistent concentration of the alkylating agent, preventing local hotspots of high reactivity that could degrade the product. This meticulous attention to thermodynamic and kinetic parameters results in an enantiomer content of less than 0.01%, a specification that is critical for the safety and efficacy of the final respiratory medication.
How to Synthesize Ipratropium Bromide Efficiently
Implementing this advanced synthesis route requires strict adherence to the defined process parameters to maximize yield and purity. The procedure begins with the acetylation of tropic acid, followed immediately by chlorination without intermediate workup, leveraging the efficiency of telescoped reactions. The subsequent esterification utilizes precise molar ratios of isopropyltropine alcohol and DMAP to ensure complete conversion. Following the reaction, a controlled hydrolysis and extraction sequence isolates the free base, which is then subjected to the final methylation step. The detailed standardized synthesis steps, including specific reagent quantities, temperature ramping rates, and stirring times validated in the patent examples, are provided in the guide below to assist technical teams in replicating this high-performance process.
- Acetylation of tropic acid with acetyl chloride or acetic anhydride at 20-40°C followed by vacuum distillation.
- Chlorination of the concentrated intermediate using thionyl chloride or oxalyl chloride at 50-70°C.
- Esterification with isopropyltropine alcohol and DMAP catalyst in chloroform at 35-55°C.
- Hydrolysis using dilute hydrochloric acid followed by pH adjustment and extraction.
- Methylation with methyl bromide in dichloromethane at -5 to 5°C to form the final quaternary ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis method translates into tangible strategic benefits that extend beyond simple chemical yield. The elimination of toxic reagents like titanium tetrachloride removes a major regulatory and safety hurdle, simplifying waste disposal and reducing the environmental compliance burden associated with hazardous material handling. Moreover, the ability to recover and reuse excess acetylating and chlorinating agents directly impacts the cost of goods sold (COGS) by minimizing raw material consumption. The streamlined nature of the process, which avoids multiple isolation and purification stages, significantly shortens the production cycle time, thereby enhancing supply chain responsiveness and reducing lead time for high-purity pharmaceutical intermediates. This operational efficiency ensures a more stable and predictable supply of Ipratropium Bromide, mitigating the risks of production delays that can impact downstream formulation schedules.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its solvent-free initial steps and the recyclability of key reagents. By conducting acetylation and chlorination without additional solvents and recovering excess reagents via vacuum distillation, the method drastically reduces solvent purchase and disposal costs. Additionally, the high catalytic efficiency of DMAP means lower catalyst loading is required to achieve full conversion, further optimizing material costs. The avoidance of complex purification steps for unstable intermediates also reduces labor and equipment usage time, contributing to substantial cost savings in the overall manufacturing budget without compromising on the stringent quality requirements of the pharmaceutical industry.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials such as tropic acid and isopropyltropine alcohol. Unlike processes dependent on specialized or hazardous reagents that may face supply constraints, the reagents in this route are commodity chemicals with robust global availability. The simplified operational workflow reduces the likelihood of batch failures due to procedural complexity, ensuring consistent output. This reliability is crucial for maintaining continuous production schedules and meeting the demands of global markets for respiratory therapies, providing a secure source of high-quality intermediates for long-term contracts.
- Scalability and Environmental Compliance: The process has been successfully validated at the kilogram scale, demonstrating its readiness for commercial scale-up of complex pharmaceutical intermediates. The mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with modern sustainability goals. Furthermore, the reduction in hazardous waste generation, particularly the elimination of titanium-containing waste streams, simplifies environmental permitting and wastewater treatment. This green chemistry approach not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of safety, efficiency, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement specialists assessing the quality credentials of potential suppliers.
Q: How does the new one-pot method improve upon traditional synthesis routes?
A: Traditional routes often rely on highly toxic and corrosive reagents like titanium tetrachloride (TiCl4) and require harsh high-temperature conditions that degrade product quality. The new one-pot method described in CN115572290A eliminates TiCl4, utilizes milder temperatures (20-70°C), and avoids intermediate isolation, significantly reducing impurity formation and operational complexity.
Q: What represents the key advantage regarding impurity control in this process?
A: The process achieves exceptional purity levels, with HPLC purity reaching 99.99% and enantiomer content controlled below 0.01%. This is largely due to the low-temperature methylation step (-5 to 5°C) and the efficient DMAP-catalyzed esterification, which minimizes side reactions like transesterification and dehydration that plague older solvent-free high-temperature methods.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly validates the method through kilogram-level scale-up trials. The elimination of intermediate purification steps, the ability to recover and reuse excess reagents like acetyl chloride, and the use of common solvents make the process highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ipratropium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot synthesis of Ipratropium Bromide can be seamlessly transitioned from the laboratory to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex quaternary ammonium salt synthesis allows us to offer a reliable supply of high-purity Ipratropium Bromide that adheres to the highest international standards, providing our partners with a competitive edge in the respiratory therapy sector.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to verify the superior impurity profile and yield advantages of our process. By partnering with NINGBO INNO PHARMCHEM, you gain access to a dedicated team focused on engineering excellence and supply chain stability, ensuring your production needs are met with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
