Advanced Manufacturing of Terbutaline Sulfate: A Breakthrough in Process Efficiency and Safety
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational safety, particularly for critical respiratory medications like Terbutaline Sulfate. The recent disclosure in patent CN110981739B presents a transformative approach to synthesizing this beta-2 adrenergic agonist, addressing long-standing inefficiencies in legacy production methods. By utilizing 3,5-dihydroxyacetophenone as a direct starting material, this novel protocol bypasses the cumbersome hydroxyl protection steps that have historically plagued the synthesis of catechol-containing intermediates. This strategic simplification not only streamlines the reaction sequence but also significantly mitigates the environmental and safety risks associated with handling highly toxic reagents such as diazomethane or organolithium compounds found in earlier art. For global procurement teams and R&D directors, this represents a pivotal shift towards greener, more cost-effective API manufacturing that aligns with modern regulatory expectations for impurity control and process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Terbutaline Sulfate has been burdened by complex multi-step sequences that introduce significant operational hazards and cost inefficiencies. Prominent legacy routes, such as those described in US3937838, rely heavily on the use of diazomethane for esterification and benzyl protection strategies that necessitate subsequent hydrogenolysis for deprotection. These traditional pathways are inherently flawed due to the extreme toxicity of diazomethane, which poses severe explosion risks and requires specialized containment infrastructure that drives up capital expenditure. Furthermore, alternative routes employing methyl lithium or selenium dioxide introduce dangerous metal reagents that complicate waste stream management and increase the burden on downstream purification processes to meet stringent heavy metal limits. The cumulative effect of these protection-deprotection cycles results in extended lead times, lower overall yields due to material loss at each stage, and a larger carbon footprint, rendering these methods increasingly obsolete in a competitive market focused on sustainability.
The Novel Approach
In stark contrast, the methodology outlined in CN110981739B leverages the inherent reactivity of the phenolic hydroxyl groups to enable a direct functionalization strategy that eliminates the need for protecting groups entirely. By employing copper(II) bromide as a selective brominating agent, the process achieves alpha-bromination of the ketone moiety while leaving the phenolic rings intact, a feat that was previously difficult to control without protection. This direct approach collapses the synthetic timeline by removing at least two major unit operations—protection and deprotection—thereby reducing solvent consumption and energy usage per kilogram of product. The subsequent transformation involves a clever tandem reduction and cyclization using sodium borohydride and sodium hydroxide, which efficiently constructs the critical epoxide intermediate in a single pot. This streamlined logic not only enhances the atom economy of the synthesis but also drastically simplifies the isolation procedures, offering a clear pathway to higher throughput and reduced manufacturing costs for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into CuBr2-Mediated Direct Bromination and Epoxide Formation
The core innovation of this synthesis lies in the selective alpha-bromination of 3,5-dihydroxyacetophenone using copper(II) bromide in a mixed solvent system of chloroform and ethyl acetate. Mechanistically, the copper salt acts as both an oxidant and a source of bromine radicals or electrophilic bromine species that target the enolizable alpha-position of the ketone. The presence of the electron-donating hydroxyl groups on the aromatic ring could theoretically lead to ring bromination; however, the specific reaction conditions and the nature of the copper reagent favor side-chain functionalization. This selectivity is crucial because it preserves the structural integrity of the resorcinol core, which is essential for the biological activity of the final API. Following bromination, the intermediate 5-bromoacetyl resorcinol undergoes reduction by sodium borohydride to generate the corresponding bromohydrin. In the presence of a base like sodium hydroxide, an intramolecular SN2 reaction occurs where the alkoxide oxygen attacks the adjacent carbon bearing the bromine leaving group, resulting in the formation of the strained three-membered epoxide ring. This cascade transformation is highly efficient and minimizes the formation of oligomeric byproducts that often plague epoxide syntheses.
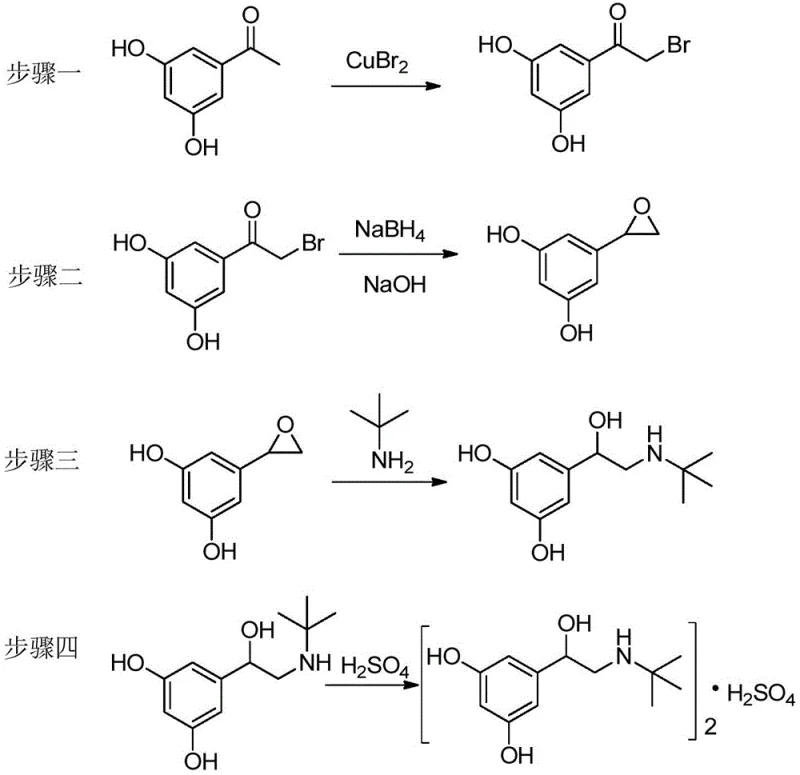
Following the formation of the epoxide intermediate, the synthesis proceeds through a regioselective ring-opening reaction with tert-butylamine. The nucleophilic attack of the amine on the less hindered carbon of the epoxide ring ensures the correct placement of the amino-alcohol side chain characteristic of beta-agonists. This step is conducted in acetonitrile at elevated temperatures to overcome the activation energy barrier of the ring strain without causing degradation of the sensitive catechol structure. Finally, the free base is converted to the sulfate salt through careful pH control in an ethanol medium. This salification step is critical for stabilizing the molecule and ensuring the appropriate crystalline form required for pharmaceutical formulation. The entire sequence demonstrates a sophisticated understanding of physical organic chemistry, leveraging reagent compatibility to achieve high purity without the need for chromatographic purification, which is a key requirement for cost reduction in API manufacturing.
How to Synthesize Terbutaline Sulfate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity profiles, particularly regarding the control of dibrominated byproducts and regioisomers during the initial bromination step. Operators must maintain the reaction temperature between 40-80°C during the copper bromide addition to ensure complete conversion while preventing thermal degradation of the phenolic starting material. The subsequent reduction and cyclization steps demand strict temperature monitoring, typically initiating at 0-10°C to manage the exotherm of the borohydride addition before warming to ambient temperatures to drive the ring closure to completion. Detailed standard operating procedures regarding solvent ratios, specifically the chloroform to ethyl acetate mix, are essential for optimizing the solubility of the copper salts and the organic intermediates. For a comprehensive breakdown of the specific molar ratios, quenching protocols, and isolation techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform alpha-bromination of 3,5-dihydroxyacetophenone using copper(II) bromide in a chloroform and ethyl acetate solvent system at 40-80°C.
- Reduce the resulting 5-bromoacetyl resorcinol with sodium borohydride in ethanol, followed by base-catalyzed cyclization using sodium hydroxide to form the epoxide intermediate.
- Conduct ring-opening reaction of the epoxide with tert-butylamine in acetonitrile at elevated temperatures to yield the free base alcohol.
- Finalize the process by salifying the free base with dilute sulfuric acid in ethanol under controlled pH conditions to obtain high-purity Terbutaline Sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented route offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for expensive and hazardous protecting group reagents such as benzyl bromide or chloromethyl methyl ether, the raw material cost profile is significantly optimized. Furthermore, the removal of high-risk reagents like diazomethane reduces the insurance premiums and specialized facility costs associated with their storage and handling, leading to indirect overhead savings that accumulate over long-term production campaigns. The simplified workflow also translates to reduced utility consumption, as fewer heating and cooling cycles are required compared to the elongated traditional routes, contributing to a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic shortening of the synthetic sequence, which inherently reduces labor hours, reactor occupancy time, and solvent volumes. By avoiding the protection and deprotection stages, the process eliminates the yield losses typically associated with these additional unit operations, thereby increasing the overall mass efficiency of the production line. Additionally, the use of commodity chemicals like copper bromide and sodium borohydride, rather than bespoke organometallic reagents, ensures that raw material pricing remains stable and predictable, shielding the supply chain from volatility in the specialty chemical market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 3,5-dihydroxyacetophenone and tert-butylamine ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. Traditional routes dependent on specific protected intermediates often face bottlenecks if a single supplier of a niche precursor encounters production issues; this new method diversifies the supply risk by utilizing bulk chemicals that are produced by multiple vendors globally. This redundancy is critical for maintaining continuous API supply to meet market demand, especially for essential respiratory medications where stockouts can have severe clinical consequences.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, operating under atmospheric pressure and moderate temperatures that do not require specialized high-pressure reactors or cryogenic equipment. This ease of scaling allows manufacturers to rapidly ramp up production capacity from pilot plant to commercial tonnage without significant capital investment in new infrastructure. Moreover, the avoidance of heavy metals like selenium and toxic gases like diazomethane simplifies wastewater treatment and废气 (exhaust gas) abatement systems, ensuring compliance with increasingly stringent environmental regulations and reducing the costs associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The answers provided reflect the specific advantages of the CuBr2-mediated pathway over conventional methods, focusing on safety, purity, and operational simplicity.
Q: How does this new method improve upon traditional Terbutaline Sulfate synthesis?
A: Traditional methods often require hazardous reagents like diazomethane or methyl lithium and involve lengthy protection-deprotection sequences. This patented route utilizes direct bromination with CuBr2, eliminating the need for hydroxyl protection, thereby reducing step count, minimizing toxic waste, and improving overall process safety.
Q: What are the critical reaction conditions for the epoxide formation step?
A: The epoxide formation involves a tandem reduction-cyclization sequence. It requires careful temperature control between 0-40°C using sodium borohydride as the reducing agent in ethanol, followed by the addition of sodium hydroxide to facilitate the intramolecular nucleophilic substitution that closes the epoxide ring.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It avoids high-pressure hydrogenation and extremely toxic reagents, uses common organic solvents like ethyl acetate and ethanol, and operates under mild thermal conditions, making it highly viable for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to deliver high-quality active pharmaceutical ingredients to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110981739B are fully realized in practical manufacturing environments. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Terbutaline Sulfate meets the highest international pharmacopoeial standards, providing our partners with absolute confidence in the safety and efficacy of their final drug products.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined process for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and your competitive position in the respiratory therapy market.
