Advanced Manufacturing of Terbutaline Sulfate: A Safer, Scalable Route for Global Pharma Supply Chains
Advanced Manufacturing of Terbutaline Sulfate: A Safer, Scalable Route for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance high purity with operational safety, particularly for critical respiratory medications like Terbutaline Sulfate. Patent CN113045437A introduces a groundbreaking preparation method that fundamentally restructures the synthesis of this vital beta-2 adrenergic agonist. By utilizing 3,5-dihydroxyacetophenone as a starting material, this novel process achieves direct oxidation to aromatic glyoxal without the cumbersome need for protecting phenolic hydroxyl groups. This technical breakthrough addresses long-standing inefficiencies in prior art, specifically eliminating hazardous hydrogenation debenzylation units and environmentally damaging bromination reactions. For R&D directors and supply chain leaders, this represents a significant leap forward in process intensification, offering a cleaner, shorter, and more economically viable route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Terbutaline Sulfate has been plagued by convoluted synthetic routes that introduce unnecessary risk and cost. Traditional methodologies, such as those disclosed in early patents like US3937838, rely heavily on 3,5-dihydroxybenzoic acid, necessitating a lengthy sequence of esterification, benzyl protection, hydrolysis, and acylation. A critical bottleneck in these legacy processes is the reliance on diazomethane, an extremely toxic and explosive reagent that poses severe safety liabilities in a manufacturing setting. Furthermore, subsequent steps often involve bromination reactions which generate substantial hazardous waste, and hydrogenation debenzylation which requires high-pressure equipment and precious metal catalysts. These factors collectively result in low atom economy, extended lead times, and a heavy environmental footprint, rendering many conventional methods unsuitable for modern, sustainable API intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN113045437A streamlines the synthesis into a concise four-step sequence that prioritizes safety and efficiency. The core innovation lies in the direct oxidation of 3,5-dihydroxyacetophenone using a system of concentrated hydrobromic acid and dimethyl sulfoxide (DMSO). This clever chemical design allows for the formation of the key glyoxal intermediate without the need to protect the sensitive phenolic hydroxyl groups, effectively cutting out multiple protection and deprotection stages. The subsequent condensation with tert-butylamine and reduction using mild hydride agents proceeds under atmospheric pressure and moderate temperatures. This approach not only simplifies the operational workflow but also drastically reduces the generation of solid waste and toxic byproducts, positioning it as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into HBr/DMSO Oxidation and Reductive Amination
The heart of this synthetic strategy is the efficient conversion of the acetophenone derivative into the corresponding phenylglyoxal. In the first step, the reaction between 3,5-dihydroxyacetophenone and DMSO activated by HBr facilitates an alpha-oxidation of the ketone. This mechanism avoids the harsh conditions typically required for such transformations, operating effectively at temperatures between 20°C and 55°C. The absence of phenolic protection is particularly noteworthy; usually, the electron-rich aromatic ring would be susceptible to over-oxidation or side reactions, but the specific stoichiometry and solvent system employed here ensure high selectivity for the alpha-carbon. This precision minimizes the formation of polymeric tars or over-oxidized acids, ensuring a cleaner reaction profile that simplifies downstream purification.

Following the oxidation, the process moves to a condensation reaction where the newly formed glyoxal reacts with tert-butylamine. This step forms an imine intermediate, specifically 2-(tert-butylimino)-1-(3,5-dihydroxyphenyl)ethane-1-ketone. The choice of solvent, preferably isopropyl acetate, plays a crucial role in driving the equilibrium towards the imine while maintaining the solubility of the polar intermediates. The subsequent reduction step utilizes sodium borohydride or potassium borohydride in alcoholic solvents like methanol or ethanol. This reduction is chemoselective, targeting both the ketone and the imine functionalities to yield the amino-alcohol backbone of Terbutaline. The mild nature of borohydride reduction ensures that the stereochemistry is managed effectively without the need for chiral catalysts at this stage, relying instead on the inherent stability of the transition states.
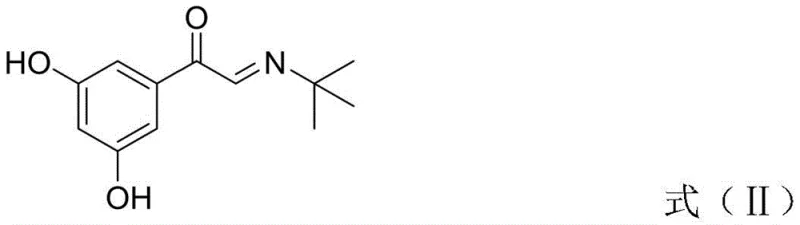
Impurity control is inherently built into this mechanism by avoiding the use of heavy metal catalysts often found in hydrogenation steps. In traditional routes, residual palladium or platinum from debenzylation can be difficult to remove to ppm levels required for injectables or inhalants. By utilizing a purely organic oxidation and hydride reduction pathway, the metal impurity profile is significantly cleaner. Furthermore, the avoidance of bromine gas or liquid bromine in favor of hydrobromic acid in a controlled oxidation system reduces the risk of ring bromination, a common side reaction that creates hard-to-remove halogenated impurities. This results in a crude product with a higher purity profile, reducing the burden on crystallization and recrystallization steps.
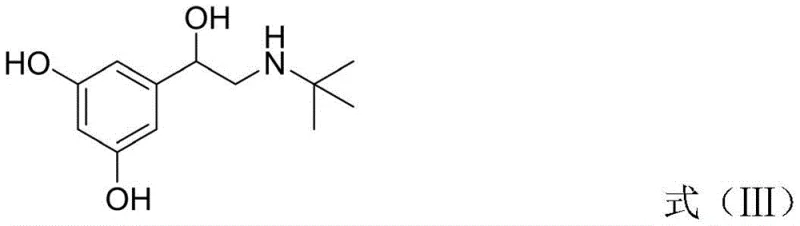
How to Synthesize Terbutaline Sulfate Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the novel route. The process begins with the dissolution of the starting material in DMSO, followed by the controlled addition of hydrobromic acid to initiate the oxidation. Once the glyoxal intermediate is isolated, it is immediately subjected to condensation with tert-butylamine in a water-immiscible ester solvent to facilitate easy workup. The final reduction and salification steps are designed to be telescoped where possible, minimizing solvent swaps and handling time. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Oxidize 3,5-dihydroxyacetophenone using concentrated HBr and DMSO to obtain 3,5-dihydroxyphenylglyoxal.
- Perform condensation with tert-butylamine in isopropyl acetate to form the imine intermediate.
- Reduce the carbonyl and imine groups using sodium borohydride in ethanol to yield the free base.
- Salify the free base with sulfuric acid at pH 4-6 to crystallize pure Terbutaline Sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing 3,5-dihydroxyacetophenone, a commodity chemical, and avoiding specialized reagents like diazomethane or selenium dioxide, the process mitigates the risk of supply disruptions caused by the scarcity of exotic precursors. Additionally, the elimination of high-pressure hydrogenation units means that production can be outsourced to a wider range of CDMOs that do not possess specialized high-pressure infrastructure, thereby increasing competition and potentially lowering manufacturing costs. The reduced step count directly correlates to lower labor costs and reduced utility consumption per kilogram of finished API.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive protection and deprotection sequences. In traditional synthesis, the reagents required for benzyl protection and the subsequent hydrogenolytic removal add significant material costs and extend cycle times. By bypassing these steps entirely, the new method achieves a substantial reduction in raw material expenditure. Furthermore, the use of common solvents like DMSO, isopropyl acetate, and ethanol replaces more expensive or regulated solvents, contributing to a leaner cost structure. The overall yield improvement resulting from fewer unit operations also means less waste of valuable starting materials, enhancing the gross margin potential for the final product.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions. The process operates at near-ambient temperatures and atmospheric pressure, reducing the likelihood of equipment failure or batch loss due to thermal runaways or pressure breaches. The raw materials involved are widely available in the global chemical market, reducing dependency on single-source suppliers for niche reagents. This stability ensures consistent production schedules and reliable delivery timelines, which is critical for maintaining inventory levels of essential respiratory medications. The simplified workflow also allows for faster batch turnover, enabling manufacturers to respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The avoidance of elemental bromine and the minimization of brominated waste streams significantly lower the cost and complexity of wastewater treatment. The absence of heavy metal catalysts eliminates the need for costly scavenging resins and extensive testing for metal residues. This 'green' profile aligns with increasingly stringent global environmental regulations, reducing the risk of regulatory shutdowns or fines. The process is inherently scalable, as the exothermic profiles of the oxidation and reduction steps are manageable in large reactors, facilitating a smooth transition from pilot plant to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Terbutaline Sulfate synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield expectations, safety protocols, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this new route improve safety compared to traditional Terbutaline synthesis?
A: This method eliminates the need for high-risk hydrogenation debenzylation and avoids the use of toxic bromination reagents and diazomethane, significantly reducing operational hazards and environmental pollution.
Q: What are the key yield advantages of the direct oxidation strategy?
A: By bypassing protection and deprotection steps for phenolic hydroxyl groups, the process shortens the reaction sequence, leading to improved overall atom economy and reduced material loss typically associated with multi-step purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (20°C to 55°C), utilize readily available solvents like DMSO and isopropyl acetate, and avoid high-pressure equipment, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for essential medicines like Terbutaline Sulfate. Our technical team has thoroughly analyzed the pathway described in CN113045437A and validated its potential for high-volume production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of monitoring the specific impurity profiles associated with this oxidation-reduction sequence, guaranteeing stringent purity specifications that meet or exceed international pharmacopoeia standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By optimizing this route further, we can deliver significant value through improved cost structures and supply security. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot batches and conduct comprehensive route feasibility assessments to demonstrate how we can become your preferred partner for high-quality Terbutaline Sulfate.
