Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
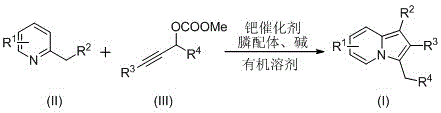
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly indolizine scaffolds which serve as critical cores in numerous bioactive molecules. Patent CN110357879B, published in April 2021, introduces a significant technological advancement in the preparation of C-3 benzyl indolizine compounds, addressing long-standing challenges in yield and operational simplicity. This innovation leverages a palladium-catalyzed coupling strategy that transforms readily available 2-alkylpyridines and propargyl carbonates into complex heterocyclic structures with high efficiency. For R&D directors and procurement managers alike, this methodology represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates. The ability to access these structurally diverse compounds through a one-pot reaction sequence eliminates the need for multi-step protections and deprotections often associated with traditional heterocycle synthesis.
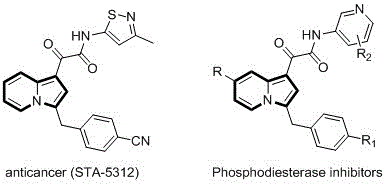
Indolizine derivatives are renowned for their potent biological activities, ranging from antitumor properties to phosphodiesterase inhibition, making them indispensable in modern drug discovery pipelines. The structural versatility offered by the C-3 benzyl substitution pattern allows for extensive SAR (Structure-Activity Relationship) studies, enabling medicinal chemists to fine-tune pharmacokinetic profiles. However, the historical reliance on inefficient synthetic pathways has often bottlenecked the supply of these critical intermediates. The disclosed method in CN110357879B circumvents these issues by employing a transition metal-catalyzed annulation that tolerates a wide array of functional groups, including esters, cyano groups, and various aryl substituents. This broad substrate scope is essential for a reliable pharmaceutical intermediate supplier aiming to support diverse drug development programs without compromising on purity or delivery timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C-3 substituted indolizines has been plagued by significant operational hazards and chemical inefficiencies. Prior art methods frequently relied on the reaction of 2-vinylpyridine with chlorocarbene under rigorous heating or irradiation conditions. This traditional approach suffers from severe limitations, including poor applicability regarding substituent groups and consistently low yields that render it economically unviable for large-scale production. Furthermore, the generation and handling of chlorocarbene intermediates pose substantial safety risks in an industrial setting, requiring specialized equipment and stringent safety protocols that drive up operational costs. The harsh reaction conditions often lead to the formation of complex impurity profiles, necessitating laborious purification steps that further erode profit margins and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a palladium-catalyzed cyclization of 2-alkylpyridines with propargyl carbonates, offering a streamlined and safer alternative. This reaction proceeds smoothly in polar organic solvents such as dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) at temperatures between 100°C and 140°C. The use of propargyl carbonates as electrophilic partners not only enhances the reaction rate but also improves the overall atom economy compared to carbene-based routes. By simply mixing the metal catalyst, phosphine ligand, substrates, and a mild base like potassium carbonate, manufacturers can achieve high conversion rates with minimal byproduct formation. This operational simplicity translates directly into reduced manufacturing costs and enhanced supply chain reliability, as the process does not require exotic reagents or extreme pressure conditions.
Mechanistic Insights into Pd-Catalyzed Cyclization
The success of this transformation hinges on the precise orchestration of the palladium catalytic cycle, which facilitates the construction of the indolizine core through a series of well-defined organometallic steps. The reaction initiates with the oxidative addition of the zero-valent palladium species into the propargyl carbonate, generating a reactive pi-allyl palladium intermediate. This is followed by the nucleophilic attack of the 2-alkylpyridine nitrogen or the subsequent C-H activation at the methyl group, depending on the specific mechanistic pathway favored by the ligand environment. The presence of bulky bidentate phosphine ligands, such as 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), plays a crucial role in stabilizing the active palladium species and promoting the reductive elimination step that closes the five-membered ring. Understanding these mechanistic nuances allows process chemists to optimize catalyst loading and ligand ratios to maximize turnover numbers.
Furthermore, the choice of base and solvent significantly influences the impurity profile and the final purity of the C-3 benzyl indolizine product. The patent highlights that using cesium carbonate or potassium carbonate in polar aprotic solvents effectively suppresses side reactions such as polymerization of the alkyne moiety or hydrolysis of the ester groups. This control over the reaction environment ensures that the resulting crude material is of sufficient quality for standard column chromatography purification, avoiding the need for preparative HPLC which is costly and difficult to scale. For R&D teams focused on impurity control, this method offers a predictable pathway where major impurities can be easily identified and minimized through slight adjustments in reaction temperature or time, ensuring compliance with stringent regulatory standards for API intermediates.
How to Synthesize C-3 Benzyl Indolizine Efficiently
To implement this synthesis effectively, operators must adhere to specific stoichiometric ratios and thermal profiles outlined in the patent data. The process begins by charging a reaction vessel with the zero-valent palladium catalyst, the selected phosphine ligand, the 2-alkylpyridine substrate, the propargyl carbonate derivative, and the inorganic base. The mixture is then suspended in an appropriate volume of organic solvent, typically 10-15 mL per mmol of limiting reagent, to ensure adequate solubility and heat transfer. Heating the system to the optimized temperature range initiates the catalytic cycle, which generally requires 10 to 20 hours to reach full conversion. Detailed standardized synthesis steps follow below.
- Charge a reaction vessel with zero-valent palladium catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in an organic solvent such as DMSO or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit lies in the significant reduction of raw material costs, as both 2-alkylpyridines and propargyl carbonates are commodity chemicals available from multiple global suppliers. This abundance prevents supply bottlenecks and allows for competitive pricing strategies, ensuring cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality. Additionally, the elimination of hazardous chlorocarbene reagents simplifies the regulatory compliance landscape, reducing the administrative burden and insurance costs associated with handling dangerous substances. The straightforward workup procedure, involving simple filtration and silica gel treatment, minimizes solvent consumption and waste generation, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and a highly efficient catalyst system that operates at low loadings. By avoiding expensive specialty reagents and complex multi-step sequences, the overall cost of goods sold (COGS) is drastically lowered. The high yields reported in the patent examples indicate that very little raw material is wasted, further enhancing the economic efficiency of the production line. Moreover, the ability to use common solvents like DMF and DMSO, which are easily recovered and recycled in industrial settings, contributes to substantial long-term savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which tolerate minor variations in temperature and reagent quality without catastrophic failure. The use of stable solid reagents and catalysts simplifies logistics and storage requirements, reducing the risk of degradation during transit. Since the reaction does not rely on sensitive moisture-sensitive reagents that require cryogenic storage, the inventory management becomes more flexible and less prone to disruption. This reliability ensures consistent delivery schedules for downstream API manufacturers, fostering stronger long-term partnerships between suppliers and clients.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards typical of carbene chemistry. The moderate temperature range of 100-140°C is easily achievable with standard industrial heating jackets, removing the need for specialized high-pressure reactors. Furthermore, the simplified post-treatment reduces the volume of aqueous waste streams, making wastewater treatment more manageable and cost-effective. This environmental compatibility is increasingly critical for maintaining operating licenses and meeting the sustainability goals of major pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process development teams. Understanding these specifics is vital for ensuring successful technology transfer and scale-up operations.
Q: What are the key advantages of this Pd-catalyzed method over traditional chlorocarbene routes?
A: Unlike traditional methods requiring hazardous chlorocarbene and harsh conditions, this novel approach utilizes readily available propargyl carbonates and 2-alkylpyridines under milder thermal conditions, resulting in higher yields and simpler post-processing.
Q: Which ligands and catalysts are optimal for this transformation?
A: The patent specifies zero-valent palladium sources such as tris(dibenzylideneacetone)dipalladium or tetrakis(triphenylphosphine)palladium, paired with bidentate phosphine ligands like Xantphos derivatives to maximize catalytic efficiency.
Q: Is this process suitable for large-scale manufacturing of anticancer intermediates?
A: Yes, the process demonstrates high substrate tolerance and uses common organic solvents like DMSO and DMF, making it highly adaptable for commercial scale-up with straightforward purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-3 Benzyl Indolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering C-3 benzyl indolizine compounds with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies like the one described in CN110357879B, we can offer customized synthesis solutions that optimize both cost and timeline for your specific application.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities can support your pipeline. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to our optimized manufacturing processes. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions for your supply chain strategy. Partner with us to secure a reliable source of complex heterocyclic intermediates that drive innovation in the pharmaceutical industry.
