Advanced Thioxanthone-Based Oxime Esters for High-Performance Visible Light Curing
The landscape of photochemical synthesis and polymer curing is undergoing a significant transformation, driven by the urgent demand for efficient, visible-light-active catalysts that minimize environmental impact while maximizing performance. Patent CN115109028B introduces a groundbreaking class of oxime ester compounds derived from the thioxanthone scaffold, representing a pivotal advancement in the field of fine chemical intermediates. Unlike traditional photoinitiators that often rely on harsh UV conditions or unstable radical precursors, this novel technology leverages a robust conjugated system to generate highly reactive anthrone benzyl free radicals under mild visible light irradiation. For R&D directors and procurement specialists alike, this patent outlines a pathway to materials that not only exhibit superior catalytic activity in olefin addition reactions but also offer a streamlined, cost-effective manufacturing route. The strategic integration of these compounds into supply chains promises to enhance the efficiency of polymer additive production, providing a reliable foundation for next-generation coating and adhesive technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of effective photoinitiators has been plagued by significant technical hurdles, particularly regarding spectral sensitivity and thermal stability. Conventional oxime esters reported in prior art often suffer from insufficient absorption in the visible light spectrum, necessitating the use of high-energy UV sources that can damage sensitive substrates or pose safety risks in industrial settings. Furthermore, many existing compounds exhibit poor stability during storage or processing, leading to premature decomposition and inconsistent curing performance. From a supply chain perspective, the synthesis of these legacy materials frequently involves complex multi-step sequences with low overall yields, driving up the cost of goods sold and creating bottlenecks in procurement. The reliance on expensive transition metal catalysts or difficult-to-handle reagents further complicates the manufacturing process, introducing impurities that require rigorous and costly purification steps to meet the stringent quality standards demanded by high-performance applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN115109028B presents a refined synthetic strategy that directly addresses these inefficiencies through the design of a thioxanthone-based core. This innovative approach utilizes a straightforward acylation followed by an oxime coupling reaction, eliminating the need for exotic catalysts and enabling operation under mild thermal conditions. The resulting compounds possess a larger conjugated structure that dramatically expands the absorption window into the visible range, specifically demonstrating strong efficacy around 405 nanometers. 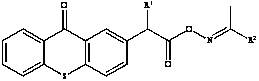 This structural optimization not only enhances photocatalytic activity but also improves the solubility profile of the final product, ensuring seamless integration into diverse resin matrices. By simplifying the reaction pathway and utilizing readily available starting materials such as thionyl chloride and acetophenone oxime, this novel method significantly lowers the barrier to entry for large-scale production, offering a compelling value proposition for manufacturers seeking to optimize their cost structures without compromising on performance metrics.
This structural optimization not only enhances photocatalytic activity but also improves the solubility profile of the final product, ensuring seamless integration into diverse resin matrices. By simplifying the reaction pathway and utilizing readily available starting materials such as thionyl chloride and acetophenone oxime, this novel method significantly lowers the barrier to entry for large-scale production, offering a compelling value proposition for manufacturers seeking to optimize their cost structures without compromising on performance metrics.
Mechanistic Insights into Visible-Light-Induced Radical Generation
The exceptional performance of these thioxanthone oxime esters can be attributed to their unique electronic configuration, which facilitates efficient homolytic cleavage of the nitrogen-oxygen bond upon photon absorption. When exposed to visible light, the excited state of the molecule undergoes a rapid fragmentation process, generating an anthrone benzyl free radical alongside an iminyl radical. This specific radical species is characterized by higher catalytic activity compared to standard aromatic or aliphatic radicals, enabling it to initiate olefin addition reactions with remarkable speed and efficiency. The thioxanthone moiety acts as a potent chromophore, effectively harvesting light energy and transferring it to the reactive center, thereby maximizing the quantum yield of the initiation process. This mechanism ensures that even at low concentrations, typically around 1 mol%, the compound can drive high conversion rates in polymerization reactions, as evidenced by real-time infrared monitoring showing double bond conversions reaching up to 75% within seconds of irradiation.
Furthermore, the stability of the generated radicals plays a crucial role in the overall efficacy of the curing process. Electron spin resonance studies confirm the formation of stable methylene free radicals with distinct hyperfine splitting constants, indicating a controlled and sustained release of active species. 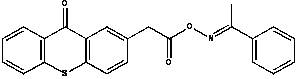 This controlled generation minimizes side reactions and ensures a uniform cure throughout the material depth, which is critical for applications requiring precise mechanical properties. The ability to tune the R groups on the oxime and the thioxanthone backbone allows chemists to fine-tune the absorption maxima and radical reactivity, providing a versatile platform for customizing photoinitiators for specific industrial needs. Such mechanistic clarity offers R&D teams the confidence to integrate these intermediates into complex formulations, knowing that the underlying chemistry is robust, predictable, and scalable.
This controlled generation minimizes side reactions and ensures a uniform cure throughout the material depth, which is critical for applications requiring precise mechanical properties. The ability to tune the R groups on the oxime and the thioxanthone backbone allows chemists to fine-tune the absorption maxima and radical reactivity, providing a versatile platform for customizing photoinitiators for specific industrial needs. Such mechanistic clarity offers R&D teams the confidence to integrate these intermediates into complex formulations, knowing that the underlying chemistry is robust, predictable, and scalable.
How to Synthesize Thioxanthone Oxime Esters Efficiently
The synthesis protocol detailed in the patent provides a clear, reproducible roadmap for producing these high-value intermediates with excellent purity and yield. The process begins with the activation of the carboxylic acid precursor, followed by a coupling step that requires careful control of temperature and stoichiometry to maximize efficiency. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different production batches.
- Activate the thioxanthone carboxylic acid precursor using thionyl chloride or oxalyl dichloride in an ice-water bath, then warm to room temperature to form the acid chloride intermediate.
- React the crude acid chloride with specific oximes (such as acetophenone oxime) in the presence of triethylamine in dichloromethane at low temperatures.
- Purify the resulting oxime ester precipitate via recrystallization using a petroleum ether and ethyl acetate mixed solvent system to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology translates into tangible operational improvements and risk mitigation. The synthesis route described avoids the use of precious metal catalysts, which are subject to volatile market pricing and supply constraints, thereby stabilizing the cost base for raw materials. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a more sustainable and economical manufacturing footprint. The high yields reported in the examples demonstrate a process that is not only chemically efficient but also commercially viable, minimizing waste generation and maximizing throughput. These factors collectively enhance the reliability of the supply chain, ensuring that critical photoinitiator components are available when needed without the delays often associated with complex chemical syntheses.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like thionyl chloride and triethylamine significantly lower the direct material costs associated with production. The process operates at near-ambient temperatures after an initial cooling phase, reducing the energy load required for heating or cryogenic cooling, which further drives down utility expenses. Moreover, the high isolated yields, consistently exceeding 85% across multiple examples, mean that less raw material is wasted, improving the overall atom economy of the process. This economic efficiency allows for competitive pricing strategies while maintaining healthy margins, making it an attractive option for cost-sensitive applications in the coatings and adhesives sectors.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that the supply chain is resilient against disruptions that often affect specialized reagents. The simplicity of the two-step synthesis allows for flexible manufacturing scheduling, enabling producers to respond quickly to fluctuations in demand. Purification is achieved through straightforward recrystallization or precipitation techniques, avoiding the need for complex chromatographic separations that can bottleneck production lines. This operational simplicity translates to shorter lead times and a more dependable supply of high-purity intermediates, giving downstream customers greater certainty in their own production planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The use of common solvents like dichloromethane and petroleum ether, which can be recovered and recycled, aligns with modern environmental compliance standards and reduces the burden of hazardous waste disposal. The absence of heavy metal residues in the final product simplifies regulatory approval for end-use applications, particularly in industries with strict toxicity limits. This combination of scalability and environmental stewardship positions the technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxime ester technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this thioxanthone oxime ester over traditional photoinitiators?
A: The primary advantage lies in its extended conjugated structure, which provides a wider spectral absorption range (200 to 500 nanometers) and significantly stronger absorption in the visible light region compared to conventional aryl or aliphatic radicals.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes simple, economically viable reagents like thionyl chloride and common oximes, operating under mild conditions (ice bath to room temperature) with high yields ranging from 85% to 93%, making it highly suitable for commercial scale-up.
Q: How does the solubility of this compound compare to other anthrone derivatives?
A: This specific oxime ester exhibits superior solubility due to its smaller molecular size and optimized structural properties, allowing for better dissolution in various resin systems compared to bulkier anthrone-based compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxime Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the thioxanthone-based oxime esters described in CN115109028B and are fully equipped to bring this innovation to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot scale to full industrial realization. Our state-of-the-art facilities are designed to handle the specific requirements of photochemical intermediate synthesis, including strict light-control protocols and advanced purification capabilities. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of quality and consistency, providing you with a reliable source of high-performance photoinitiators for your most demanding applications.
We invite you to collaborate with us to optimize your supply chain and unlock new possibilities in your product development. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. By partnering with us, you gain access to deep technical expertise and a commitment to excellence that sets us apart in the industry. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
