Advanced Catalytic Oxidation for High-Purity Triazolethione Derivatives and Commercial Scale-Up
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of key agrochemical intermediates, particularly triazolethione derivatives which serve as critical precursors for compounds like prothioconazole. A pivotal advancement in this domain is detailed in Chinese Patent CN108912062B, which discloses a novel preparation method that fundamentally shifts the paradigm from stoichiometric oxidation to efficient catalytic aerobic oxidation. This technology addresses long-standing challenges in the industry, specifically regarding yield optimization, waste reduction, and process safety. By leveraging a controlled oxygen atmosphere combined with transition metal catalysts, the patented process achieves exceptional conversion rates while operating under remarkably mild conditions. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic superiority of this route is essential for securing a competitive supply chain. The following analysis dissects the technical nuances of this invention, highlighting its potential to redefine cost structures and quality standards in fungicide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(1-chloro-cyclopropan-1-yl)-1-(2-chloro-phenyl)-3-(4,5-dihydro-1,2,4-triazol-5-thiocarbonyl-1-yl)-propan-2-ol has been plagued by inefficient and environmentally burdensome protocols. One early approach, disclosed in WO1999/18087A1, relied on the reaction of the triazolidine precursor with oxygen in the presence of sulfur powder and potassium hydroxide. As illustrated in the reaction scheme below, this method necessitated elevated temperatures to drive conversion, which unfortunately triggered significant peroxidation side reactions. These harsh conditions often led to the dissociation of sulfur from the target molecule, resulting in poor yields and a crude product that was notoriously difficult to purify to pharmaceutical or agrochemical grade standards.
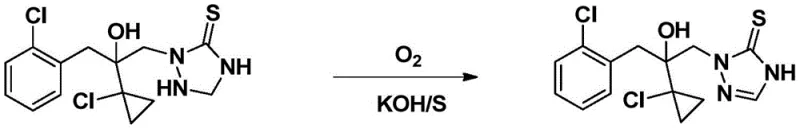
Subsequent attempts to improve this process, such as the method described in WO2001/46158A1, shifted towards using ferric chloride as an oxidant. However, this approach required stoichiometric or even excess amounts of iron salts—often two molar equivalents relative to the substrate. While this mitigated some thermal issues, it introduced a massive waste burden. The reaction generated substantial quantities of acidic wastewater and solid waste containing Fe2+ and Fe3+ ions, creating severe environmental compliance hurdles and escalating disposal costs. Furthermore, the separation of the target product from the large excess of iron salts proved technically challenging, often requiring extensive washing and purification steps that eroded overall process efficiency.
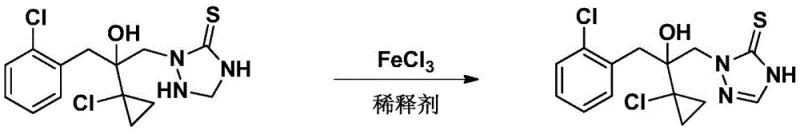
More recent literature, such as a 2017 publication in 'Pesticides', attempted to reduce catalyst loading by using catalytic amounts of anhydrous ferric chloride under open-air stirring. Despite reducing iron usage, this method suffered from inherently slow reaction kinetics due to the low partial pressure of oxygen in ambient air. To achieve full conversion, reaction times extended up to seven days in scale-up trials, with yields plummeting to as low as 72%. Additionally, the requirement for anhydrous solvents and catalysts added unnecessary complexity and cost, as any moisture ingress could deactivate the catalyst or alter the reaction pathway, making this method unsuitable for robust industrial application.
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN108912062B introduces a highly efficient catalytic system that utilizes high-concentration oxygen gas (50%-100%) in conjunction with minimal amounts of transition metal catalysts. The core innovation lies in the synergistic effect between the enriched oxygen environment and catalysts such as FeCl3, CuCl2, or CoCl2. By increasing the oxygen concentration, the reaction kinetics are dramatically accelerated, allowing the process to proceed rapidly at mild temperatures ranging from 10°C to 65°C, with optimal results often observed at ambient conditions (10-20°C). This eliminates the energy-intensive heating steps required by sulfur-based methods and avoids the thermal degradation of sensitive functional groups.

Furthermore, this novel approach drastically reduces the catalyst loading to a range of 0.5 mol% to 8.5 mol%, a significant improvement over the stoichiometric requirements of prior art. The use of common organic solvents like methanol or ethyl acetate, which do not require rigorous anhydrous treatment, further simplifies the operational protocol. The result is a clean, high-yielding process that consistently delivers the target triazolethione derivative in yields exceeding 95%, often reaching up to 99.3%. This leap in efficiency not only enhances the economic viability of the synthesis but also aligns with modern green chemistry principles by minimizing heavy metal waste and solvent consumption.
Mechanistic Insights into FeCl3-Catalyzed Aerobic Oxidation
The chemical transformation at the heart of this patent involves the oxidative dehydrogenation of the 4,5-dihydro-1,2,4-triazolidine ring to form the aromatic 1,2,4-triazole ring. In the presence of a transition metal catalyst like ferric chloride (FeCl3), the reaction likely proceeds through a single-electron transfer mechanism. The Fe(III) species acts as a Lewis acid and an oxidant, coordinating with the nitrogen atoms of the triazolidine ring to facilitate the removal of hydrogen atoms. Under normal atmospheric conditions, the regeneration of the active Fe(III) species from the reduced Fe(II) state is slow, limiting the turnover number of the catalyst. However, by introducing a gas stream with 50%-100% oxygen concentration, the re-oxidation of Fe(II) back to Fe(III) is kinetically favored, effectively closing the catalytic cycle and sustaining high reaction rates throughout the process.
This mechanistic efficiency is critical for impurity control. In traditional methods involving sulfur or excessive heat, the thione group (-C=S) is vulnerable to nucleophilic attack or thermal decomposition, leading to desulfurized byproducts that are structurally similar to the target and difficult to remove. The mild, oxygen-driven oxidation of the new method preserves the integrity of the thione moiety while selectively aromatizing the heterocyclic ring. The precise control over oxygen partial pressure ensures that the oxidation stops at the desired triazole stage without progressing to over-oxidized species such as sulfones or N-oxides. This selectivity is paramount for achieving the high purity levels (>97.5%) required for downstream coupling reactions in the synthesis of final fungicide active ingredients.
How to Synthesize Triazolethione Derivatives Efficiently
Implementing this advanced synthesis route requires careful attention to gas-liquid mass transfer and catalyst selection to maximize the benefits of the patented technology. The process is designed to be operationally simple, avoiding the need for specialized anhydrous setups or extreme temperature controls, which makes it highly attractive for commercial adoption. Operators can utilize standard stainless steel reactors equipped with gas spargers to ensure efficient dissolution of the oxygen-rich gas mixture into the solvent phase. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational protocol for process development teams aiming to replicate these high-yield results in a pilot or production setting.
- Prepare the reaction mixture by combining the triazolidine precursor (Formula II) with a transition metal catalyst (e.g., FeCl3) in an organic solvent such as methanol.
- Introduce a gas mixture containing 50%-100% oxygen into the reactor while maintaining the temperature between 10°C and 65°C.
- Monitor the reaction progress via HPLC until completion (typically 8-15 hours), then isolate the product through crystallization or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic aerobic oxidation method represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of stoichiometric oxidants and the reduction in catalyst loading directly translate to lower raw material costs and simplified logistics. Unlike methods requiring sulfur powder or large quantities of iron salts, this process utilizes widely available industrial gases and commodity chemicals, reducing the risk of supply bottlenecks. Furthermore, the mild reaction conditions and reduced solvent volumes decrease the energy footprint of the manufacturing process, contributing to significant operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The drastic reduction in catalyst usage from stoichiometric amounts to as low as 0.5 mol% fundamentally alters the cost equation. By avoiding the purchase and subsequent disposal of tons of iron waste, manufacturers can realize substantial cost savings in both raw materials and waste treatment. Additionally, the ability to use non-anhydrous solvents like commercial-grade methanol removes the need for expensive solvent drying processes, further lowering the cost of goods sold (COGS) without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of this method against moisture and its operation at near-ambient temperatures make it less susceptible to batch failures caused by environmental fluctuations or equipment limitations. This reliability ensures consistent output volumes, allowing supply chain planners to maintain tighter inventory controls and meet delivery commitments with greater confidence. The use of standard equipment and common reagents also means that production can be easily scaled or transferred between facilities without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner alternative to legacy methods. The minimization of heavy metal waste streams simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. The absence of sulfur powder eliminates the risk of hydrogen sulfide generation and associated safety hazards. These factors collectively enhance the sustainability profile of the manufacturing site, ensuring long-term compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazolethione synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: What are the primary advantages of using high-concentration oxygen in this synthesis?
A: Using oxygen concentrations between 50% and 100% significantly accelerates the reaction rate and allows for a drastic reduction in catalyst loading (down to 0.5 mol%), minimizing metal waste and simplifying downstream purification compared to traditional stoichiometric methods.
Q: How does this method improve product purity compared to prior art?
A: By avoiding harsh conditions like high temperatures or sulfur powder usage found in older methods, this process prevents sulfur dissociation and over-oxidation, achieving yields of 95%-99% with minimal byproduct formation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method operates at mild temperatures (10-20°C) and uses common solvents like methanol without requiring rigorous anhydrous conditions, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolethione Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of technologies like CN108912062B are fully realized in practical manufacturing environments. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of triazolethione derivative meets the exacting standards required by global agrochemical formulators.
We invite you to collaborate with us to leverage this innovative synthesis route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this catalytic method can reduce your overall manufacturing expenses. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and cost-effective supply of high-purity agrochemical intermediates.
