Advanced Palladium-Catalyzed Carbonylation for Scalable Diaryl Ketone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable Diaryl Ketone Production
The chemical industry is constantly seeking more efficient pathways to synthesize complex structural motifs that serve as the backbone for pharmaceuticals and agrochemicals. Patent CN110483223B introduces a groundbreaking method for the preparation of diaryl ketone compounds utilizing a highly efficient pyridine-palladium catalytic system. This technology leverages aryl benzene boronic acids, iodobenzene derivatives, and carbon monoxide as primary feedstocks, operating under mild conditions with potassium carbonate or potassium hydroxide as the base. The innovation lies in the specific use of pyridine-palladium or 3-chloropyridine-palladium complexes, which demonstrate exceptional catalytic activity and stability against air exposure. By optimizing reaction parameters such as temperature and CO pressure, this process achieves high yields while maintaining a simplified operational protocol that is highly attractive for industrial scale-up.
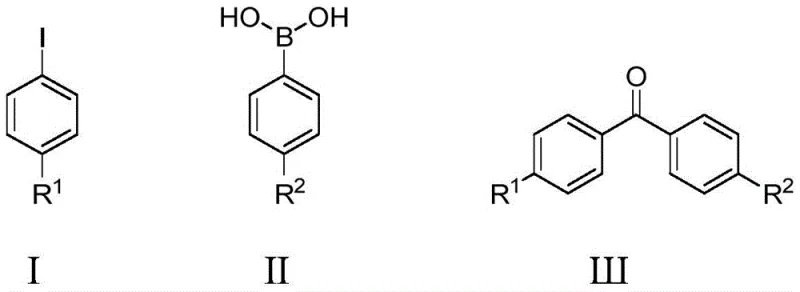
This synthetic route represents a significant advancement over conventional methods by addressing critical pain points related to atom economy and reagent toxicity. The use of carbon monoxide as a carbonyl source is particularly strategic, as it is a cost-effective C1 building block readily available from natural gas or biomass processing. Furthermore, the aryl boronic acid partners employed in this reaction are generally non-toxic and exhibit remarkable stability in the presence of air and moisture, contrasting sharply with more sensitive organometallic reagents like Grignard or organolithium species. The combination of these stable starting materials with a robust catalyst system creates a reliable supply chain foundation for producing high-purity diaryl ketones essential for drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl ketones has relied heavily on Friedel-Crafts acylation, a process that suffers from significant drawbacks regarding environmental impact and selectivity. The traditional approach necessitates the use of stoichiometric or excess amounts of Lewis acids, such as aluminum chloride, which generates substantial quantities of corrosive waste and complicates downstream purification efforts. Moreover, Friedel-Crafts reactions often struggle with regioselectivity, particularly when attempting to functionalize substituted aromatic rings, leading to mixtures of ortho and para isomers that require energy-intensive separation techniques. Alternative transition metal-mediated methods have also faced challenges, specifically regarding the failure of carbon monoxide insertion in certain substrates, which results in the formation of undesired biaryl by-products instead of the target ketone. These limitations are especially pronounced when reacting with electron-deficient aryl halides, where the electronic nature of the substrate hinders the oxidative addition or migratory insertion steps crucial for carbonylation.
The Novel Approach
The methodology disclosed in patent CN110483223B overcomes these historical barriers by employing a sophisticated palladium-catalyzed carbonylation Suzuki-Miyaura coupling strategy. This novel approach utilizes a specialized pyridine-palladium catalyst that facilitates the smooth insertion of carbon monoxide between the aryl halide and the aryl boronic acid moieties. By operating at moderate temperatures ranging from 60°C to 100°C and under controlled CO pressures of 3 to 6 atm, the reaction proceeds with high efficiency and minimal side product formation. The system is remarkably versatile, accommodating a wide range of substituents including halogens, alkoxy groups, and trifluoromethyl groups without compromising yield. This flexibility allows for the streamlined production of diverse diaryl ketone derivatives, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing where structural diversity is often required for SAR studies.
Mechanistic Insights into Pyridine-Palladium Catalyzed Carbonylation
The success of this catalytic system is deeply rooted in the unique electronic properties of the N-heterocyclic carbene (NHC) ligands coordinated to the palladium center. Unlike traditional phosphine ligands, NHC ligands possess high dissociation energies and act as strong sigma donors, which significantly strengthens the bond between the ligand and the transition metal. This robust interaction prevents the decomposition of the catalyst into inactive palladium black, a common deactivation pathway that plagues many homogeneous catalytic processes. The pyridine moiety within the catalyst structure further enhances the stability and initiation of the catalytic precursor, ensuring that the active species remains available throughout the reaction cycle. Consequently, the catalyst loading can be kept extremely low, typically between 0.1% and 0.2% relative to the substrate, without sacrificing reaction kinetics or conversion rates.
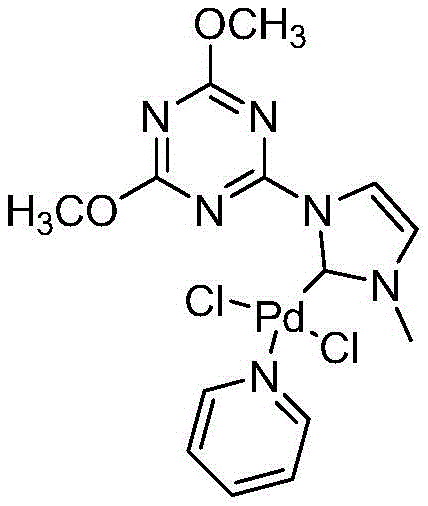
From an impurity control perspective, the mechanism favors the desired carbonylative cross-coupling over direct biaryl formation. The precise tuning of the ligand environment around the palladium atom facilitates the migratory insertion of carbon monoxide into the palladium-aryl bond before the transmetallation with the boronic acid occurs. This sequence is critical for suppressing the formation of biaryl by-products, which are often difficult to separate from the target ketone due to similar polarity. The use of inorganic bases like potassium carbonate or potassium hydroxide in solvents such as 1,4-dioxane or toluene further optimizes the transmetallation step, ensuring a clean reaction profile. This mechanistic clarity provides R&D teams with confidence in the reproducibility of the process, as the factors influencing selectivity are well-understood and controllable through standard parameter adjustments.
How to Synthesize Diaryl Ketones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward protocol that balances safety with high throughput. The process begins by charging a reaction vessel with the aryl iodide substrate, the corresponding aryl boronic acid, and the pyridine-palladium catalyst in the presence of an inorganic base and organic solvent. Once the mixture is prepared, carbon monoxide gas is introduced to establish the necessary pressure, and the system is heated to initiate the catalytic cycle. Detailed standardized synthesis steps follow below, outlining the precise molar ratios and workup procedures required to achieve the reported high yields.
- Combine aryl iodide, aryl boronic acid, pyridine-palladium catalyst (0.1-0.2 mol%), and inorganic base (K2CO3 or KOH) in an organic solvent like 1,4-dioxane or toluene.
- Introduce carbon monoxide (CO) gas into the reaction vessel and maintain a pressure of 3-6 atm while heating the mixture to 60-100°C.
- Stir the reaction for 6-8 hours, then cool to room temperature, remove solvent via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for catalysts and reagents, as the pyridine-palladium complex is air-stable and does not require the specialized handling infrastructure associated with pyrophoric or highly moisture-sensitive catalysts. This stability translates directly into reduced logistical costs and lower risks of material degradation during storage and transport. Furthermore, the ability to use inexpensive and readily available carbon monoxide as a C1 source eliminates the need for costly acyl chlorides or anhydrides, driving down the raw material cost per kilogram of the final product significantly.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the exceptionally low catalyst loading required, which is typically in the range of 0.1% to 0.2% molar equivalent. This minimal usage rate means that the expensive palladium metal contributes negligibly to the overall bill of materials, especially when considering the potential for catalyst recovery and recycling in large-scale operations. Additionally, the elimination of stoichiometric Lewis acids removes the substantial costs associated with waste disposal and neutralization of corrosive by-products. The simplified workup procedure, which often involves basic filtration and solvent removal, reduces the consumption of utilities and labor hours, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures a consistent supply of high-purity intermediates, which is critical for maintaining uninterrupted production schedules in downstream API manufacturing. Since the aryl boronic acids and iodobenzene derivatives used are commodity chemicals with established global supply chains, the risk of raw material shortage is minimized. The tolerance of the catalyst to air and moisture further mitigates the risk of batch failures due to environmental excursions, providing a more predictable and reliable production timeline. This reliability allows supply chain planners to optimize inventory levels and reduce the safety stock required for key intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the use of standard reactor equipment capable of handling moderate pressures and temperatures. The high atom economy of the carbonylation reaction means that fewer atoms end up as waste, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. The avoidance of heavy metal waste streams associated with traditional Lewis acid chemistry simplifies regulatory compliance and wastewater treatment requirements. This environmental compatibility not only reduces operational costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for process chemists. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the advantages of using pyridine-palladium catalysts over traditional phosphine ligands?
A: Pyridine-palladium catalysts, particularly those with N-heterocyclic carbene (NHC) ligands, exhibit higher dissociation energies and stronger metal-ligand bonds compared to phosphine ligands. This results in superior thermal and chemical stability, preventing the formation of inactive palladium black and ensuring consistent catalytic activity throughout the reaction.
Q: Can this method tolerate electron-deficient aryl halides?
A: Yes, unlike some traditional carbonylation reactions that struggle with electron-deficient substrates due to failed CO insertion, this patented method demonstrates excellent substrate applicability. It successfully couples various aryl iodides, including those with trifluoromethyl and halogen substituents, achieving high yields up to 98%.
Q: Is the catalyst sensitive to air and moisture?
A: One of the significant operational benefits of this technology is the air stability of the pyridine-palladium catalyst. This stability simplifies handling and storage requirements, reducing the need for stringent inert atmosphere conditions during the setup phase, which contributes to overall process robustness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development timelines. Our team of expert chemists has extensively evaluated the pyridine-palladium catalyzed carbonylation route and possesses the technical expertise to implement this technology seamlessly across various scales. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop optimization to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of diaryl ketone intermediate meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecule and volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that will demonstrate the clear competitive advantage of our manufacturing capabilities. Let us help you secure a reliable supply of high-quality intermediates while optimizing your overall production costs.
