Advanced One-Pot Synthesis of Benzopyran-2-ol Derivatives for Scalable API Manufacturing
Advanced One-Pot Synthesis of Benzopyran-2-ol Derivatives for Scalable API Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of active pharmaceutical ingredients (APIs), particularly for high-volume therapeutic areas such as urology. Patent CN101454304B discloses a groundbreaking process for the manufacture of benzopyran-2-ol derivatives, which serve as critical intermediates in the synthesis of Tolterodine and Fesoterodine, widely used muscarinic receptor antagonists for treating overactive bladder. This technology represents a significant departure from traditional multi-step syntheses by employing a novel amine-catalyzed cyclization strategy that streamlines the production workflow. The core innovation lies in the ability to construct the complex chroman ring system directly from simple phenolic precursors and trans-cinnamaldehyde in a single operational unit, thereby minimizing material handling and processing time. 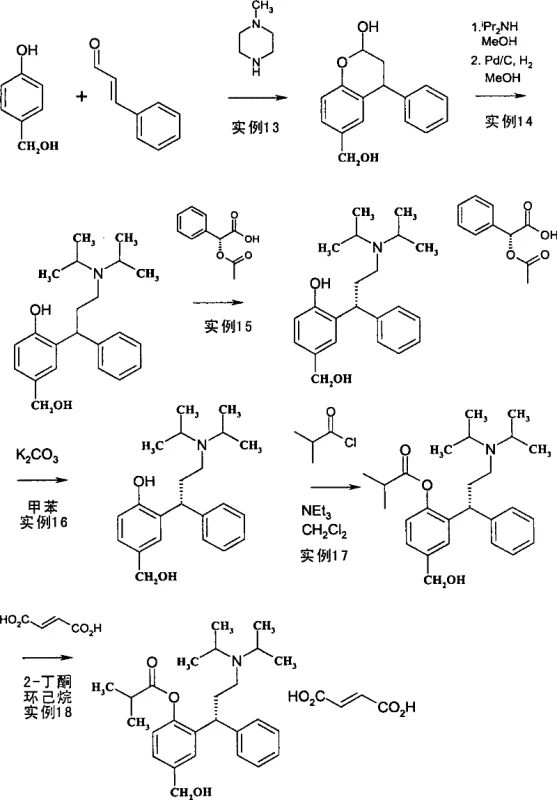
For procurement managers and supply chain directors, the implications of this patented methodology are profound, as it addresses key pain points related to raw material costs, safety hazards associated with hazardous reagents, and overall process throughput. By replacing expensive stoichiometric reducing agents with catalytic amounts of readily available secondary amines, the process inherently lowers the cost of goods sold (COGS) while enhancing the environmental profile of the manufacturing site. Furthermore, the versatility of the method allows for the synthesis of various derivatives where the substituent Y can be methyl, hydroxymethyl, or other functional groups, providing a flexible platform for producing not just Tolterodine but also its prodrug Fesoterodine and potential analogues. This adaptability ensures a reliable pharmaceutical intermediate supplier can maintain continuity of supply even as market demands shift between different salt forms or related therapeutic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 98/29402, typically rely on a multi-step sequence that begins with the condensation of p-cresol with cinnamic acid to form a lactone, followed by a critical reduction step to generate the corresponding chromen-2-ol compound. This conventional approach is heavily dependent on the use of potent and costly reducing agents like diisobutylaluminum hydride (DIBAL), sodium bis(2-methoxyethoxy)aluminum hydride, or lithium tri-tert-butoxyaluminum hydride. The reliance on these pyrophoric and moisture-sensitive reagents introduces significant safety risks and requires specialized equipment and handling protocols, which inevitably drives up capital expenditure and operational costs. Moreover, the multi-step nature of the traditional route means that yield losses accumulate at each stage, and the isolation of intermediates adds to the processing time and solvent consumption. 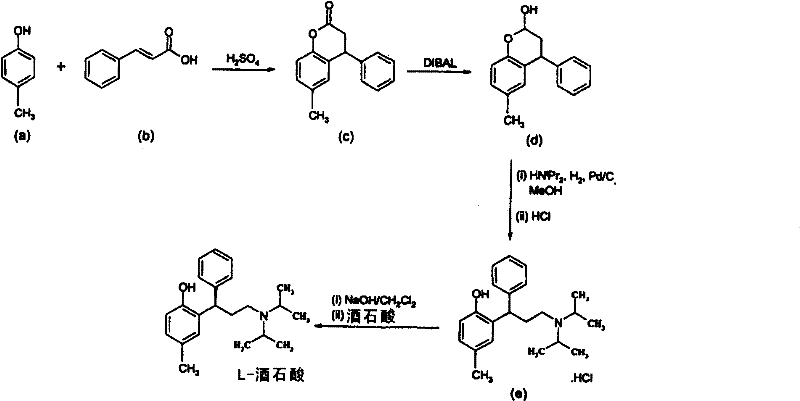
The Novel Approach
In stark contrast, the process described in CN101454304B achieves the formation of the key benzopyran-2-ol intermediate in a remarkably efficient one-pot reaction by reacting a phenolic compound directly with trans-cinnamaldehyde in the presence of a secondary amine catalyst. This innovative strategy bypasses the need for pre-forming a lactone and subsequently reducing it, effectively collapsing multiple synthetic operations into a single vessel. The use of simple secondary amines such as piperazine or N-methylpiperazine not only acts as a catalyst for the cyclization but also facilitates the formation of isolatable intermediates that are easily hydrolyzed to the desired product upon acid treatment. This simplification of the synthetic route translates directly into cost reduction in API manufacturing by eliminating the purchase and disposal of expensive aluminum-based reducing agents and reducing the overall number of unit operations required to reach the final intermediate.
Mechanistic Insights into Amine-Catalyzed Cyclization
The mechanistic foundation of this novel process involves the activation of the alpha,beta-unsaturated aldehyde (trans-cinnamaldehyde) by the secondary amine catalyst to form a reactive iminium or enamine species, which then undergoes conjugate addition with the electron-rich phenolic ring. Unlike previous methods that required strongly activated substrates or aniline nucleophiles, this invention successfully utilizes simple phenols, demonstrating the high efficacy of the amine catalysis system in promoting the 1,4-addition and subsequent intramolecular cyclization. The reaction proceeds through a transition state where the amine catalyst organizes the reactants in a specific orientation that favors the formation of the six-membered chroman ring, often yielding a mixture of diastereomers at the newly formed chiral center which can be managed in downstream processing. 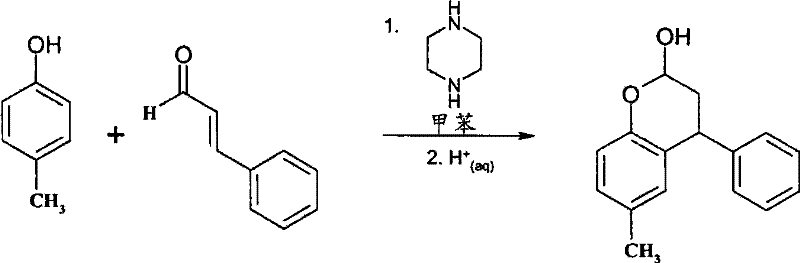
Impurity control is inherently improved in this system because the reaction conditions are milder and more selective compared to the harsh reduction conditions of the prior art. The use of Dean-Stark conditions to remove water during the reflux in toluene drives the equilibrium towards the formation of the cyclic product, minimizing the presence of open-chain aldehyde byproducts. Furthermore, the subsequent acid hydrolysis step is highly specific, cleaving the amine adducts to release the free hydroxyl group of the benzopyran-2-ol without degrading the sensitive aromatic framework. This level of chemical precision ensures that the resulting intermediate possesses high non-chiral purity, often exceeding 97% as demonstrated in the examples, which reduces the burden on purification steps and enhances the overall yield of the final active pharmaceutical ingredient.
How to Synthesize Benzopyran-2-ol Derivatives Efficiently
The practical implementation of this synthesis involves dissolving the phenolic starting material and the secondary amine catalyst in a suitable organic solvent such as toluene, followed by heating to reflux to remove residual water. Trans-cinnamaldehyde is then added slowly to the reaction mixture, maintaining the reflux temperature to ensure complete conversion while continuously removing the water generated during the condensation. After the reaction is complete, the mixture is cooled and treated with an aqueous acid solution to hydrolyze the intermediate amine adducts, followed by phase separation and washing to isolate the crude benzopyran-2-ol derivative which can be used directly in the next step or purified further.
- React a phenolic compound with trans-cinnamaldehyde in the presence of a secondary amine catalyst like piperazine or N-methylpiperazine in a solvent such as toluene under reflux with water removal.
- Treat the resulting reaction mixture with an aqueous acid, such as hydrochloric acid, to hydrolyze intermediate species and isolate the benzopyran-2-ol derivative.
- Proceed to reductive amination using diisopropylamine and hydrogenation catalysts to convert the intermediate into the final active pharmaceutical ingredient precursor.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this amine-catalyzed process offers substantial strategic benefits for organizations looking to optimize their supply chain for urology drugs. The elimination of hazardous reducing agents like DIBAL not only reduces the direct cost of raw materials but also significantly lowers the costs associated with safety compliance, waste disposal, and specialized storage infrastructure. This shift towards safer, more benign chemistry aligns with modern green chemistry principles and reduces the regulatory burden on manufacturing sites, facilitating faster approval times for new production lines. Additionally, the use of commodity chemicals like piperazine and toluene ensures a stable and reliable supply of inputs, mitigating the risk of supply chain disruptions that can occur with specialty reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of stoichiometric amounts of expensive aluminum hydrides with catalytic quantities of inexpensive secondary amines. By removing the need for cryogenic conditions or strict anhydrous environments required for DIBAL reductions, the process allows for operation in standard glass-lined or stainless steel reactors, thereby maximizing asset utilization and reducing energy consumption. The consolidation of multiple steps into a one-pot procedure further decreases labor costs and solvent usage, leading to a leaner and more cost-effective manufacturing footprint that enhances profit margins for high-volume generic production.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as p-cresol, 4-hydroxyphenethyl alcohol, and trans-cinnamaldehyde ensures that the supply chain is robust and less susceptible to geopolitical or logistical bottlenecks. The process tolerance for various substituents on the phenolic ring allows manufacturers to produce a family of related intermediates on the same equipment, providing flexibility to respond to market fluctuations in demand for Tolterodine versus Fesoterodine. This versatility strengthens the position of a reliable pharmaceutical intermediate supplier by enabling rapid scale-up and diversification of the product portfolio without significant capital investment in new technology.
- Scalability and Environmental Compliance: The reaction conditions, which operate at ambient pressure and moderate temperatures (reflux of toluene), are inherently scalable from kilogram to multi-tonne production scales without the engineering challenges associated with handling pyrophoric reagents. The simplified workup procedure, involving basic aqueous washes and crystallization, generates less hazardous waste compared to the quenching of aluminum residues, supporting sustainability goals and reducing the environmental impact of the manufacturing process. This ease of scale-up ensures that commercial production can meet global demand consistently while adhering to increasingly stringent environmental regulations regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology for the production of benzopyran-2-ol derivatives. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route.
Q: What are the primary advantages of this new process over conventional DIBAL reduction methods?
A: The novel amine-catalyzed process eliminates the need for expensive and hazardous reducing agents like DIBAL, reduces the number of synthetic steps from multiple stages to a one-pot reaction, and significantly simplifies downstream processing and waste management.
Q: Which catalysts are most effective for this benzopyran-2-ol synthesis?
A: Secondary amines containing two basic nitrogen atoms, particularly piperazine and N-methylpiperazine, have been identified as highly effective catalysts that facilitate high yields and easy hydrolysis of intermediate species.
Q: Is this process suitable for large-scale commercial production of Tolterodine?
A: Yes, the process utilizes common solvents like toluene and operates at ambient pressure with standard reflux conditions, making it highly scalable and economically viable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran-2-ol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of pharmaceutical intermediates. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards for identity, assay, and impurity profiles. By leveraging advanced technologies like the amine-catalyzed cyclization described in CN101454304B, we can offer our partners a distinct competitive advantage through superior quality and consistent supply reliability.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements for Tolterodine and Fesoterodine intermediates. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs, and let us provide you with specific COA data and route feasibility assessments that demonstrate the tangible value of partnering with a leader in fine chemical synthesis. Together, we can accelerate your drug development timelines and secure a sustainable supply chain for your vital therapeutic products.
