Advanced Biocatalytic Synthesis of Chiral Alcohols for Industrial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards sustainable and highly selective manufacturing processes, driven by the urgent need for greener synthesis routes. Patent CN114807246A introduces a groundbreaking methodology for the production of chiral alcohols, specifically leveraging advanced ketoreductase mutants to catalyze the asymmetric reduction of prochiral ketone compounds. This technology addresses critical bottlenecks in the synthesis of vital intermediates such as (R)-4-chloro-3-hydroxybutyric acid ethyl ester ((R)-CHBE), which serves as a key precursor for medications like L-carnitine. By utilizing directed evolution to engineer enzymes with superior catalytic properties, this invention offers a robust alternative to traditional chemical synthesis, promising enhanced efficiency and environmental compatibility for large-scale industrial applications.
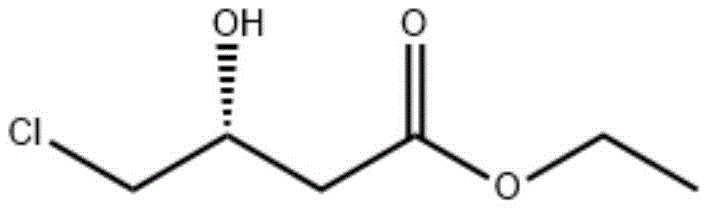
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of optically active alcohols has relied heavily on chemical asymmetric reduction methods that utilize transition metal catalysts such as rhodium and ruthenium complexes. These conventional processes are fraught with significant operational challenges, including the necessity for high-pressure hydrogenation equipment which poses safety risks and increases capital expenditure. Furthermore, chemical catalysts often struggle to achieve the stringent optical purity required for pharmaceutical-grade intermediates, frequently resulting in products with suboptimal enantiomeric excess that require costly and wasteful purification steps. The reliance on precious metals also introduces supply chain vulnerabilities and environmental concerns regarding heavy metal residue disposal, making these routes increasingly unsustainable in the context of modern green chemistry mandates.
The Novel Approach
In stark contrast, the novel biocatalytic approach detailed in the patent utilizes specifically engineered ketoreductase mutants that operate under mild, ambient conditions without the need for hazardous reagents or high-pressure infrastructure. This biological method demonstrates exceptional specificity, capable of distinguishing between enantiomers with precision that chemical catalysts cannot match, thereby yielding products with extremely high optical purity directly from the reaction vessel. The use of recombinant microorganisms expressing these optimized enzymes allows for a scalable fermentation-based production model that significantly lowers the barrier to entry for manufacturing complex chiral molecules. This shift not only simplifies the downstream processing by eliminating metal removal steps but also aligns perfectly with the industry's move towards carbon-neutral and waste-minimized manufacturing protocols.
Mechanistic Insights into Ketoreductase-Catalyzed Reduction
The core of this technological advancement lies in the rational design and directed evolution of the ketoreductase enzyme, specifically targeting amino acid residues that influence substrate binding and catalytic turnover. The patent highlights specific mutation sites such as V14, I42, A97, G170, A242, I262, and F286, which when modified, create a more accommodating active site for bulky or halogenated substrates. These structural modifications enhance the enzyme's affinity for the prochiral ketone while simultaneously stabilizing the transition state during the hydride transfer from the cofactor (NADH or NADPH) to the carbonyl group. This precise molecular engineering ensures that the reduction proceeds with strict stereocontrol, favoring the formation of the desired (R)-configuration alcohol over its (S)-counterpart through a highly organized enzyme-substrate complex.
Furthermore, the integration of an efficient cofactor regeneration system is pivotal to the economic viability of this enzymatic process. The reaction mechanism involves the oxidation of the reduced cofactor (NADH/NADPH) to its oxidized form (NAD+/NADP+), which must be continuously recycled to sustain catalytic activity without stoichiometric addition of expensive cofactors. The patent describes coupling the ketoreductase with auxiliary enzymes like glucose dehydrogenase or formate dehydrogenase, or using chemical regenerants like isopropanol, to maintain a constant pool of active reducing equivalents. This coupled system prevents the accumulation of inhibitory byproducts and ensures that the reaction kinetics remain favorable throughout the conversion, allowing for high substrate loading concentrations that are critical for industrial throughput and cost-effectiveness.
How to Synthesize Chiral Alcohol Efficiently
The implementation of this biocatalytic route requires a systematic approach to reaction setup, focusing on the optimization of pH, temperature, and enzyme loading to maximize space-time yield. The process begins with the preparation of a buffered aqueous system containing the prochiral ketone substrate, where the concentration can be adjusted dynamically based on the specific solubility and tolerance of the enzyme mutant being employed. Operators must carefully control the reaction environment to maintain the stability of the biocatalyst while ensuring sufficient mass transfer of the substrate into the aqueous phase, often aided by the presence of co-solvents or surfactants if necessary. For a comprehensive understanding of the specific operational parameters and step-by-step execution, please refer to the standardized synthesis guide provided below.
- Prepare the reaction system by mixing the prochiral ketone substrate (concentration 1-200 g/L) with a suitable buffer solution (pH 5.0-9.0) and a cofactor regeneration system.
- Add the specific ketoreductase mutant (e.g., SEQ ID NO: 1 variants) and the necessary coenzyme (NADH or NADPH) to the reaction mixture.
- Maintain the reaction temperature between 4-60°C with agitation until conversion exceeds 99%, then proceed to downstream isolation of the chiral alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits that extend far beyond simple reaction yield improvements. The elimination of precious metal catalysts removes a major cost driver and supply risk associated with fluctuating prices of rhodium and ruthenium, leading to a more predictable and stable cost structure for long-term production contracts. Additionally, the mild reaction conditions significantly reduce energy consumption related to heating, cooling, and pressurization, contributing to a lower overall carbon footprint and reduced utility costs for the manufacturing facility. These factors combined create a compelling business case for switching from legacy chemical processes to this modern biocatalytic platform.
- Cost Reduction in Manufacturing: The transition to enzyme-based catalysis fundamentally alters the cost equation by removing the need for expensive transition metal catalysts and the associated ligand systems that drive up raw material expenses. By operating at ambient temperatures and atmospheric pressure, the process drastically reduces energy consumption and eliminates the requirement for specialized high-pressure reactor vessels, thereby lowering both capital investment and operational expenditures. Furthermore, the high stereoselectivity minimizes the formation of unwanted isomers, reducing the burden on downstream purification units and increasing the overall yield of the saleable product, which directly enhances profit margins.
- Enhanced Supply Chain Reliability: Relying on biologically derived catalysts produced via fermentation decouples the supply chain from the geopolitical and mining-related instabilities that often plague the availability of rare earth metals and precious catalysts. The ability to produce the enzyme in-house or source it from stable biological suppliers ensures a consistent and renewable supply of the critical catalytic component, mitigating the risk of production stoppages due to raw material shortages. Moreover, the robustness of the engineered mutants allows for more flexible logistics and storage conditions, ensuring that the catalyst remains active and effective throughout the distribution network.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the patent demonstrating efficacy at high substrate concentrations that facilitate efficient use of reactor volume and reduce solvent waste generation. The aqueous nature of the reaction medium and the biodegradability of the enzymatic components simplify wastewater treatment and ensure compliance with increasingly stringent environmental regulations regarding heavy metal discharge and volatile organic compound emissions. This environmental compatibility not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key differentiator in securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ketoreductase technology in industrial settings. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this biocatalytic route into existing manufacturing workflows.
Q: What represents the primary advantage of this ketoreductase mutant over wild-type enzymes?
A: The engineered mutants exhibit significantly improved enzyme activity and thermal stability, allowing for higher substrate concentrations (up to 200 g/L) and reduced catalyst loading compared to wild-type strains.
Q: Does this process require expensive transition metal catalysts?
A: No, this biocatalytic method completely eliminates the need for precious metal catalysts like rhodium or ruthenium, relying instead on renewable enzymatic systems under mild aqueous conditions.
Q: What level of stereoselectivity can be achieved with this method?
A: The process consistently achieves enantiomeric excess (ee) values greater than 99%, ensuring high optical purity essential for pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic strategies described in patent CN114807246A and are fully equipped to leverage this technology for your specific project needs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are staffed by expert chemists and engineers who specialize in optimizing enzymatic processes, maintaining stringent purity specifications, and operating rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to explore how this advanced ketoreductase technology can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality chiral alcohols for the global market.
