Advanced Synthesis of 5-Cyanophthalide: A Strategic Upgrade for Antidepressant API Manufacturing
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of critical antidepressant intermediates, particularly for high-volume drugs like Citalopram. Patent CN1331686A introduces a transformative methodology for the preparation of 5-cyanophthalide, a pivotal building block in the synthesis of this widely prescribed selective serotonin reuptake inhibitor (SSRI). Unlike historical approaches that relied on hazardous heavy metal chemistry, this invention leverages a convenient and cost-effective dehydration strategy starting from 5-carboxyphthalide. By converting the carboxylic acid precursor into an amide intermediate followed by a controlled dehydration step, the process achieves high yields while maintaining exceptional chemical purity. This technical breakthrough addresses long-standing challenges in impurity control and operational safety, positioning it as a preferred route for modern API manufacturing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-cyanophthalide and related nitriles often depended on classical Sandmeyer-type reactions or similar diazotization protocols involving copper cyanide. These traditional methods present significant drawbacks for large-scale industrial application, primarily due to the generation of toxic heavy metal waste streams that require expensive and complex remediation procedures. The use of copper salts not only poses environmental compliance risks but also complicates the purification process, as residual metal traces must be rigorously removed to meet stringent pharmaceutical quality standards. Furthermore, diazotization reactions are inherently unstable and exothermic, requiring precise temperature control and specialized equipment to mitigate safety hazards. The cumulative effect of these factors results in elevated production costs, extended cycle times, and a larger environmental footprint, making conventional routes increasingly unattractive for cost-sensitive generic drug manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent offers a streamlined alternative that bypasses the need for transition metal catalysts entirely. The core innovation lies in the direct conversion of 5-carboxyphthalide to an amide derivative, which is subsequently dehydrated using reagents such as thionyl chloride (SOCl2) or phosphorus oxychloride (POCl3). This approach utilizes readily available commodity chemicals that are easy to source and handle on a commercial scale. The reaction conditions are mild yet effective, typically proceeding in common organic solvents like toluene or acetonitrile at reflux temperatures. By avoiding the formation of diazonium salts and copper complexes, the new route drastically simplifies the workup procedure, often requiring only filtration and recrystallization to achieve high-purity product. This shift represents a fundamental improvement in process chemistry, aligning with green chemistry principles by reducing waste and enhancing atom economy.
Mechanistic Insights into Amide Dehydration Cyclization
The chemical elegance of this process is rooted in the selective dehydration of the amide functionality without compromising the sensitive lactone ring present in the phthalide structure. The mechanism typically initiates with the activation of the amide oxygen by the dehydrating agent, such as thionyl chloride, forming a reactive imidoyl chloride intermediate. This activated species then undergoes elimination of hydrogen chloride to generate the carbon-nitrogen triple bond of the nitrile group. Crucially, the reaction parameters are tuned to ensure chemoselectivity; the lactone carbonyl, which could theoretically react with chlorinating agents, remains intact under the specified conditions. This selectivity is vital for maintaining the structural fidelity of the intermediate, as ring-opening or side reactions would lead to difficult-to-remove impurities that could compromise the efficacy of the final antidepressant drug.
Experimental evidence from the patent highlights the robustness of this mechanistic pathway, demonstrating that substantially no reaction occurs on the lactone ring even under prolonged heating. The use of catalytic amounts of N,N-dimethylformamide (DMF) further enhances the reactivity of the dehydrating agent, facilitating the formation of the Vilsmeier-Haack type active species which accelerates the dehydration rate. This catalytic effect allows the reaction to proceed efficiently at lower stoichiometric ratios of the dehydrating agent, typically between 1.0 to 1.5 equivalents. The result is a clean transformation that minimizes the formation of by-products, thereby reducing the burden on downstream purification units and ensuring a consistent impurity profile suitable for regulatory submission.
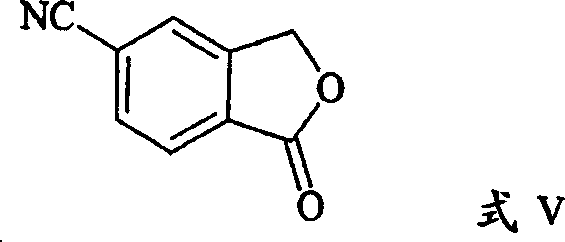
How to Synthesize 5-Cyanophthalide Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to maximize yield and purity. The process generally begins with the activation of the carboxylic acid, followed by amidation and final dehydration. Each step is designed to be operationally simple, utilizing standard reactor configurations found in most fine chemical facilities. The following guide outlines the critical operational phases derived from the preferred embodiments of the patent, providing a framework for process engineers to establish robust manufacturing protocols.
- Convert 5-carboxyphthalide to an acid chloride using thionyl chloride (SOCl2) with a catalytic amount of DMF in toluene under reflux.
- React the resulting acid chloride with ammonia or a C1-6 alkylamine (e.g., tert-butylamine) to form the corresponding amide intermediate.
- Dehydrate the amide intermediate using a dehydrating agent such as SOCl2, POCl3, or PCl5 in a solvent like toluene or acetonitrile to yield 5-cyanophthalide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of copper-based catalysts removes a significant variable from the raw material sourcing matrix, reducing exposure to volatile metal markets and complex waste disposal contracts. Furthermore, the reliance on common solvents and dehydrating agents ensures a stable supply base, minimizing the risk of production stoppages due to specialty chemical shortages. The simplified isolation procedure, which often involves straightforward crystallization, translates directly into reduced processing time and lower utility consumption per kilogram of product. These factors collectively contribute to a more resilient and cost-efficient supply chain for antidepressant intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive and toxic heavy metal catalysts from the process flow. By eliminating the need for copper cyanide and the associated heavy metal scavenging steps, manufacturers can achieve substantial cost savings in both raw material procurement and waste treatment operations. The simplified workup procedure reduces the consumption of solvents and energy required for extensive purification, leading to a lower overall cost of goods sold (COGS). Additionally, the high yields reported in the patent examples indicate efficient raw material utilization, further enhancing the economic viability of the process for high-volume production.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as thionyl chloride, phosphorus oxychloride, and toluene, are commodity chemicals produced by a wide range of global suppliers. This broad availability mitigates the risk of supply disruptions that often plague processes dependent on niche or single-source catalysts. The robustness of the reaction conditions also allows for flexibility in manufacturing scheduling, as the process is less sensitive to minor variations in temperature or reagent grade compared to sensitive organometallic reactions. This reliability ensures consistent delivery timelines for downstream API manufacturers, supporting just-in-time inventory strategies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a cleaner profile that facilitates easier regulatory approval and permitting for manufacturing sites. The absence of cyanide salts and heavy metals significantly reduces the toxicity of the effluent stream, lowering the cost and complexity of wastewater treatment. The process is inherently scalable, as demonstrated by the use of standard reflux and filtration techniques that translate seamlessly from pilot plant to multi-ton commercial reactors. This scalability ensures that the supply of 5-cyanophthalide can be rapidly ramped up to meet surging market demand for generic citalopram without requiring capital-intensive process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this chemistry into their existing manufacturing portfolios.
Q: What are the primary advantages of this new method over traditional Sandmeyer reactions?
A: The novel method described in CN1331686A eliminates the need for toxic copper cyanide (CuCN) and hazardous diazotization steps. This significantly reduces heavy metal contamination risks, simplifies downstream purification, and improves overall process safety and environmental compliance compared to conventional routes.
Q: How does the process ensure the integrity of the lactone ring during dehydration?
A: The process utilizes specific dehydrating agents like thionyl chloride under controlled conditions that exhibit high chemoselectivity. Experimental data confirms that substantially no reaction occurs on the sensitive lactone ring, ensuring the structural integrity of the phthalide core while efficiently converting the amide to the nitrile group.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable. It relies on readily available industrial reagents such as thionyl chloride and common solvents like toluene. The workup involves simple filtration and crystallization steps, avoiding complex chromatographic separations, which makes it ideal for multi-ton manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Cyanophthalide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 5-cyanophthalide with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific reagents and conditions required by this dehydration route, guaranteeing a consistent and reliable supply for your antidepressant API manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can support your long-term production goals and enhance your competitive position in the pharmaceutical market.
