Advanced Synthesis of 5-Cyanophthalide: Technical Upgrade and Commercial Scalability for Global Pharma
The pharmaceutical industry continuously seeks robust synthetic routes for critical antidepressant intermediates, and patent CN1423643A presents a significant technological advancement in the preparation of 5-cyanophthalide. This compound serves as a pivotal building block in the manufacturing of citalopram, a widely prescribed selective serotonin reuptake inhibitor. The disclosed method introduces a novel dehydration strategy that bypasses traditional hazardous cyanation steps, offering a cleaner and more efficient pathway for production. By leveraging oxazoline or thiazoline intermediates, this process addresses long-standing challenges related to toxicity and waste management in fine chemical synthesis. For R&D directors and procurement specialists, understanding this technology is crucial for optimizing supply chains and ensuring the consistent availability of high-purity pharmaceutical intermediates. The shift away from heavy metal catalysts represents a meaningful step towards greener chemistry without compromising on yield or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-cyanophthalide relied heavily on diazotization reactions followed by treatment with copper cyanide, a process fraught with significant operational and safety hazards. The use of copper cyanide introduces severe toxicity concerns, requiring stringent safety protocols and specialized waste treatment facilities to handle cyanide-containing effluents. Furthermore, the diazotization step is inherently unstable and sensitive to reaction conditions, often leading to variable yields and the formation of difficult-to-remove impurities. These conventional methods also involve multiple reduction steps starting from 4-aminophthalimide, which elongates the production timeline and increases the overall cost of goods. The accumulation of heavy metal residues in the final product necessitates additional purification stages, further driving up manufacturing expenses and complicating regulatory compliance for API production. Consequently, reliance on these outdated techniques poses substantial risks to supply chain continuity and environmental sustainability.
The Novel Approach
In contrast, the method described in patent CN1423643A utilizes a dehydration reaction of 2-(1-oxo-1,3-dihydroisobenzofuran-5-yl)oxazoline or thiazoline intermediates to directly form the nitrile group. This innovative route eliminates the need for toxic copper cyanide reagents, thereby drastically simplifying the waste treatment process and reducing the environmental footprint of the manufacturing facility. The reaction conditions are milder and more controllable, utilizing common dehydrating agents such as thionyl chloride in the presence of catalytic amounts of tertiary amides. This approach not only improves the safety profile of the operation but also enhances the overall yield by minimizing side reactions associated with radical cyanation. By streamlining the synthetic sequence, manufacturers can achieve cost reduction in API manufacturing while maintaining high standards of product quality. The versatility of this method allows for the use of various substituents on the oxazoline ring, providing flexibility for derivative synthesis.
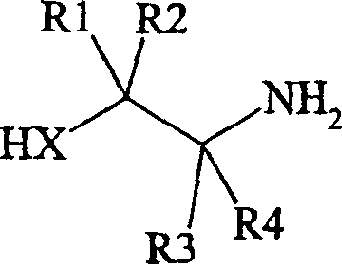
Mechanistic Insights into SOCl2-Catalyzed Dehydration
The core of this technological breakthrough lies in the efficient conversion of the oxazoline or thiazoline ring into a nitrile functionality through a dehydration mechanism. When using thionyl chloride as the dehydrating agent, the reaction likely proceeds via the activation of the hydroxyl or thiol group within the heterocyclic ring, facilitating the elimination of water or hydrogen sulfide. The presence of a catalytic amount of N,N-dimethylformamide (DMF) generates a Vilsmeier-type reagent in situ, which significantly enhances the electrophilicity of the dehydrating agent and accelerates the reaction rate. This catalytic cycle ensures that the transformation occurs under relatively mild thermal conditions, typically involving reflux in solvents like toluene or acetonitrile. The mechanistic pathway avoids the formation of free radical species that are common in copper-catalyzed cyanations, thereby reducing the generation of tarry by-products and simplifying downstream purification. Understanding this mechanism is vital for process chemists aiming to replicate and optimize the reaction for large-scale production environments.
Impurity control is another critical aspect where this novel mechanism offers distinct advantages over traditional methods. By avoiding the use of copper salts, the process eliminates the risk of heavy metal contamination, which is a major concern for regulatory agencies governing pharmaceutical ingredients. The dehydration reaction is highly selective, primarily targeting the specific functional groups required for nitrile formation without affecting other sensitive moieties on the benzofuranone core. This selectivity results in a cleaner crude product profile, reducing the burden on crystallization and chromatography steps during purification. Furthermore, the thermal stability of the intermediates allows for robust process control, minimizing the formation of degradation products that could compromise the safety of the final antidepressant drug. For quality assurance teams, this means more consistent batch-to-batch reproducibility and easier compliance with stringent purity specifications required for global markets.
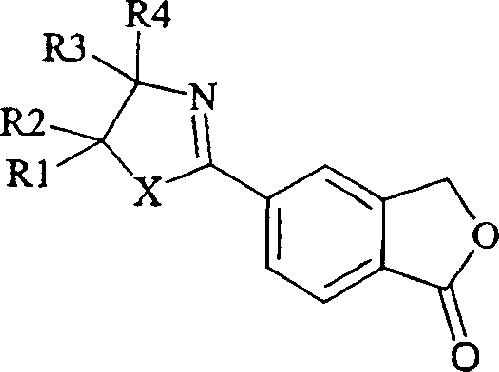
How to Synthesize 5-Cyanophthalide Efficiently
The practical implementation of this synthesis route involves a straightforward sequence that begins with the preparation of the key oxazoline intermediate from readily available carboxy-benzofuranone derivatives. Once the intermediate is secured, the dehydration step is performed using thionyl chloride in a suitable organic solvent, often with the addition of a catalytic amide to drive the reaction to completion. The reaction mixture is then quenched, and the product is isolated through filtration and washing, followed by optional recrystallization to achieve the desired purity levels. Detailed standard operating procedures for this transformation are essential for ensuring safety and consistency during scale-up. The following guide outlines the critical steps for executing this synthesis effectively in a laboratory or pilot plant setting.
- Prepare the oxazoline intermediate by reacting 5-carboxy-2-benzo[c]furanone derivatives with 2-hydroxyethylamine.
- Treat the isolated oxazoline intermediate with a dehydrating agent such as thionyl chloride (SOCl2) in the presence of a catalytic amide.
- Quench the reaction mixture, filter the solid product, and purify via recrystallization to achieve high-purity 5-cyanophthalide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patent-protected synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their vendor networks. The elimination of copper cyanide not only reduces the cost associated with hazardous waste disposal but also mitigates the regulatory risks linked to handling toxic materials. This shift allows for a more streamlined manufacturing process that is less dependent on specialized containment equipment, thereby lowering capital expenditure requirements for production facilities. Additionally, the use of common reagents like thionyl chloride and toluene ensures that raw material sourcing is stable and less susceptible to market volatility compared to specialized cyanation agents. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding timelines of the pharmaceutical industry.
- Cost Reduction in Manufacturing: The removal of expensive and toxic copper cyanide reagents significantly lowers the raw material costs associated with the synthesis of 5-cyanophthalide. By simplifying the reaction sequence and reducing the number of purification steps required to remove heavy metal residues, manufacturers can achieve substantial cost savings in overall production. The higher yields reported in the patent examples further contribute to economic efficiency, ensuring that more product is obtained from the same amount of starting material. This economic advantage is critical for maintaining competitive pricing in the global market for antidepressant intermediates while preserving profit margins.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as thionyl chloride and organic amines enhances the reliability of the supply chain by reducing dependency on niche suppliers. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages or logistical bottlenecks. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing across different geographic locations, supporting a diversified sourcing strategy. For supply chain heads, this means greater confidence in meeting delivery commitments and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor equipment and solvent systems that are common in the fine chemical industry. The absence of heavy metals simplifies the environmental compliance process, making it easier to obtain necessary permits and maintain operational licenses. This environmental compatibility aligns with the growing corporate demand for sustainable manufacturing practices, enhancing the brand reputation of companies that adopt this technology. The ability to scale up complex pharmaceutical intermediates without significant process redesign ensures a smooth transition from laboratory development to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 5-cyanophthalide based on the disclosed patent data. These answers are derived from the specific experimental conditions and beneficial effects described in the documentation, providing clarity for stakeholders evaluating this technology. Understanding these details is essential for making informed decisions about process adoption and supplier selection. The information below reflects the current state of the art as presented in the patent literature.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel process eliminates the use of toxic copper cyanide (CuCN) and complex diazotization steps, resulting in a cleaner impurity profile and reduced environmental burden compared to traditional methods described in older literature.
Q: What dehydrating agents are suitable for this transformation?
A: The patent specifies several effective dehydrating agents including thionyl chloride (SOCl2), phosphorus oxychloride (POCl3), and phosphorus pentachloride (PCl5), with SOCl2 being the most preferred for optimal yield and convenience.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes robust reagents and standard solvent systems like toluene and acetonitrile, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Cyanophthalide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced synthetic methodologies like the one described in CN1423643A to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-cyanophthalide meets the highest industry standards. By integrating innovative dehydration technologies, we offer a reliable pharmaceutical intermediate supplier solution that balances cost efficiency with uncompromising quality.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational improvements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements, demonstrating how our advanced processes can enhance your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Together, we can drive innovation and efficiency in the production of critical antidepressant intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
